Abstract
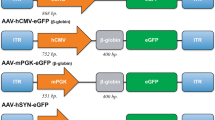
The neurogenetic, lysosomal enzyme (LSE) deficiency diseases are characterized by storage lesions throughout the brain; therefore, gene transfer needs to provide widespread distribution of the normal enzyme. Adeno-associated virus (AAV) vectors can be effective in the brain despite limited transduction because LSEs are exported to neighboring cells (cross-correction) to reverse the metabolic deficit. The extent of correction is determined by a combination of the total amount of LSE produced by a vector and the spatial distribution of the vector within the brain. Neuron-specific promoters have been used in the brain because AAV predominantly transduces neurons. However, these promoters are large, using up a substantial amount of the limited cloning capacity of AAV vector genomes. A small promoter that is active in all cells, from the LSE β-glucuronidase (GUSB), has been used for long-term expression in AAV vectors in the brain but the natural promoter is expressed at very low levels. The amount of LSE exported from a cell is proportional to the level of transcription, thus more active promoters would export more LSE for cross-correction, but direct comparisons have not been reported. In this study, we show that in long-term experiments (>6 months) the GUSB minimal promoter (hGBp) expresses the hGUSB enzyme in brain at similar levels as the neuron-specific enolase promoter or the promoter from the latency-associated transcript of herpes simplex virus. The hGBp minimal promoter thus may be useful for long-term expression in the central nervous system of large cDNAs, bicitronic transcription units, self-complimentary or other designs with size constraints in the AAV vector system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pierson TM, Wolfe JH . Gene therapy for inherited diseases of the central nervous system. In: Lynch D (ed). Neurogenetics: Scientific and Clinical Advances. Marcel Dekker, Inc.: New York, NY, 2005, pp 43–85.
Mandel RJ, Manfredsson FP, Foust KD, Rising A, Reimsnider S, Nash K et al. Recombinant adeno-associated viral vectors as therapeutic agents to treat neurological disorders. Mol Ther 2006; 13: 463–483.
Taylor RM, Wolfe JH . Decreased lysosomal storage in the adult MPS VII mouse brain in the vicinity of grafts of retroviral vector-corrected fibroblasts secreting high levels of β-glucuronidase. Nat Med 1997; 3: 771–774.
Passini MA, Lee EB, Heuer GG, Wolfe JH . Distribution of a lysosomal enzyme in the adult brain by axonal transport and by cells of the rostral migratory stream. J Neurosci 2002; 22: 6437–6446.
Cearley CN, Wolfe JH . A single injection of an adeno-associated virus vector into nuclei with divergent connections results in widespread vector distribution in the brain and global correction of a neurogenetic disease. J Neurosci 2007; 27: 9928–9940.
Wolfe JH, Kyle JW, Sands MS, Sly WS, Markowitz DG, Parente MK . High level expression and export of beta-glucuronidase from murine mucopolysaccharidosis VII cells corrected by a double-copy retrovirus vector. Gene Therapy 1995; 2: 70–78.
Frisella WA, O'Connor LH, Vogler CA, Roberts M, Walkley S, Levy B et al. Intracranial injection of recombinant adeno-associated virus improves cognitive function in a murine model of mucopolysaccharidosis type VII. Mol Ther 2001; 3: 351–358.
Berges BK, Wolfe JH, Fraser NW . Transduction of brain by herpes simplex virus vectors. Mol Ther 2007; 15: 20–29.
Zwaagstra JC, Ghiasi H, Slanina SM, Nesburn AB, Wheatley SC, Lillycrop K et al. Activity of herpes simplex virus type 1 latency-associated transcript (LAT) promoter in neuron-derived cells: evidence for neuron specificity and for a large LAT transcript. J Virol 1990; 64: 5019–5028.
Fu H, Samulski RJ, McCown TJ, Picornell YJ, Fletcher D, Muenzer J . Neurological correction of lysosomal storage in a mucopolysaccharidosis IIIB mouse model by adeno-associated virus-mediated gene delivery. Mol Ther 2002; 5: 42–49.
