Abstract
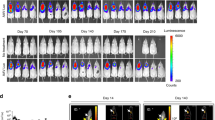
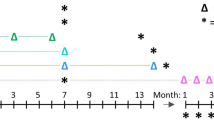
The development of a strategy to deliver a gene to pulmonary endothelium will be useful for gene function study and for pulmonary gene therapy. Cationic lipidic vectors are efficient in gene transfer to pulmonary endothelium via the vascular route; however, gene expression is transient and lasts for only a few days. In this study, we show that pulmonary gene transfer via cationic lipidic vectors can be significantly improved using an Epstein–Barr virus (EBV)-based expression plasmid. Systemic administration of cationic liposomes followed by the EBV-based plasmid led to gene expression in the lung that lasted for more than 3 weeks. Prolonged and high levels of gene expression can also be obtained in primary mouse lung endothelial cells (MLEC) following lipofection with an EBV-based plasmid. These results suggest the utility of this gene transfer protocol in studying the expression of cloned genes in lung endothelial cells and in pulmonary gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lemarchand P et al. In vivo adenovirus-mediated gene transfer to lungs via pulmonary artery. J Appl Physiol 1994; 76: 2840–2845.
Rodman DM et al. In vivo gene delivery to the pulmonary circulation in rats: transgene distribution and vascular inflammatory response. Am J Respir Cell Mol Biol 1997; 16: 640–649.
Zhu N, Liggitt D, Liu Y, Debs R . Systemic gene expression after intravenous DNA delivery into adult mice. Science 1993; 261: 209–211.
Canonico AE, Conary JT, Meyrick BO, Brigham KL . Aerosol and intravenous transfection of human alpha 1-antitrypsin gene to lungs of rabbits. Am J Respir Cell Mol Biol 1994; 10: 24–29.
Liu Y et al. Factors influencing the efficiency of cationic liposome-mediated intravenous gene delivery. Nat Biotechnol 1997; 15: 167–173.
Liu F, Qi H, Huang L, Liu D . Factors controlling efficiency of cationic lipid-mediated transfection in vivo via intravenous administration. Gene Ther 1997; 4: 517–523.
Templeton NS et al. Improved DNA:liposome complexes for increased systemic delivery and gene expression. Nat Biotechnol 1997; 15: 647–652.
Li S, Huang L . In vivo gene transfer via intravenous administration of cationic lipid–protamine–DNA (LPD) complexes. Gene Ther 1997; 4: 891–900.
Barron LG, Meyer KB, Szoka Jr FC . Effects of complement depletion on the pharmacokinetics and gene delivery mediated by cationic lipid–DNA complexes. Hum Gene Ther 1998; 9: 315–323.
Hong K, Zheng W, Baker A, Papahadjopoulos D . Stabilization of cationic liposome–plasmid DNA complexes by polyamines and poly(ethylene glycol)-phospholipid conjugates for efficient in vivo gene delivery. FEBS Lett 1997; 400: 233–237.
Yates JL, Warren N, Sugden B . Stable replication of plasmids derived from Epstein–Barr virus in various mammalian cells. Nature 1985; 313: 812–815.
Lupton S, Levine AJ . Mapping genetic elements of Epstein–Barr virus that facilitate extrachromosomal persistence of Epstein–Barr virus-derived plasmids in human cells. Mol Cell Biol 1985; 5: 2533–2542.
Margolskee RF, Kavathas P, Berg P . Epstein–Barr virus shuttle vector for stable episomal replication of cDNA expression libraries in human cells. Mol Cell Biol 1988; 8: 2837–2847.
Krysan PJ, Calos MP . Epstein–Barr virus-based vectors that replicate in rodent cells. Gene 1993; 136: 137–143.
Tsukamoto H et al. Enhanced expression of recombinant dystrophin following intramuscular injection of Epstein–Barr virus (EBV)-based mini-chromosome vectors in mdx mice. Gene Ther 1999; 6: 1331–1335.
Tu G et al. Non-replicating Epstein–Barr virus-based plasmids extend gene expression and can improve gene therapy in vivo. J Biol Chem 2000; 275: 30408–30416.
Tomiyasu K et al. Direct intra-cardiomuscular transfer of beta2-adrenergic receptor gene augments cardiac output in cardiomyopathic hamsters. Gene Ther 2000; 7: 2087–2093.
Cui FD et al. Highly efficient gene transfer into murine liver achieved by intravenous administration of naked Epstein–Barr virus (EBV)-based plasmid vectors. Gene Ther 2001; 8: 1508–1513.
Tan Y et al. Sequential injection of cationic liposome and plasmid DNA effectively transfects the lung with minimal inflammatory toxicity. Mol Ther 2001; 3: 673–682.
Ghazizadeh S, Carroll JM, Taichman LB . Repression of retrovirus-mediated transgene expression by interferons: implications for gene therapy. J Virol 1997; 71: 9163–9169.
Qin L et al. Promoter attenuation in gene therapy: interferon-gamma and tumor necrosis factor-alpha inhibit transgene expression. Hum Gene Ther 1997; 8: 2019–2029.
Li S et al. Effect of immune response on gene transfer to the lung via systemic administration of cationic lipidic vectors. Am J Physiol 1999; 276: L796–L804.
Tan Y, Li S, Pitt BR, Huang L . The inhibitory role of CpG immunostimulatory motifs in cationic lipid vector-mediated transgene expression in vivo. Hum Gene Ther 1999; 10: 2153–2161.
Yew NS et al. High and sustained transgene expression in vivo from plasmid vectors containing a hybrid ubiquitin promoter. Mol Ther 2001; 4: 75–82.
Newman PJ . The biology of PECAM-1. J Clin Invest 1997; 99: 3–8.
Dong QG et al. A general strategy for isolation of endothelial cells from murine tissues: characterization of two endothelial cell lines from the murine lung and subcutaneous sponge implants. Arterioscler Thromb Vasc Biol. 1997; 17:1599–1604.
Satoh E et al. Efficient gene transduction by Epstein–Barr-virus-based vectors coupled with cationic liposome and HVJ-liposome. Biochem Biophys Res Commun 1997; 238: 795–799.
Wilson JB, Bell JL, Levine AJ . Expression of Epstein–Barr virus nuclear antigen-1 induces B cell neoplasia in transgenic mice. EMBO J 1996; 15: 3117–3126.
Hammerschmidt W, Sugden B . Genetic analysis of immortalizing functions of Epstein–Barr virus in human B lymphocytes. Nature 1989; 340: 393–397.
Tomkinson B, Robertson E, Kieff E . Epstein–Barr virus nuclear proteins EBNA-3A and EBNA-3C are essential for B-lymphocyte growth transformation. J Virol 1993; 67: 2014–2025.
Kaye KM, Izumi KM, Kieff E . Epstein–Barr virus latent membrane protein 1 is essential for B-lymphocyte growth transformation. Proc Natl Acad Sci USA 1993; 90: 9150–9154.
Saeki Y, Wataya-Kaneda M, Tanaka K, Kaneda Y . Sustained transgene expression in vitro and in vivo using an Epstein–Barr virus replicon vector system combined with HVJ liposomes. Gene Ther 1998; 5: 1031–1037.
Acknowledgements
This work was supported by NIH Grants HL RO1 63080 (to S Li), AI RO1 48851 (to L Huang), and HL RO1 32154 (to B Pitt).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, J., Wilson, A., Alber, S. et al. Prolonged gene expression in mouse lung endothelial cells following transfection with Epstein–Barr virus-based episomal plasmid. Gene Ther 10, 822–826 (2003). https://doi.org/10.1038/sj.gt.3301958
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301958