Abstract
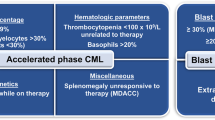
The chronological history of the important discoveries leading to our present understanding of the essential clinical, biological, biochemical, and molecular features of chronic myelogenous leukemia (CML) are first reviewed, focusing in particular on abnormalities that are responsible for the massive myeloid expansion. CML is an excellent target for the development of selective treatment because of its highly consistent genetic abnormality and qualitatively different fusion gene product, p210bcr-abl. It is likely that the multiple signaling pathways dysregulated by p210bcr-abl are sufficient to explain all the initial manifestations of the chronic phase of the disease, although understanding of the circuitry is still very incomplete. Evidence is presented that the signaling pathways that are constitutively activated in CML stem cells and primitive progenitors cooperate with cytokines to increase the proportion of stem cells that are activated and thereby increase recruitment into the committed progenitor cell pool, and that this increased activation is probably the primary cause of the massive myeloid expansion in CML. The cooperative interactions between Bcr-Abl and cytokine-activated pathways interfere with the synergistic interactions between multiple cytokines that are normally required for the activation of stem cells, while at the same time causing numerous subtle biochemical and functional abnormalities in the later progenitors and precursor cells. The committed CML progenitors have discordant maturation and reduced proliferative capacity compared to normal committed progenitors, and like them, are destined to die after a limited number of divisions. Thus, the primary goal of any curative strategy must be to eliminate all Philadelphia positive (Ph+) primitive cells that are capable of symmetric division and thereby able to expand the Ph+ stem cell pool and recreate the disease. Several highly potent and moderately selective inhibitors of Bcr-Abl kinase have recently been discovered that are capable of killing the majority of actively proliferating early CML progenitors with minimal effects on normal progenitors. However, like their normal counterparts, most of the CML primitive stem cells are quiescent at any given time and are relatively invulnerable to the Bcr-Abl kinase inhibitors as well as other drugs. We propose that survival of dormant Ph+ stem cells may be the most important reason for the inability to cure the disease during initial treatment, while resistance to the inhibitors and other drugs becomes increasingly important later. An outline of a possible curative strategy is presented that attempts to take advantage of the subtle differences in the proliferative behavior of normal and Ph+ stem cells and the newly discovered selective inhibitors of Bcr-Abl.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout













Similar content being viewed by others
References
Bennett JH . Case of hypertrophy of the spleen and liver in which death took place from suppuration of the blood. Edinburgh Med Surg J 1845; 64: 413.
Craigie D . Case of disease of the spleen in which death took place in consequence of the presence of purulent matter in the blood. Edinburgh Med Surg J 1845; 64: 400.
Virchow R . Weisses Blut. Froiep's Notizen 1845; 36: 151.
Virchow R . Weisses Blut und Milztumoren I. Med Ztg 1846; 15: 157.
Friedreich N . Ein never fall von leukämie. Arch Pathol Anat 1857; 12: 37.
Fraenkel A . Veber acute leukämie. Dtsch Med Wochenschr 1895; 21: 639.
Neumann E . Veber myelogene leukämie. Berl Klin Wochenschr 1878; 15: 69.
Ehrlich P . Farbenanolytische untersuchungen zur histologic and klinik des blutes. Berlin: Hirschwald, 1891.
Naegli O . Veber rotes knochenmark and myeloblasten. Dtsch Med Wochenschr 1890; 26: 287.
Türk W . Ein systen der lymphomatosen. Wien Klin Wochenschr 1903; 16: 1073.
Baike AG, Court Brown WM, Buckton KE . A possible specific chromosome abnormality in human chronic myeloid leukemia. Nature 1960; 188: 1165.
Nowell PC, Hungerford DA . Chromosome studies on normal and leukemic human leukocytes. J Natl Cancer Inst 1960; 25: 85–109.
Nowell PC, Hungerford DA . Chromosome studies in human leukemia. II. Chronic granulocytic leukemia. J Natl Cancer Inst 1961; 27: 1013.
Tough IM, Court Brown WM, Buckton KE . Cytogenetic studies in chronic leukemia and acute leukemia associated with mongolism. Lancet 1961; 1: 411.
Caspersson T, Gahrton G, Lindsten J, Zech L . Identification of the Philadelphia chromosome on a number 22 by quinacrine mustard fluorescence analysis. Exp Cell Res 1970; 63: 238–240.
Rowley JD . A new consistent chromosomal abnormality in chronic myelogenous leukaemia identified by quinacrine, fluorescence and Giemsa staining. Nature 1973; 243: 290–293.
Bartram CR, de Klein A, Hagemeijer A, van Agthoven T, Geurts von Kessel A, Bootsma D et al. Translocation of c-abl oncogene correlates with the presence of a Philadelphia chromosome in chronic myelocytic leukaemia. Nature 1983; 306: 277–280.
DeKlein A, Van Kessel AG, Grosveld G . A cellular oncogene is translocated to the Philadelphia chromosome in chronic myelocytic leukemia. Nature 1982; 300: 765.
Groffen J, Stephenson JR, Heisterkamp N, de Klein A, Bartram CR, Grosveld G . Philadelphia chromosomal breakpoints are clustered within a limited region, bcr, on chromosome 22. Cell 1984; 36: 93–99.
Shtivelman E, Lifshitz B, Gale RP, Canaani E . Fused transcript of abl and bcr genes in chronic myeloid leukaemia. Nature 1985; 315: 550–554.
Konopka JB, Watanabe SM, Witte ON . An alteration of the human c-abl protein in K562 leukemia cells unmasks associated tyrosine kinase activity. Cell 1984; 37: 1035–1042.
Konopka JB, Witte ON . Detection of c-abl tyrosine kinase activity in vitro permits direct comparison of normal and altered abl gene products. Mol Cell Biol 1985; 5: 3116–3123.
Dreazen O, Klisak I, Rassool F, Goldman JM, Sparkes RS, Gale RP . Do oncogenes determine clinical features in chronic myeloid leukaemia? Lancet 1987; 1: 1402–1405.
Guo JQ, Wang JYJ, Arlinghaus RB . Detection of BCR-ABL proteins in blood cells of benign phase chronic myelogenous leukemia patients. Cancer Res 1991; 51: 3048.
Kurzrock R, Blick MB, Talpaz M . Rearrangement in the breakpoint cluster region and the clinical course in Philadelphia-negative chronic myelogenous leukemia. Ann Intern Med 1986; 105: 673.
Morris CM, Reeve AE, Fitzgerald PH, Hollings PE, Beard MEJ, Heaton DC . Genomic diversity correlates with clinical variation in Ph1 negative chronic myeloid leukaemia. Nature 1986; 320: 281–283.
Weinstein ME, Grossman A, Perle MA . The karyotype of Philadelphia chromosome-negative, bcr rearrangement-positive chronic myeloid leukemia. Cancer Genet Cytogenet 1988; 35: 223.