Xu R, Janson CG, Mastakov M, Lawlor P, Young D, Mouravlev A et al. Quantitative comparison of expression with adeno-associated virus (AAV-2) brain-specific gene cassettes. Gene Therapy 2001; 8: 1323–1332.
Peel AL, Zolotukhin S, Schrimsher GW, Muzyczka N, Reier PJ . Efficient transduction of green fluorescent protein in spinal cord neurons using adeno-associated virus vectors containing cell type-specific promoters. Gene Therapy 1997; 4: 16–24.
Shipley JM, Miller RD, Wu BM, Grubb JH, Christensen SG, Kyle JW et al. Analysis of the 5′ flanking region of the human beta-glucuronidase gene. Genomics 1991; 10: 1009–1018.
Kyle JW, Birkenmeier EH, Gwynn B, Vogler C, Hoppe PC, Hoffmann JW et al. Correction of murine mucopolysaccharidosis VII by a human beta-glucuronidase transgene. Proc Natl Acad Sci USA 1990; 87: 3914–3918.
Passini MA, Wolfe JH . Widespread gene delivery and structure-specific patterns of expression in the brain after intraventricular injections of neonatal mice with an adeno-associated virus vector. J Virol 2001; 75: 12382–12392.
Prasad Alur RK, Foley B, Parente MK, Tobin DK, Heuer GG, Avadhani AN et al. Modification of multiple transcriptional regulatory elements in a Moloney murine leukemia virus gene transfer vector circumvents silencing in fibroblast grafts and increases levels of expression of the transferred enzyme. Gene Therapy 2002; 9: 1146–1154.
Watson DJ, Kobinger GP, Passini MA, Wilson JM, Wolfe JH . Targeted transduction patterns in the mouse brain by lentivirus vectors pseudotyped with VSV, Ebola, Mokola, LCMV, or MuLV envelope proteins. Mol Ther 2002; 5: 528–537.
Watson G, Paigen K . Genetic variations in kinetic constants that describe beta-glucuronidase mRNA induction in androgen-treated mice. Mol Cell Biol 1987; 7: 1085–1090.
Vite CH, Passini MA, Haskins ME, Wolfe JH . Adeno-associated virus vector-mediated transduction in the cat brain. Gene Therapy 2003; 10: 1874–1881.
Cearley CN, Wolfe JH . Transduction characteristics of adeno-associated virus vectors expressing cap serotypes 7, 8, 9, and Rh10 in the mouse brain. Mol Ther 2006; 13: 528–537.
Cearley CN, Vandenberghe LH, Parente MK, Carnish ER, Wilson JM, Wolfe JH . Expanded repertoire of AAV vector serotypes mediate unique patterns of transduction in mouse brain. Mol Ther 2008; 16: 1710–1718.
Berges BK, Wolfe JH, Fraser NW . Stable levels of long-term transgene expression driven by the latency-associated transcript promoter in a herpes simplex virus type 1 vector. Mol Ther 2005; 12: 1111–1119.
Passini MA, Watson DJ, Vite CH, Landsburg DJ, Feigenbaum AL, Wolfe JH . Intraventricular brain injection of adeno-associated virus type 1 (AAV1) in neonatal mice results in complementary patterns of neuronal transduction to AAV2 and total long-term correction of storage lesions in the brains of beta-glucuronidase-deficient mice. J Virol 2003; 77: 7034–7040.
Wang C, Wang CM, Clark KR, Sferra TJ . Recombinant AAV serotype 1 transduction efficiency and tropism in the murine brain. Gene Therapy 2003; 10: 1528–1534.
Burger C, Gorbatyuk OS, Velardo MJ, Peden CS, Williams P, Zolotukhin S et al. Recombinant AAV viral vectors pseudotyped with viral capsids from serotypes 1, 2, and 5 display differential efficiency and cell tropism after delivery to different regions of the central nervous system. Mol Ther 2004; 10: 302–317.
