Abstract
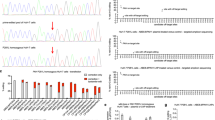
Novel recombinant adeno-associated virus vectors pseudotyped with serotype 8 capsid (rAAV2/8) have recently shown exciting promise as effective liver-directed gene transfer reagents. We have produced a novel liver-specific rAAV2/8 vector expressing the mouse phenylalanine hydroxylase (Pah) cDNA and have administered this vector to hyperphenylalaninemic PAH-deficient Pahenu2 mice, a model of human phenylketonuria (PKU). Our hypothesis was that this vector would produce sufficient hepatocyte transduction frequency and PAH activity to correct blood phenylalanine levels in murine PKU. Portal vein injection of recombinant AAV2/8 vector into five adult Pahenu2 mice yielded complete and stable (up to 17 weeks) correction of serum phenylalanine levels. Liver PAH activity was corrected to 11.5±2.4% of wild type liver activity and was associated with a significant increase in phenylalanine clearance following parenteral phenylalanine challenge. Although questions of long-term safety and stability of expression remain, recombinant AAV2/8-mediated, liver-directed gene therapy is a promising novel treatment approach for PKU and allied inborn errors of metabolism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Abbreviations
- AAV:
-
adeno-associated virus
- PKU:
-
phenylketonuria
- Phe:
-
phenylalanine
- PAH:
-
phenylalanine hydroxylase.
References
Scriver CR, Kaufman S . Hyperphenylalaninemia: phenylalanine hydroxylase deficiency. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The Metabolic & Molecular Bases of Inherited Disease. McGraw-Hill: New York, 2001, pp. 1667–1724.
Udenfriend S, Cooper JR . The enzymatic conversion of phenylalanine to tyrosine. J Biol Chem 1952; 194: 503–511.
Azen CG, Koch R, Friedman EG, Berlow S, Coldwell J, Krause W et al. Intellectual development in 12-year-old children treated for phenylketonuria. Am J Dis Child 1991; 145: 35–39.
Koch R, Hanley W, Levy H, Matalon K, Matalon R, Rouse B et al. The Maternal Phenylketonuria International Study: 1984–2002. Pediatrics 2003; 112: 1523–1529.
National Institutes of Health Consensus Development Conference Statement. Phenylketonuria: screening and management, October 16–18, 2000. Pediatrics 2001; 108: 972–982.
Ding Z, Harding CO, Thöny B . State-of-the-art 2003 on PKU gene therapy. Mol Genet Metab 2004; 81: 3–8.
McDonald JD, Bode VC, Dove WF, Shedlovsky A . Pahhph-5: a mouse mutant deficient in phenylalanine hydroxylase. Proc Natl Acad Sci USA 1990; 87: 1965–1967.
Liu T-J, Kay MA, Darlington GJ, Woo SLC . Reconstitution of enzymatic activity in hepatocytes of phenylalanine hydroxylase-deficient mice. Somat Cell Mol Genet 1992; 18: 89–96.
Fang B, Eisensmith RC, Li XHC, Finegold MJ, Shedlovsky A, Dove W et al. Gene therapy for phenylketonuria: phenotypic correction in a genetically deficient mouse model by adenovirus-mediated hepatic gene therapy. Gene Therapy 1994; 1: 247–254.
Laipis PJ, Reyes L, Embury JE, Alexander JJ, Hurt CB, Wein DA et al. Long term reduction of serum phenylalanine levels in a mouse model of PKU by rAAV-mediated gene therapy. Mol Ther 2001; 3: S293.
Oh HJ, Park ES, Kang S, Jo I, Jung SC . Long-term enzymatic and phenotypic correction in the phenylketonuria mouse model by adeno-associated virus vector-mediated gene transfer. Pediatr Res 2004; 56: 278–284.
Mochizuki S, Mizukami H, Ogura T, Kure S, Ichinohe A, Kojima K et al. Long-term correction of hyperphenylalaninemia by AAV-mediated gene transfer leads to behavioral recovery in phenylketonuria mice. Gene Therapy 2004; 11: 1081–1086.
Laipis PJ, Charron CE, Embury JE, Perera OP, Porvasnik SL, Fields CR et al. Correction of maternal phenylketonuria syndrome in the Pahenu2 missense mutant mouse by r-AAV mediated gene therapy. Mol Ther 2004; 9: S334.
Gao GP, Alvira MR, Wang L, Calcedo R, Johnston J, Wilson JM . Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc Natl Acad Sci USA 2002; 99: 11854–11859.
Sarkar R, Tetreault R, Gao G, Wang L, Bell P, Chandler R et al. Total correction of hemophilia A mice with canine FVIII using an AAV 8 serotype. Blood 2004; 103: 1253–1260.
Wang L, Takabe K, Bidlingmaier SM, Ill CR, Verma IM . Sustained correction of bleeding disorder in hemophilia B mice by gene therapy. Proc Natl Acad Sci USA 1999; 96: 3906–3910.