Vander Plas DC, Hermans ABC, Goekarman D . Cytogenetic and molecular analysis in Philadelphia negative CML. Blood 1989; 73: 1038.
Wisniewski D, Strife A, Wojciechowicz D, Lambek C, Clarkson B . Rapid Communication: A 62-kilodalton tyrosine phosphoprotein constitutively present in primary chronic phase chronic myelogenous leukemia enriched lineage negative blast populations. Leukemia 1994; 8: 688–693.
Bartram CR, Anger B, Carbonell F, Kleihauer E . Involvement of chromosome 9 in variant Ph1 translocation. Leukemia Res 1985; 9: 1133.
Morris CM, Rosman I, Archer SA . A cytogenetic and molecular analysis of five variant Philadelphia translocations in chronic myeloid leukemia. Cancer Genet Cytogenet 1988; 35: 179.
Boveri T . Zur Frage der Entstehung maligner Tumoren. Jena: Gustav Fischer, 1914.
Heim S, Mitelman F (eds). Cancer Cytogenetics, 2nd edn. New York: Wiley-Liss Inc., 1995.
Rowley JD . Molecular cytogenetics: Rosetta stone for understanding cancer – twenty-ninth G.H.A. Clowes Memorial Award Lecture. Cancer Res 1990; 50: 3816–3825.
Sandberg AA, Turc-Carel C, Gemmill RM . Chromosomes in solid tumors and beyond. Cancer Res 1988; 48: 1049–1059.
Yunis JJ . Recurrent chromosomal defects are found in most patients with acute nonlymphocytic leukemia. Cancer Genet Cytogenet 1984; 11: 125–137.
Yunis JJ, Oken MM, Theologides A, Howe RB, Kaplan ME . Recurrent chromosomal defects are found in most patients with non-Hodgkin's-lymphoma. Cancer Genet Cytogenet 1984; 13: 17–28.
Le Beau MM, Larson RA . Cytogenetics and neoplasia (Chapter 59). In: Hoffman, R, Benz Jr EJ, Shattil, SJ, Furie, B, Cohen, HJ, Silberstein LE (eds). Hematology – Basic Principles and Practice, 2nd edn. New York: Churchill Livingstone Inc., 1995, pp 878–898.
Sandberg AA, Gemmill RM, Hecht BK . The Philadelphia chromosome: A model of cancer and molecular cytogenetics. Cancer Genet Cytogenet 1986; 21: 129–146.
Singh S, Wass J, Vincent PC, Young GAR, Gunz FW . Significance of secondary cytogenetic changes in patients with Ph-positive chronic granulocytic leukemia in the acute phase. Cancer Genet Cytogenet 1986; 21: 209–220.
Sokal JE, Gomez GA, Baccarani M, Tura S, Clarkson BD, Cervantes F et al. Prognostic significance of additional cyto-genetic abnormalities at diagnosis of Philadelphia chromosome-positive chronic granulocytic leukemia. Blood 1988; 72: 294–298.
Swolin B, Weinfeld A, Westin J, Waldenstrom J, Magnusson B . Karyotypic evolution in Ph-positive chronic myeloid leukemia in relation to management and disease progression. Cancer Genet Cytogenet 1985; 18: 65–79.
Laneuville P, Heisterkamp N, Groffen J . Expression of the chronic myelogenous leukemia-associated p210bcr/abl oncoprotein in a murine IL-3 dependent myeloid cell line. Oncogene 1991; 6: 275–282.
Bernstein R . Cytogenetics of chronic myelogenous leukemia. Semin Hematol 1988; 25: 20–34.
Sandberg A . The leukemias – chronic granulocytic leukemia. In: The Chromosomes in Human Cancer and Leukemia. New York: Elsevier, 1980, pp 183–261.
Sandberg AA . The cytogenetics of chronic myelocytic leukemia (CML): Chronic phase and blastic crisis. Cancer Genet Cytogenet 1980; 1: 217.
Stoll C, Oberline G . Non-random clonal evolution in 45 cases of chronic myeloid leukemia. Leukemia Res 1980; 46: 61.
Bartram CR, Janssen JWG, Becher R, De Klein A, Grosveld G . Persistence of chronic myelocytic leukemia despite deletion of rearranged bcr/c-abl sequences in blast crisis. J Exp Med 1986; 164: 1389–1396.
Hagemeijer A, Smith EME, Lowenberg B, Abels J . Chronic myeloid leukemia with permanent disappearance of the Ph1 chromosome and development of new clonal subpopulations. Blood 1979; 53: 1–14.
Laneuville P, Sullivan AK . Clonal succession and deletion of bcr/abl sequences in chronic myelogenous leukemia with recurrent lymphoid blast crisis. Leukemia 1991; 5: 752.
Alimena G, De Cuia MR, Diverio D, Gastaldi R, Nanni M . The karyotype of blastic crisis. Cancer Genet Cytogenet 1987; 26: 39–50.
Aventin A, Mecucci C, VanOrshonen A . Variant (3,21) translocation and megakaryocytic involvement in blastic crisis of Philadelphia positive chronic myeloid leukaemia. Br J Haematol 1989; 71: 562.
Bernstein R, Bagg A, Pinto M, Lewis D, Mendelow B . Chromosome 3q21 abnormalities associated with hyperactive thrombopoiesis in acute blastic transformation of chronic myeloid leukemia. Blood 1986; 68: 652–657.
Diez-Martin JL, DeWald GW, Pierre RV . Possible cytogenetic distinction between lymphoid and myeloid blast crisis in chronic granulocytic leukemia. Am J Hematol 1988; 27: 194.
Hogge DE, Misawa S, Schiffer CA, Testa JR . Promyelocytic blast crisis in chronic granulocytic leukemia with 15,17 translocation. Leukemia Res 1984; 8: 1019–1023.
Mitani K, Miyazono K, Urabe A, Takaku F . Karyotypic changes during the course of blastic crisis of chronic myelogenous leukemia. Cancer Genet Cytogenet 1989; 39: 299.
O'Malley FM, Garson OM . Chronic granulocytic leukemia: Correlation of blastic transformation type with karyotypic evolution. Am J Hematol 1985; 20: 313–323.
Parreira L, Kearney L, Rassool F, Babapulle VB, Matutes E, Parreira A et al. Correlation between chromosomal abnormalities and blast phenotype in the blast crisis of Ph-positive CGL. Cancer Genet Cytogenet 1986; 22: 29–34.
Whang-Peng J, Knutsen T . Chromosomal abnormalities. In: Shaw MT (ed). Chronic Granulocytic Leukemia. UK: Praeger, East Sussex, 1982, pp 49–92.
Ahuja H, Bar-Eli M, Arlin Z, Advani S, Allen SL, Goldman J et al. The spectrum of molecular alterations in the evolution of chronic myelocytic leukemia. J Clin Invest 1991; 87: 2042.
