Abstract
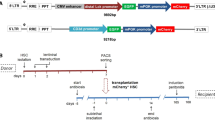
B-lymphocytes play a key role in the pathogenesis of many immune-mediated diseases, such as autoimmune and atopic diseases. Therefore, targeting B-lymphocytes provides a rationale for refining strategies to treat such diseases for long-term clinical benefits and minimal side effects. In this study we describe a protocol for repopulating irradiated mice with B-lymphocytes engineered for restricted expression of transgenes using haematopoietic stem cells. A self-inactivating lentiviral vector, which encodes enhanced green fluorescence protein (EGFP) from the spleen focus-forming virus (SFFV) promoter, was used to generate new vectors that permit restricted EGFP expression in B-lymphocytes. To achieve this, the SFFV promoter was replaced with the B-lymphocyte-restricted CD19 promoter. Further, an immunoglobulin heavy chain enhancer (Eμ) flanked by the associated matrix attachment regions (MARs) was inserted upstream of the CD19 promoter. Incorporation of the Eμ-MAR elements upstream of the CD19 promoter resulted in enhanced, stable and selective transgene expression in human and murine B-cell lines. In addition, this modification permitted enhanced selective EGFP expression in B-lymphocytes in vivo in irradiated mice repopulated with transduced bone marrow haematopoietic stem cells (BMHSCs). The study provides evidence for the feasibility of targeting B-lymphocytes for therapeutic restoration of normal B-lymphocyte functions in patients with B-cell-related diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Browning JL . B cells move to centre stage: novel opportunities for autoimmune disease treatment. Nat Rev Drug Discov 2006; 5: 564–576.
Cunningham-Rundles C, Ponda PP . Molecular defects in T- and B-cell primary immunodeficiency diseases. Nat Rev Immunol 2005; 11: 880–892.
Mageed RA, Prud'homme GJ . Immunopathology and the gene therapy of lupus. Gene Therapy 2003; 10: 861–874.
Youinou P, Jamin C, Pers JO, Berthou C, Saraux A, Renaudineau Y . B lymphocytes are required for development and treatment of autoimmune diseases. Ann NY Acad Sci 2005; 1050: 19–33.
Ng KP, Cambridge G, Leandro MJ, Edwards JC, Ehrenstein M, Isenberg DA . B cell depletion therapy in systemic lupus erythematosus: long term follow up and predictors of response. Ann Rheum Dis 2007; 66: 1259–1262.
Zouali M, Sarmay G . B lymphocyte signaling pathways in systemic autoimmunity: implications for pathogenesis and treatment. Arthritis Rheum 2004; 50: 1259–1262.
Nimmerjahn F, Ravetch JV . Fcgamma receptors: old friends and new family members. Immunity 2006; 24: 19–28.
Yan J, Harvey BP, Gee RJ, Shlomchik MJ, Mamula MJ . B cells drive early T cell autoimmunity in vivo prior to dendritic cell-mediated autoantigen presentation. J Immunol 2006; 177: 4481–4487.
Plosker GL, Figgitt DP . Rituximab: a review of its use in non-Hodgkin's lymphoma and chronic lymphocytic leukaemia. Drugs 2003; 63: 803–843.
Perceau G, Diris N, Estines O, Derancourt C, Lévy S, Bernard P . Late lethal hepatitis B virus reactivation after rituximab treatment of low-grade cutaneous B-cell lymphoma. Br J Dermatol 2006; 155: 1053–1056.
Messmer D, Kipps TJ . CD154 gene therapy for human B-cell malignancies. Ann NY Acad Sci 2005; 1062: 51–60.
Morgan RA, Dudley ME, Wunderlich JR, Hughes MS, Yang JC, Sherry RM et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science 2006; 314: 126–129.
Cavazzana-Calvo M, Lagresle C, Hacein-Bey-Abina S, Fischer A . Gene therapy for severe combined immunodeficiency. Annu Rev Med 2005; 56: 585–602.
Aiuti A, Slavin S, Aker M, Ficara F, Deola S, Mortellaro A et al. Correction of ADA-SCID by stem cell gene therapy combined with nonmyeloablative conditioning. Science 2002; 296: 2410–2413.
Zufferey R, Dull T, Mandel RJ, Bukovsky A, Quiroz D, Naldini L et al. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery. J Virol 1998; 72: 9873–9880.
May C, Rivella S, Chadburn A, Sadelain M . Successful treatment of murine beta-thalassemia intermedia by transfer of the human beta-globin gene. Blood 2002; 99: 1902–1908.
Kowolik CM, Hu J, Yee JK . Locus control region of the human CD2 gene in a lentivirus vector confers position-independent transgene expression. J Virol 2001; 75: 4641–4648.