McDonald JD, Charlton CK . Characterization of mutations at the mouse phenylalanine hydroxylase locus. Genomics 1997; 39: 402–405.
Harding CO, Wild K, Chang D, Messing A, Wolff JA . Metabolic engineering as therapy for inborn errors of metabolism – development of mice with phenylalanine hydroxylase expression in muscle. Gene Therapy 1998; 5: 677–683.
McCaman MW, Robins E . Fluorimetric method for the determination of phenylalanine in serum. J Lab Clin Med 1962; 59: 885–890.
Nakai H, Fuess S, Storm TA, Muramatsu S, Nara Y, Kay MA . Unrestricted hepatocyte transduction with adeno-associated virus serotype 8 vectors in mice. J Virol 2005; 79: 214–224.
Miyamoto M, Fitzpatrick T . Competitive inhibition of mammalian tyrosinase by phenylalanine and its relationship to hair pigmentation in phenylketonuria. Nature 1957; 179: 199–200.
Laipis PJ, Charron CE, Ross K, Reyes L, Alexander JJ, Song S et al. Long-term correction of phenylketonuria in an animal model by recombinant AAV-based gene therapy. J Inher Metab Dis 2002; 25: 615–616.
Davidoff AM, Ng CY, Zhou J, Spence Y, Nathwani AC . Sex significantly influences transduction of murine liver by recombinant adeno-associated viral vectors through an androgen-dependent pathway. Blood 2003; 102: 480–488.
Hamman K, Clark H, Montini E, Al-Dhalimy M, Grompe M, Finegold M et al. Low therapeutic threshold for hepatocyte replacement in murine phenylketonuria. Mol Ther 2005; 12: 337–344.
Salter M, Knowles RG, Pogson CI . Transport of the aromatic amino acids into isolated rat liver cells. Properties of uptake by two distinct systems. Biochem J 1986; 233: 499–506.
Halbert CL, Allen JM, Miller AD . Adeno-associated virus type 6 (AAV6) vectors mediate efficient transduction of airway epithelial cells in mouse lungs compared to that of AAV2 vectors. J Virol 2001; 75: 6615–6624.
Rabinowitz JE, Rolling F, Li C, Conrath H, Xiao W, Xiao X et al. Cross-packaging of a single adeno-associated virus (AAV) type 2 vector genome into multiple AAV serotypes enables transduction with broad specificity. J Virol 2002; 76: 791–801.
Xiao W, Chirmule N, Berta SC, McCullough B, Gao G, Wilson JM . Gene therapy vectors based on adeno-associated virus type 1. J Virol 1999; 73: 3994–4003.
Ding Z, Georgier P, Thöny B . Administration-route and gender-independent long-term therapeutic correction of phenylketonuria (PKU) in a mouse model by recombinant adeno-associated virus 8 pseudotyped vector-mediated gene transfer. Gene Therapy 2005 [E-pub ahead of print: 17 November 2005; doi:10.1038/sj.gt.3302684].
Nakai H, Montini E, Fuess S, Storm TA, Grompe M, Kay MA . AAV serotype 2 vectors preferentially integrate into active genes in mice. Nat Genet 2003; 34: 297–302.
Miller DG, Rutledge EA, Russell DW . Chromosomal effects of adeno-associated virus vector integration. Nat Genet 2002; 30: 147–148.
Donsante A, Vogler C, Muzyczka N, Crawford JM, Barker J, Flotte T et al. Observed incidence of tumorigenesis in long-term rodent studies of rAAV vectors. Gene Therapy 2001; 8: 1343–1346.
Ledley FD, Hahn T, Woo SL . Selection for phenylalanine hydroxylase activity in cells transformed with recombinant retroviruses. Somat Cell Mol Genet 1987; 13: 145–154.
Acknowledgements
This work was supported by Doernbecher Children's Hospital Foundation, Portland, OR (COH). Dr Koeberl was supported by the Muscular Dystrophy Association and Genzyme Corporation. The authors would like to thank Drs Markus Grompe and David Koeller for critical reviews of the manuscript. Dr Harding would like to thank the many families affected by PKU, who provided constant encouragement and inspiration for this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harding, C., Gillingham, M., Hamman, K. et al. Complete correction of hyperphenylalaninemia following liver-directed, recombinant AAV2/8 vector-mediated gene therapy in murine phenylketonuria. Gene Ther 13, 457–462 (2006). https://doi.org/10.1038/sj.gt.3302678
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302678
Keywords
This article is cited by
-
Phenylketonuria
Nature Reviews Disease Primers (2021)
-
Phenylketonuria: Current Treatments and Future Developments
Drugs (2019)
-
Treatment of a metabolic liver disease by in vivo genome base editing in adult mice
Nature Medicine (2018)
-
Preclinical Dose-Finding Study With a Liver-Tropic, Recombinant AAV-2/8 Vector in the Mouse Model of Galactosialidosis
Molecular Therapy (2012)
-
In search of proof‐of‐concept: gene therapy for glycogen storage disease type Ia
Journal of Inherited Metabolic Disease (2012)