Marshal R, Shtalrid M, Talpaz M, Kantarjian H, Smith L, Beran M et al. Rearrangement and expression of p53 in the chronic phase and blast crisis of chronic myelogenous leukemia. Blood 1990; 75: 180.
Trotta R, Vignudelli T, Pecorari L, Candini O, Guerzoni C, Santilli G et al. BCR/ABL activates mdm2 mRNA translation via the La antigen. Blood 2002; 100: 582a (Abstract 2289).
Towatari M, Adachi K, Kato H, Saito H . Absence of the human retinoblastoma gene product in the megakaryoblastic crisis of chronic myelogenous leukemia. Blood 1991; 78: 2178.
LeMaistre A, Lee MS, Talpaz M, Kantarjian HM, Freireich EJ, Deisseroth AB et al. Ras oncogene mutations are rare late stage events in chronic myelogenous leukemia. Blood 1989; 73: 889.
McCarthy DM, Rassool FV, Goldman JM, Graham SV, Birnie GD . Genomic alterations involving the c-myc protooncogene locus during the evolution of a case of chronic granulocytic leukaemia. Lancet 1984; 2: 1362.
Sill H, Goldman JM, Cross NC . Homozygous deletions of the p16 tumor-suppressor gene are associated with lymphoid transformation of chronic myeloid leukemia. Blood 1995; 85: 2013–2016.
Ahuja HG, Popplewell L, Tcheurekdjian L, Slovak ML . NUP98 gene rearrangements and the clonal evolution of chronic myelogenous leukemia. Genes Chromosomes Cancer 2001; 30: 410–415.
Deininger MW, Goldman JM, Melo JV . The molecular biology of chronic myeloid leukemia. Blood 2000a; 96: 3343–3356.
Faderl S, Talpaz M, Estrov Z, O'Brien S, Kurzrock R, Kantarjian HM . The biology of chronic myeloid leukemia. N Engl J Med 1999; 341: 164–172.
Dierov JK, Dierova R, Carroll M . BCR/ABL translocates to the nucleus after DNA damage and interacts with the ataxia-telangiectasia mutant (ATM) protein to modify DNA repair. Blood 2002; 100: 204a (Abstract 764).
Romana DL, Nowicki MO, Falinski R, Stoklosa T, Blasiak J, Skorski T . BCR/ABL stimulates reactive oxygen species to induce DNA double strand breaks, resulting in unfaithful recombination repair and genomic instability. Blood 2002; 100: 581a (Abstract 2284).
Slupianek A, Gurdek E, Koptyra M, Nowicki MO, Skorski T . BCR/ABL stimulates bloom syndrome (BLM) RecQ Helicase: role in resistance to DNA damage. Blood 2002; 100: 583a (Abstract 2294).
Canellos GP, Griffin JD . Chronic granulocytic leukemia: the heterogeneity of stem cell differentiation within a single disease entity. Semin Oncol 1985; 12: 281.
Champlin RE, Golde DW . Chronic myelogenous leukemia: recent advances. Blood 1985; 65: 1039.
Clarkson B . Hematologic malignancies: editorial overview. In: Adamson JW (ed). Current Opinion in Hematology. Philadelphia: Current Science, 1993, pp 167–171.
Clarkson B, Strife A . Review: linkage of proliferative and maturational abnormalities in chronic myelogenous leukemia and relevance to treatment. Leukemia 1993a; 7: 1683–1721.
Griffin JD, Tantravahi R, Canellos GP, Wisch JS, Reinherz EL, Sherwood G et al. T cell surface antigens in a patient with blast crisis of chronic myeloid leukemia. Blood 1983; 61: 640.
Janossy G, Greaves MF, Revesz T, Lister TA, Roberts M, Durrant J et al. Blast crisis of chronic myeloid leukaemia (CML): cell surface marker analysis of ‘lymphoid’ and myeloid cases. Br J Haematol 1976; 34: 179.
Armitage P, Doll R . The age distribution of cancer and a multistage theory of cancer. Br J Cancer 1954; 8: 1–12.
Doll R . The age distribution of cancer: implications for models of carcinogenesis. J R Stat Soc Ser A 1971; 134: 133.
Feinberg AP . Human colorectal cancer. In: Cossman J (ed). Molecular Genetics in Cancer Diagnosis. New York: Elsevier, 1990, pp 419–430.
Hanahan D, Weinberg RA . Review. The hallmarks of cancer. Cell 2000; 100: 57–70.
Peto R, Roe FJC, Lee PN, Levy L, Clack J . Cancer and aging in mice and men. Br J Cancer 1975; 32: 411–426.
Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M et al. Genetic alterations during colorectal-tumor development. N Engl J Med 1988; 319: 525–532.
Weinberg RA . Oncogenes and multistep carcinogenesis. In: Weinberg RA (ed). Oncogenes and The Molecular Origins of Cancer. New York: Cold Spring Harbor Laboratory Press, 1989, pp 307–326.
Folkman J . Editorial: what is the evidence that tumors are angiogenesis dependent? J Natl Cancer Inst 1990; 82: 4–6.
Steeg PS, Bevilacqua G, Sobel ME, Liotta L . Invasion and metastasis. In: Cossman J (ed). Molecular Genetics in Cancer Diagnosis. New York: Elsevier, 1990, pp 104–112.
Clarkson B . New pharmacologic approaches to treatment of leukemia. Semin Hematol 1991; 28(Suppl 4): 99–115.
Clarkson B . Retinoic acid in acute promyelocytic leukemia: the promise and the paradox. Cancer Cells 1991; 3: 211–220.
Nowell P . The clonal evolution of tumor populations. Science 1976; 194: 23–28.
Weinstein IB . Disorders in cell circuitry during multistage carcinogenesis: the role of homeostasis. Carcinogenesis 2000; 21: 857–864.
Weinstein IB . Addiction to oncogenes – the Achilles heel of cancer. Science 2002; 297: 63–64.
Chaganti RSK, Bailey RB, Jhanwar SC, Arlin ZA, Clarkson BD . Chronic myelogenous leukemia in the monosomic cell line of a fertile Turner syndrome mosaic (45,X/46,XX). Cancer Genet Cytogenet 1982; 5: 215–221.
Chaganti RSK, Jhanwar SC, Arlin ZA, Clarkson BD . Chronic myelogenous leukemia in an XYY male. Cancer Genet Cytogenet 1982; 5: 223–226.
Fialkow PJ, Garther SM, Yoshida A . Clonal origin of chronic myelocytic leukemia in men. Proc Natl Acad Sci USA 1967; 58: 1468.
Fialkow PJ, Martin PJ, Najfeld V . Evidence of a multi-step pathogenesis of chronic myelogenous leukemia. Blood 1981; 58: 158–163.
Fitzgerald PH, Pickering AF, Eiby JR . Clonal origin of the Philadelphia chromosome and chronic myeloid leukemia. Evidence from a sex chromosome mosaic. Br J Haematol 1971; 21: 473–480.