Skorupa AF, Fisher KJ, Wilson JM, Parente MK, Wolfe JH . Sustained production of beta-glucuronidase from localized sites after AAV vector gene transfer results in widespread distribution of enzyme and reversal of lysosomal storage lesions in a large volume of brain in mucopolysaccharidosis VII mice. Exp Neurol 1999; 160: 17–27.
Karolewski BA, Wolfe JH . Genetic correction of the fetal brain increases the lifespan of mice with the severe multisystemic disease mucopolysaccharidosis type VII. Mol Ther 2006; 14: 14–24.
Luca T, Givogri MI, Perani L, Galbiati F, Follenzi A, Naldini L et al. Axons mediate the distribution of arylsulfatase A within the mouse hippocampus upon gene delivery. Mol Ther 2005; 12: 669–679.
Hennig AK, Levy B, Ogilvie JM, Vogler CA, Galvin N, Bassnett S et al. Intravitreal gene therapy reduces lysosomal storage in specific areas of the CNS in mucopolysaccharidosis VII mice. J Neurosci 2003; 23: 3302–3307.
Smith C, Lachmann RH, Efstathiou S . Expression from the herpes simplex virus type 1 latency-associated promoter in the murine central nervous system. J Gen Virol 2000; 81: 649–662.
Chen X, Schmidt MC, Goins WF, Glorioso JC . Two herpes simplex virus type 1 latency-active promoters differ in their contributions to latency-associated transcript expression during lytic and latent infections. J Virol 1995; 69: 7899–7908.
Lokensgard JR, Berthomme H, Feldman LT . The latency-associated promoter of herpes simplex virus type 1 requires a region downstream of the transcription start site for long-term expression during latency. J Virol 1997; 71: 6714–6719.
Berthomme H, Thomas J, Texier P, Epstein A, Feldman LT . Enhancer and long-term expression functions of herpes simplex virus type 1 latency-associated promoter are both located in the same region. J Virol 2001; 75: 4386–4393.
Raol YH, Lund IV, Bandyopadhyay S, Zhang G, Roberts DS, Wolfe JH et al. Enhancing GABA(A) receptor alpha 1 subunit levels in hippocampal dentate gyrus inhibits epilepsy development in an animal model of temporal lobe epilepsy. J Neurosci 2006; 26: 11342–11346.
Heuer GG, Passini MA, Jiang K, Parente MK, Lee VM, Trojanowski JQ et al. Selective neurodegeneration in the murine mucopolysaccharidosis VII is progressive and reversible. Ann Neurol 2002; 52: 762–770.
Wolfe JH, Sands MS, Harel N, Weil MA, Parente MK, Polesky AC et al. Gene transfer of low levels of beta-glucuronidase corrects hepatic lysosomal storage in a large animal model of mucopolysaccharidosis VII. Mol Ther 2000; 2: 552–561.
Acknowledgements
We thank A Polesky, E Cabacungan and T Clarke for excellent technical assistance. This work was supported by NIH grants R01-NS038690 (JHW) and R01-NS029390 (NWF). TH and MAP were partially supported by NIH training grant T32-DK007748.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Gene Therapy website (http://www.nature.com/gt)
Rights and permissions
About this article
Cite this article
Husain, T., Passini, M., Parente, M. et al. Long-term AAV vector gene and protein expression in mouse brain from a small pan-cellular promoter is similar to neural cell promoters. Gene Ther 16, 927–932 (2009). https://doi.org/10.1038/gt.2009.52
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2009.52
Keywords
This article is cited by
-
Quantitative, noninvasive, in vivo longitudinal monitoring of gene expression in the brain by co-AAV transduction with a PET reporter gene
Molecular Therapy - Methods & Clinical Development (2014)
-
Distinct transduction profiles in the CNS via three injection routes of AAV9 and the application to generation of a neurodegenerative mouse model
Molecular Therapy - Methods & Clinical Development (2014)
-
Progress in gene therapy for neurological disorders
Nature Reviews Neurology (2013)
-
In utero administration of Ad5 and AAV pseudotypes to the fetal brain leads to efficient, widespread and long-term gene expression
Gene Therapy (2012)