Cui Y, Golob J, Kelleher E, Ye Z, Pardoll D, Cheng L . Targeting transgene expression to antigen-presenting cells derived from lentivirus-transduced engrafting human hematopoietic stem/progenitor cells. Blood 2002; 99: 399–408.
May C, Rivella S, Callegari J, Hellern G, Gaensler KM, Luzzatto L et al. Therapeutic haemoglobin synthesis in beta-thalassaemic mice expressing lentivirus-encoded human beta-globin. Nature 2000; 406: 82–86.
Ellis J, Pannell D . The beta-globin locus control region versus gene therapy vectors: a struggle for expression. Clin Genet 2001; 59: 17–24.
Werner M, Kraunus J, Baum C, Brocker T . B-cell-specific transgene expression using a self-inactivating retroviral vector with human CD19 promoter and viral post transcriptional regulatory element. Gene Therapy 2004; 11: 992–1000.
Adams JM, Harris AW, Pinkert CA, Corcoran LM, Alexander WS, Cory S et al. The c-myc oncogene driven by immunoglobulin enhancers induces lymphoid malignancy in transgenic mice. Nature 1985; 318: 533–538.
Cockerill PN, Yuen MH, Garrard WT . The enhancer of the immunoglobulin heavy chain locus is flanked by presumptive chromosomal loop anchorage elements. J Biol Chem 1987; 262: 5394–5397.
Lutzko C, Senadheera D, Skelton D, Petersen D, Kohn DB . Lentivirus vectors incorporating the immunoglobulin heavy chain enhancer and matrix attachment regions provide position-independent expression in B lymphocytes. J Virol 2003; 77: 7341–7351.
Jenuwein T, Forrester WC, Fernandez-Herrero LA, Laible G, Dull M, Grosschedl R . Extension of chromatin accessibility by nuclear matrix attachment regions. Nature 1997; 385: 269–272.
Fernandez LA, Winkler M, Grosschedl R . Matrix attachment region-dependent function of the immunoglobulin mu enhancer involves histone acetylation at a distance without changes in enhancer occupancy. Mol Cell Biol 2001; 21: 196–208.
Herrscher RF, Kaplan MH, Lelsz DL, Das C, Scheuermann R, Tucker PW . The immunoglobulin heavy-chain matrix-associating regions are bound by Bright: a B cell-specific trans-activator that describes a new DNA-binding protein family. Genes Dev 1995; 9: 3067–3082.
Dickinson LA, Joh T, Kohwi Y, Kohwi-Shigematsu T . A tissue-specific MAR/SAR DNA-binding protein with unusual binding site recognition. Cell 1992; 70: 631–645.
Miyoshi H, Blomer U, Takahashi M, Gage FH, Verma IM . Development of a self-inactivating lentivirus vector. J Virol 1998; 72: 8150–8157.
Villemure JF, Savard N, Belmaaza A . Promoter suppression in cultured mammalian cells can be blocked by the chicken beta-globin chromatin insulator 5′HS4 and matrix/scaffold attachment regions. J Mol Biol 2001; 312: 963–974.
Kulbe H, Thompson R, Wilson JL, Robinson S, Hagemann T, Fatah R et al. The inflammatory cytokine tumor necrosis factor-alpha generates an autocrine tumor-promoting network in epithelial ovarian cancer cells. Cancer Res 2007; 67: 585–592.
Acknowledgements
This study was supported by grant number RAB/PJ/05 from the St Bartholomew's and the Royal London Charitable Foundation Research Advisory Board. DJ Gould was funded by Arthritis Research Campaign (ARC). FD'Acquisto was supported by a New Investigator Award Fellowship from the Medical Research Council (MRC). We are grateful for the expert advice of Professor Yuti Chernajovsky (Bone and Joint Research Unit, Queen Mary School of Medicine and Dentistry, London). We are also indebted to Dr Carolyn Lutzko (Childrens Hospital, SABAN Research Institute, Los Angeles, USA) and Professor Thomas Brocker (Ludwig-Maximilians-Universität, München, Germany) for providing the CCL-EMP-EGFP plasmid and the retroviral vector SIN-CD19-W, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Taher, T., Tulone, C., Fatah, R. et al. Repopulation of B-lymphocytes with restricted gene expression using haematopoietic stem cells engineered with lentiviral vectors. Gene Ther 15, 998–1006 (2008). https://doi.org/10.1038/gt.2008.33
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2008.33
Keywords
This article is cited by
-
Development of B-lineage Predominant Lentiviral Vectors for Use in Genetic Therapies for B Cell Disorders
Molecular Therapy (2011)
-
Advances in the Field of Lentivector-based Transduction of T and B Lymphocytes for Gene Therapy
Molecular Therapy (2010)
-
Incorporating double copies of a chromatin insulator into lentiviral vectors results in less viral integrants
BMC Biotechnology (2009)