Lucas GS, Padua RA, Masters GS, Oscier DG, Jacobs A . The application of X-chromosome gene probes to the diagnosis of myeloproliferative disorders. Br J Haematol 1989; 72: 530–533.
Taylor KM, Shetta M, Talpaz M, Kantarjian H, Hardikar S, Chinault AC et al. Myeloproliferative disorders: Usefulness of X-linked probes in diagnosis. Leukemia 1989; 3: 419.
Gilliland DG, Blanchard KL, Bunn HF . Clonality in myeloproliferative disorders: analysis by means of the polymerase chain reaction. Proc Natl Acad Sci USA 1991; 88: 6848–6852.
Harrison CJ, Chang J, Johnson D . Chromosomal evidence of a common stem cell in acute lymphoblastic leukemia and chronic granulocytic leukemia. Cancer Genet Cytogenet 1984; 13: 331.
Leibowitz D, Schaefer-Rego K, Popenoe DW . Variable breakpoints on the Philadelphia chromosome in chronic myelogenous leukemia. Blood 1985; 66: 243.
Yoffe G, Chinault AG, Talpaz M . Clonal nature of Philadelphia chromosome positive and negative chronic myelogenous leukemia by DNA hybridization analysis. Exp Hematol 1987; 15: 725.
Nitta M, Kato Y, Strife A, Wachter M, Fried J, Perez A et al. Incidence of involvement of the B and T lymphocyte lineages in chronic myelogenous leukemia. Blood 1985; 66: 1053–1061.
Konopka JB, Clark S, McLaughlin J, Nitta M, Kato Y, Strife A et al. Variable expression of the translocated c-abl oncogene in Philadelphia-chromosome-positive B-lymphoid cell lines from chronic myelogenous leukemia patients. Proc Natl Acad Sci USA 1986; 83: 4049–4052.
Bartram CR, Raghavachar A, Anger B, Stain C, Bettelheim P . T lymphocytes lack rearrangement of the bcr gene in Philadelphia chromosome-positive chronic myelocytic leukemia. Blood 1987; 69: 1682.
Jonas D, Lubbert M, Kawasaki ES, Henke M, Bross RJ, Mertelsmann R et al. Clonal analysis of bcr-abl rearrangement in T lymphocytes from patients with chronic myelogenous leukemia. Blood 1992; 79: 1017.
Akashi K, Mizuno S-I, Harada M, Kimura N, Kinjyo M, Shibuya T et al. T lymphoid/myeloid bilineal crisis in chronic myelogenous leukemia. Exp Hematol 1993; 21: 743–748.
Schenk TM, Keyhani A, Bottcher S, Kliche K-O, Goodacre A, Guo J-Q et al. Multilineage involvement of Philadelphia chromosome positive acute lymphoblastic leukemia. Leukemia 1998; 12: 666–674.
Gunsilius E, Duba H-C, Petzer AL, K_hler CM, Gr_newald K, Stockhammer G et al. Early report: evidence from a leukae-mia model for maintenance of vascular endothelium by bone-marrow-derived endothelial cells. Lancet 2000; 355: 1688–1691.
Green AR . Commentary: haemangioblast origin of chronic myeloid leukemia? Lancet 2000; 355: 1659–1660.
Bjornson CR, Rietze RL, Reynolds BA, Magli MC, Vescovi AL . Turning brain into blood: a hematopoietic fate adopted by adult neural stem cells in vivo. Science 1999; 283: 534–537.
Ferrari G, Cusella-De Angelis G, Coletta M . Muscle regeneration by bone marrow-derived myogenic progenitors. Science 1998; 279: 1528–1530.
Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 2002; 418: 41–49.
Bartram CR, Janssen JW, Becher R . Persistence of CML despite deletion of rearranged bcr/c-abl sequences. Haematol Bluttransfus 1987; 31: 145.
Fegan C, Morgan G, Whittaker JA . Spontaneous remission in a patient with chronic myeloid leukaemia. Br J Haematol 1989; 72: 594.
Laneuville P, Sun G, Timm M, Vekemans M . Clonal evolution in a myeloid cell line transformed to interleukin-3 independent growth by retroviral transduction and expression of p210bcr/abl. Blood 1992; 80: 1788–1797.
Lisker R, Casas L, Mutchinick O . Late-appearing Philadelphia chromosome in two patients with chronic myelogenous leukemia. Blood 1980; 56: 812.
Smadja M, Krulik M, DeGramont A . Acquisition of a Philadelphia chromosome concomitant with transformation of a refractory anemia into an acute leukemia. Cancer 1985; 55: 1477.
Smadja N, Krulik M, Audebert AA . Spontaneous regression of cytogenetic and haematology anomalies in Ph1-positive chronic myelogenous leukaemia. Br J Haematol 1986; 63: 257.
Wodzinski MA, Potter AM, Lawrence ACK . Prolonged survival in chronic granulocytic leukemia associated with loss of the Philadelphia chromosome. Br J Haematol 1989; 71: 296.
Martin PJ, Najfeld V, Fialkow PJ . Review article: B-lymphoid cell involvement in chronic myelogenous leukemia: Implications for the pathogenesis of the disease. Cancer Genet Cytogenet 1982; 6: 359–368.
Clarkson B, Strife A, Perez A, Lambek C, Wisniewski D . Integration of molecular and biological abnormalities in quest for selective treatment of chronic myelogenous leukemia (CML). Leuk Lymphoma 1993b; 11(Suppl 2): 81–100.
Bumm T, Mueller C, Leiblein S, Al-Ali H-K, Krohn K, Shepherd P et al. Restoration of polyclonal hematopoiesis in most CML patients in complete cytogenetic remission to imatinib but rapid emergence of clonal cytogenetic abnormalities in Ph-negative cells in a subset of patients. Blood 2002; 100: 164a (Abstract 613).
Clarkson B, Strife A . Cytokinetic considerations relevant to development of a successful therapeutic strategy in chronic myelogenous leukemia CML). Leuk Lymphoma 1993c; 11(Suppl 1): 101–107.
Claxton D, Deisseroth A, Talpaz M, Reading C, Kantarjian H, Trujillo J et al. Polyclonal hematopoiesis in interferon-induced cytogenetic remissions of chronic myelogenous leukemia. Blood 1992; 79: 997–1002.
Cunningham I, Gee T, Dowling M, Chaganti R, Bailey R, Hopfan S et al. Results of treatment of Ph1 chronic myelogenous leukemia with an intensive treatment regimen (L-5). Blood 1979; 53: 375–395.
Dubé ID, Arlin ZA, Kalousek DK, Eaves CJ, Eaves AC . Concise report: nonclonal hemopoietic progenitor cells detected in long-term marrow cultures from a Turner syndrome mosaic with chronic myeloid leukemia. Blood 1984; 64: 1284–1287.
Dunbar CE, Stewart FM . Separating the wheat from the chaff: selection of benign hematopoietic cells in chronic myeloid leukemia. Blood 1992; 79: 1107.
Goto T, Nishikori M, Arlin Z, Gee T, Kempin S, Burchenal J et al. Growth characteristics of leukemic and normal hematopoietic cells in Ph′+ chronic myelogenous leukemia and effects of intensive treatment. Blood 1982; 59: 793–808.
Singer JW, Arlin ZA, Najfeld V, Adamson JW, Kempin SJ, Clarkson BD et al. Restoration of nonclonal hematopoiesis in chronic myelogenous leukemia (CML) following a chemotherapy-induced loss of the Ph1 chromosome. Blood 1980; 56: 356–360.
Strife A, Lambek C, Wisniewski D, Wachter M, Gulati SC, Clarkson BD . Discordant maturation as the primary biological defect in chronic myelogenous leukemia. Cancer Res 1988; 48: 1035–1041.
Hogge DE, Coulombel L, Kalousek DK, Eaves CJ, Eaves AC . Nonclonal hemopoietic progenitors in a G6PD heterozygote with chronic myelogenous leukemia revealed after long-term marrow culture. Am J Hematol 1987; 24: 389–394.
Feldman EJ, Najfeld V, Schuster MW, Roboz GJ, Chadburn A, Goodman O et al. The emergence of Philadelphia chromosome (Ph) negative, trisomy 8 positive cells in patients with chronic myelogenous leukemia (CML) treated with imatinib mesylate: clinical evidence for a multistep pathogenesis. Blood 2002; 100: 585a (Abstract 2299).
Ford AM, Bennett CA, Price CM, Bruin MCA, Van Wering ER, Greaves M . Fetal origins of the TEL-AML1 fusion gene in identical twins with leukemia. Proc Natl Acad Sci USA 1998; 95: 4584–4588.
Wiemels JL, Cazzaniga G, Daniotti M, Eden OB, Addison GM, Masera G et al. Prenatal origin of acute lymphoblastic leukaemia in children. Lancet 1999; 354: 1499–1503.
Gale KB, Ford AM, Repp R, Borkhardt A, Keller C, Eden OB et al. Backtracking leukemia to birth: identification of clonotypic gene fusion sequences in neonatal blood spots. Proc Natl Acad Sci USA 1997; 94: 13950–13954.
Maia AT, Ford AM, Jalali GR, Harrison CJ, Taylor GM, Eden OB et al. Brief report: molecular tracking of leukemogenesis in a triplet pregnancy. Blood 2001; 98: 478–482.
Wiemels JL, Ford AM, Van Wering ER, Postma A, Greaves M . Protracted and variable latency of acute lymphoblastic leukemia after TEL-AML1 gene fusion in utero. Blood 1999; 94: 1057–1062.
Mori H, Colman SM, Xiao Z, Ford AM, Healy LE, Donaldson C et al. Chromosome translocations and covert leukemic clones are generated during normal fetal development. Proc Natl Acad Sci USA 2002; 99: 8242–8247.
Biernaux C, Sels A, Huez G, Stryckmans P . Very low level of major BCR-ABL expression in blood of some healthy individuals. Bone Marrow Transplant 1996; 17(Suppl 3): S45–S47.
Bose S, Deininger M, Gora-Tybor J, Goldman JM, Melo JV . The presence of typical and atypical BCR-ABL fusion genes in leukocytes of normal individuals: biologic significance and implications for the assessment of minimal residual disease. Blood 1998; 92: 3362–3367.
Matioli GT . BCR-ABL insufficiency for the transformation of human stem cells into CML. Medical Hypotheses 2002; 59: 588–589.
Daley GQ, Van Etten RA, Baltimore D . Induction of chronic myelogenous leukemia in mice by the P210bcr-abl gene of the Philadelphia chromosome. Science 1990; 247: 824–830.
Daley GQ, Van Etten RA, Baltimore D . Blast crisis in a murine model of chronic myelogenous leukemia. Proc Natl Acad Sci USA 1991; 88: 11335–11338.
Daley GQ, Ben-Neriah Y . Implicating the bcr/abl gene in the pathogenesis of Philadelphia chromosome-positive human leukemia. Adv Cancer Res 1991; 57: 151–184.
Heisterkamp N, Jenster G, ten Hoeve J, Zovich D, Pattengale PK, Groffen J . Acute leukaemia in bcr/abl transgenic mice. Nature (London) 1990; 344: 251–253.
Li S, Ilaria Jr RL, Million RP, Daley GQ, Van Etten RA . The P190, P210, and P230 forms of the BCR/ABL oncogene induce a similar chronic myeloid leukemia-like syndrome in mice but have different lymphoid leukemogenic activity. J Exp Med 1999; 189: 1399–1412.
Lugo TG, Pendergast A-M, Muller AJ, Witte ON . Tyrosine kinase activity and transformation potency of bcr-abl oncogene products. Science 1990; 247: 1079–1108.
Pear WS, Miller JP, Xu L, Pui JC, Soffer B, Quackenbush RC et al. Efficient and rapid induction of a chronic myelogenous leukemia-like myeloproliferative disease in mice receiving P210 bcr/abl-transduced bone marrow. Blood 1998; 92: 3780–3792.
Roumiantsev S, de Aos IE, Varticovski L, Ilaria RL, Van Etten RA . The src homology 2 domain of Bcr/Abl is required for efficient induction of chronic myeloid leukemia-like disease in mice but not for lymphoid leukemogenesis or activation of phosphatidylinositol 3-kinase. Blood 2001; 97: 4–13.
Wolff NC, Ilaria Jr RL . Establishment of a murine model for therapy-treated chronic myelogenous leukemia using the tyrosine kinase inhibitor STI571. Blood 2001; 98: 2808–2816.
Zhang X, Wong R, Hao SX, Pear WS, Ren R . The SH2 domain of bcr-Abl is not required to induce a murine myeloproliferative disease: however, SH2 signaling influences disease latency and phenotype. Blood 2001; 97: 277–287.
Voncken JW, Morris C, Pattengale P . Clonal development and karyotype evolution during leukemogenesis of BCR/ABL transgenic mice. Blood 1992; 79: 1029–1036.
Ren R . Dissecting the molecular mechanism of chronic myelogenous leukemia using murine models. Leukemia Lymphoma 2002; 43: 1549–1561.
Era T, Witte ON . Regulated expression of P210 Bcr-Abl during embryonic stem cell differentiation stimulates multipotential progenitor expansion and myeloid cell fate. Proc Natl Acad Sci USA 2000; 97: 1737–1742.
Pendergast AM . The Abl kinases: mechanisms of regulation and signaling. Adv Cancer Res 2002; 85: 51–100.
Arlinghaus RB . Working hypothesis: multiple BCR-related gene products and their proposed involvement in ligand-induced signal transduction pathways. Mol Carcinogenesis 1992; 5: 171–173.
Campbell ML, Arlinghaus RB . Current status of the BCR gene and its involvement with human leukemia. Adv Cancer Res 1991; 57: 227–256.
Maru Y, Witte ON . The BCR gene encodes a novel serine/threonine kinase activity within a single exon. Cell 1991; 67: 459.
McWhirter JR, Wang JYJ . Activation of tyrosine kinase and microfilament-binding functions of c-abl by bcr sequences in bcr/abl fusion proteins. Mol Cell Biol 1991; 11: 1553–1565.
McWhirter JR,, Galasso DL, Wang JYJ . A coiled-coil oligomerization domain of Bcr is essential for the transforming function of Bcr-Abl oncoproteins. Mol Cell Biol 1993b; 13: 7587–7595.
Pendergast AM, Muller AJ, Havlik MH, Maru Y, Witte ON . BCR sequences essential for transformation by the BCR-ABL oncogene bind to the ABL SH2 regulatory domain in a non-phosphotyrosine-dependent manner. Cell 1991; 66: 161–171.
Melo JV . Overview: the molecular biology of chronic myeloid leukaemia. Leukemia 1996; 10: 751–756.
Pendergast AM . BCR/ABL protein domain function and signaling. In: Chronic Myeloid Leukaemia: Biology and Treatment. London: Martin Dunitz Ltd., 2001, pp 19–39.
Raitano AB, Whang YE, Sawyers CL . Signal transduction by wild-type and leukemogenic Abl proteins. Biochim Biophys Acta 1997; 1333: F201 F216.
Scheijen B, Griffin JD . Tyrosine kinase oncogenes in normal hematopoiesis and hematological disease. Oncogene 2002; 21: 3314–3333.
Van Etten RA . Animal models of Philadelphia-positive leukemia. In: Chronic Myeloid Leukaemia: Biology and Treatment. London: Martin Dunitz Ltd., 2001, pp 101–131.
Voss J, Heisterkamp N, Groffen J, Feller SM . Review: leukemic kinases of the Abl family – an update. Signal Transduction 2001; 1: 1–26.
Courtney KD, Grove M, Vandongen H, Vandongen A, LaMantia A-S, Pendergast AM . Localization and phosphorylation of Abl-interactor proteins, Abi-1 and Abi-2, in the developing nervous system. Mol Cell Neurosci 2000; 16: 244–257.
Koleske AJ, Gifford AM, Scott ML, Nee M, Bronson RT, Miczek KA et al. Essential roles for the Abl and Arg tyrosine kinases in neurulation. Neuron 1998; 21: 1259–1272.
Patrick GN, Zukerberg L, Nikolic M, de la Monte S, Dikkes P, Tsai L-H . Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 1999; 402: 615–622.
Zambrano N, Bruni P, Minopoli G, Mosca R, Molino D, Russo CL et al. The β-amyloid precursor protein APP is tyrosine-phosphorylated in cells expressing a constitutively active form of the Abl protooncogene. J Biol Chem 2001; 276: 19787–19792.
Kipreos ET, Wang JYJ . Cell cycle-regulated binding of c-Abl tyrosine kinase to DNA. Science 1992; 256: 382–384.
Renshaw WM, McWhirter JR, Wang JYJ . The human leukemia oncogene bcr-abl abrogates the anchorage requirement but not the growth factor requirement for proliferation. Mol Cell Biol 1995; 15: 1286–1293.
Tybulewicz VLJ, Crawford CE, Jackson PK, Bronson RT, Mulligan RC . Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 1991; 65: 1153–1163.
Rosenberg NE, Clark DR, Witte ON . Abelson murine leukemia virus mutants deficient in kinase activity and lymphoid cell transformation. J Virol 1980; 36: 766–774.
Jackson P, Baltimore D . N-terminal mutations activate the leukemogenic potential of the myristoylated form of c-abl. EMBO J 1989; 8: 449–456.
Pendergast AM . Nuclear tyrosine kinases: From Abl to WEE1. Curr Opin Cell Biol 1996; 8: 174–181.
Skorski T, Nieborowska-Skorska M, Wlodarski P, Wasik M, Trotta M, Kanakaraj P et al. The SH3 domain contributes to BCR/ABL-dependent leukemogenesis in vivo: role in adhesion invasion and homing. Blood 1998; 91: 406–418.
Van Etten RA, Jackson P, Baltimore C . The mouse type IV c-abl gene product is a nuclear protein, and activation of transforming ability is associated with cytoplasmic localization. Cell 1989; 58: 669–678.
Dikstein R, Heffetz D, Ben-Neriah Y, Shaul Y . c-abl has a sequence-specific enhancer binding activity. Cell 1992; 5: 751–757.
McWhirter JR, Wang JYJ . An actin binding function contributes to transformation by the bcr-abl oncoprotein of Philadelphia chromosome-positive human leukemias. EMBO J 1993a; 12: 1533–1546.
Kadlec L, Pendergast AM . The amphiphysin-like protein 1 (ALP1) interacts functionally with the cABL tyrosine kinase and may play a role in cytoskeletal regulation. Proc Natl Acad Sci USA 1997; 94: 12390–12395.
Salgia R, Li J-L, Ewaniuk DS, Pear W, Pisick E, Burky SA et al. BCR/ABL induces multiple abnormalities of cytoskeletal function. J Clin Invest 1997; 100: 46–57.
Wertheim JA, Forsythe KL, Hammer D, Boettiger D, Pear SW . Localization of BCR-ABL to F-actin regulates cell adhesion but does not attenuate murine CML development. Blood 2002; 100: 205a (Abstract 770).
Diekmann D, Brill S, Garrett MD, Totty N, Hsuan J, Monfries C et al. Bcr encodes a GTPase-activating protein for p21rac. Nature 1991; 351: 400.
Wu Y, Ma G, Lu D, Lin F, Xu H-J, Liu J, Arlinghaus RB . Bcr: a negative regulator of the Bcr-Abl oncoprotein. Oncogene 1999; 18: 4416–4424.
Melo JV, Hochhaus A, Yan X-H, Goldman JM . Lack of correlation between ABL-BCR expression and response to interferon-α in chronic myeloid leukaemia. Br J Haematol 1996; 92: 684–686.
Plattner R, Kadlec L, DeMali KA, Kazlauskas A, Pendergast AM . c-Abl is activated by growth factors and Src family kinases and has a role in the cellular response to PDGF. Genes Dev 1999; 13: 2400–2411.
Ghaffari S, Wu H, Gerlach M, Han Y, Lodish HF, Daley GQ . BCR-ABL and v-SRC tyrosine kinase oncoproteins support normal erythroid development in erythropoietin receptor-deficient progenitor cells. Proc Natl Acad Sci USA 1999; 96: 13186–13190.
Fainstein E, Einat M, Gokkel E, Marcelle C, Croce CM, Gale RP et al. Nucleotide sequence analysis of human abl and bcr-abl cDNAs. Oncogene 1989; 4: 1477–1481.
Franz WM, Berger P, Wang JYJ . Deletion of an N-terminal regulatory domain of the c-abl tyrosine kinase activates its oncogenic potential. EMBO J 1989; 8: 137–147.
Muller AJ, Young JC, Pendergast A-M, Pondel M, Landau NR, Littman DR et al. BCR first exon sequences specifically activate the BCR/ABL tyrosine kinase oncogene of Philadelphia chromosome-positive human leukemias. Mol Cell Biol 1991; 11: 1785–1792.
Nagar B, Hantschel O, Young MA . Scheffzek K, Veach D, Bornmann W et al. Structural basis for the autoinhibition of c-Abl tyrosine kinase. Cell 2003; 112: 859–871.
Witte ON . Special Lecture: Role of the BCR-ABL oncogene in human leukemia: fifteenth Richard and Hinda Rosenthal Foundation Award Lecture. Cancer Res 1993; 53: 485–489.
Reuther GW, Fu H, Cripe LD, Collier RJ, Pendergast AM . Association of the protein kinases c-Bcr and Bcr-Abl with proteins of the 14-3-3 family. Science 1994; 266: 129–133.
Heisterkamp N, Groffen J, Stephenson J . Chromosomal localization of human cellular homologues of two viral oncogenes. Nature 1982; 299: 747.
Iwata S, Mizutani S, Nakazawa S, Yata J . Heterogeneity of the breakpoint in the ABL gene in cases with BCR/ABL transcript lacking ABL exon a2. Leukemia 1994; 8: 1696–1702.
Kurzrock R, Kloetzer WS, Talpaz M . Identification of molecular variants of P210bcr/abl in chronic myelogenous leukemia patients. Blood 1987; 70: 233.
Kurzrock R, Gutterman JU, Talpaz M . The molecular genetics of Philadelphia chromosome-positive leukemias. Mechanisms of Disease. N Engl J Med 1988; 319: 990–998.
Melo JV . Editorial: the diversity of BCR-ABL fusion proteins and their relationship to leukemia phenotype. Blood 1996; 88: 2375–2384.
Mills KI, Benn P, Birnie GD . Review article: does the breakpoint within the major breakpoint cluster region (M-bcr) influence the duration of the chronic phase in chronic myeloid leukemia? An analytical comparison of current literature. Blood 1991; 78: 1155–1161.
Ben-Neriah Y, Daley GQ, Mes-Masson A-M . The chronic myelogenous leukemia-specific P210 protein is the product of the bcr/abl hybrid gene. Science 1985; 233: 212.
Shtivelman E, Gale RP, Dreazen O . bcr-abl RNA in patients with chronic granulocytic leukemia. Blood 1987; 69: 971.
Stam K, Heisterkamp N, Grosveld G . Evidence of a new chimeric bcr/c-abl mRNA in patients with chronic myelocytic leukemia and the Philadephia chromosome. N Engl J Med 1985; 313: 1429.
Rosenberg N, Witte ON . The viral and cellular forms of the Abelson (abl) oncogene. Adv Virus Res 1988; 35: 39–81.
Chen SJ, Chen Z, Grausz JD, Hillion J, d'Auriol L, Flandrin G et al. Molecular cloning of a 5′ segment of the genomic phl gene defines a new breakpoint cluster region (bcr2) in Philadelphia-positive acute leukemias. Leukemia 1988; 2: 634–641.
Denny CT, Shah N, Ogden S, Willman C, McConnell T, Crist W et al. Localization of preferential sites of rearrangement within the BCR gene in Philadelphia chromosome positive acute lymphoblastic leukemia. Proc Natl Acad Sci USA 1989; 86: 4254–4258.
Erikson J, Griffin CA, Ar-Rushdi A, Valtieri M, Hoxie J, Finan J et al. Heterogeneity of chromosome 22 breakpoint in Philadelphia positive (Ph1+), acute lymphocytic leukemia. Proc Natl Acad Sci USA 1986; 83: 1807.
Hermens A, Heisterkamp N, von Lindern M, von Baal S, Meijer D, van der Plas D et al. Unique fusion of bcr alpha c-abl genes in Philadelphia chromosome positive acute lymphoblastic leukemia. Cell 1987; 51: 33–40.
Pane F, Frigeri F, Sindona M, Luciano L, Ferrara F, Cimino R et al. Rapid communication. Neutrophilic-chronic myeloid leukemia: a distinct disease with a specific molecular marker (BCR/ABL with C3/A2 junction). Blood 1996; 88: 2410–2414.
Clark SS, Christ WM, Witte ON . Molecular pathogenesis of Ph-positive leukemia. Annu Rev Med 1989; 40: 113–122.
Daley GQ, McLaughlin J, Witte ON, Baltimore D . The CML-specific P210 bcr/abl protein, unlike v-abl, does not transform NIH-3T3 fibroblasts. Science 1987; 237: 532–535.
McLaughlin J, Chianese E, Witte ON . In vitro transformation of immature hematopoietic cells by the P210 BCR/ABL oncogene product of the Philadelphia chromosome. Proc Natl Acad Sci USA 1987; 84: 6558–6562.
McLaughlin J, Chianese E, Witte ON . Alternative forms of the BCR/ABL oncogene have quantitatively different potencies for stimulation of immature lymphoid cells. Mol Cell Biol 1989; 9: 1866–1874.
Elefanty AG, Hariharan IK, Cory S . bcr-abl, the hallmark of chronic myeloid leukaemia in man, induces multiple haemopoietic neoplasms in mice. EMBO J 1990; 9: 1069–1078.
Kelliher MA, McLaughlin J, Witte ON, Rosenberg N . Induction of a chronic myelogenous leukemia-like syndrome in mice with v-abl and BCR/ABL. Proc Natl Acad Sci USA 1990; 87: 6649–6653 (Erratum, Proc Natl Acad Sci USA 1990; 87: 9072).
Van Etten RA . Distinct effect of expression of the ALL-specific form of BCR/ABL, P210bcr-abl, in murine bone marrow. Blood 1991; 78: 78a (Abstract 304).
Anafi M, Gazit A, Gilon C, Ben-Neriah Y, Levitzki A . Selective interactions of transforming and normal ab/proteins with ATP, tyrosine-copolymer substrates, and tyrphostins. J Biol Chem 1992; 267: 4518–4523.
Tuohy EL . A case of splenomegaly with polymorphonuclear neutrophil hyperleucocytosis. Am J Med Sci 1920; 18: 160.
You W, Weisbrot IM . Chronic neutrophilic leukemia: report of two cases and review of the literature. Am J Clin Pathol 1979; 72: 233.
Saglio G, Guerrasio A, Rosso C, Zaccaria A, Tassinari A, Serra A et al. New type of Bcr/Abl junction in Philadelphia chromosome-positive chronic myelogenous leukemia. Blood 1990; 76: 1819.
Wada H, Mizutani S, Nishimura J, Usuki Y, Kohsaki M, Komai M et al. Establishment and molecular characterization of a novel leukemic cell line with Philadelphia chromosome expressing p230 BCR/ABL fusion protein. Cancer Res 1995; 55: 3192.
Didsbury J, Weber RF, Bokoch GM, Evans T, Snyderman R . rac, a novel ras-related family of proteins that are botulinum toxin substrates. J Biol Chem 1989; 264: 16378.
Inokuchi K, Dan K, Uchida N, Inami M, Tarusawa M, Yamaguchi H et al. Novel transgenic mice expressing P230 Bcr-Abl developed myeloproliferative disease closely resembling human CML. Blood 2002; 100: 204a (Abstract 767).
Pendergast AM . Identification of novel pathways targeted by the oncogenic bcr-abl tyrosine kinases. Proceedings of the International Symposium on Chronic Myelogenous Leukemia, Biarritz, France, July 7–9, 1999 (Abstract, p 29).
Mayer BJ, Jackson PK, Van Etten RA, Baltimore D . Point mutations in the abl SH2 domain coordinately impair phosphotyrosine binding in vitro and transforming activity in vivo. Mol Cell Biol 1992; 12: 609–618.
Mayer BJ, Baltimore D . Mutagenic analysis of the roles of SH2 and SH3 domains in regulation of the Abl tyrosine kinase. Mol Cell Biol 1994; 14: 2883–2894.
Overduln M, Rios CB, Mayer BJ, Baltimore D, Cowburn D . Three-dimensional solution structure of the src homology 2 domain of c-abl. Cell 1992; 70: 697–704.
Songyang Z, Shoelson SE, Chaudhuri M, Gish G, Pawson T, Haser WG et al. SH2 domains recognize specific phosphopeptide sequenes. Cell 1993; 72: 767–778.
Brown MT, Cooper JA . Regulation, substrates and functions of src. Biochem Biophys Acta 1996; 1287: 121–149.
Kuriyan J, Cowburn D . Modular peptide recognition domains in eukaryotic signaling. Annu Rev Biophys Biomol Struct 1997; 26: 259–288.
Pawson T . Protein modules and signaling networks. Nature 1995; 373: 573–580.
Thomas AM, Brugge JS . Cellular functions regulated by Src family kinases. Annu Rev Cell Biol 1997; 13: 513–609.
Hunter T . A tail of two src's. Mutatis mutandis. Cell 1987; 49: 1–4.
Sicheri F, Kuriyan J . Structures of Src-family tyrosine kinases. Curr Opin Struct Biol 1997; 7: 777–785.
Xu W Harrison SC, Eck MJ . Three-dimensional structure of the tyrosine kinase c-Src. Nature 1997; 385: 595–602.
Moarefi I, LaFevre-Bernt M, Sicheri F, Huse M, Lee CH, Kuriyan J et al. Activation of the Src-family tyrosine kinase Hck by SH3 domain displacement. Nature 1997; 385: 650–653.
Schindler T, Sicheri F, Pico A, Gazit A, Levitzki A, Kuriyan J . Crystal structure of Hck in complex with a Src family-selective tyrosine kinase inhibitor. Mol Cell 1999; 3: 639–648.
Dorey K, Engen JR,, Kretzschmar J, Wilm M, Neubauer G, Schindler T et al. Phosphorylation and structure-based functional studies reveal a positive and a negative role for the activation loop of the c-Abl tyrosine kinase. Oncogene 2001; 20: 8075–8084.
Hunter T . Review: oncoprotein networks. Cell 1997; 88: 333–346.
Hunter T, Plowman GD . Review: the protein kinases of budding yeast: six score and more. Trends Biochem Sci 1997; 22: 18–22.
Plowman GD, Sudarsanam S, Bingham J, Whyte D, Hunter T . Review: the protein kinases of Caenorhabditis elegans: a model for signal transduction in multicellular organisms. PNAS 1999; 96: 13603–13610.
Hunter T . Signaling – 2000 and beyond. Cell 2000; 100: 113–127.
Cantley LC, Auger KR, Carpenter C, Duckworth B, Graziani A, Kapeller R et al. Review: oncogenes and signal transduction. Cell 1991; 64: 281–302.
Carpino N, Wisniewski D, Strife A, Marshak D, Kobayashi R, Stillman B et al. p62dok: a constitutively tyrosine-phosphorylated, GAP-associated protein in chronic myelogenous leukemia progenitor cells. Cell 1997; 88: 197–204.
Clarkson BD, Strife A, Wisniewski D, Lambek C, Carpino N . Leading Article: new understanding of the pathogenesis of CML: a prototype of early neoplasia. Leukemia 1997; 11: 1404–1428.
Garton AJ, Flint AJ, Tonks NK . Identification of substrates for protein tyrosine phosphatases. In: Hardie DG (ed). Protein Phosphorylation: a Practical Approach. 2nd edn, New York: Oxford University Press, 1999, pp 183–200.
Rane SG, Reddy EP . JAKs, STATs and Src kinases in hematopoiesis. Oncogene 2002; 21: 3334–3358.
Ullrich A, Schlessinger J . Review: signal transduction by receptors with tyrosine kinase activity. Cell 1990; 61: 203–212.
Wisniewski D, Strife A, Berman E, Clarkson B . c-kit ligand stimulates tyrosine phosphorylation of a similar pattern of phosphotyrosyl proteins in primary primitive normal hematopoietic progenitors that are constitutively phosphorylated in comparable primitive progenitors in chronic phase chronic myelogenous leukemia. Leukemia 1996; 10: 229–237.
Wisniewski D, Strife A, Clarkson B . Rapid Communication: c-kit ligand stimulates tyrosine phosphorylation of the c-Cbl protein in human hematopoietic cells. Leukemia 1996; 10: 1436–1442.
Wisniewski D, Strife A, Swendeman S, Erdjument-Bromage H, Geromanos S, Kavanaugh WM et al. A novel SH2-containing phosphatidyl-inositol 3,4,5-triphosphate 5-phosphatase (SHIP2) is constitutively tyrosine phosphorylated and associated with src homologous and collagen gene (SHC) in chronic myelogenous leukemia progenitor cells. Blood 1999; 93: 2707–2720.
Charbonneau H, Tonks NK . 1002 protein phosphatases? Annu Rev Cell Biol 1992; 8: 463–493.
Adachi M, Fischer EH, Ihle JN, Imani K, Jirik F, Neel B et al. Mammalian SH2-containing protein tyrosine phosphatases. Cell 1996; 85: 15.
Wickrema A, Chen F, Namin F, Yi T, Ahmad S, Uddin S et al. Defective expression of the SHP-1 phosphatase in polycythemia vera. Exp Hematol 1999; 27: 1124–1132.
Bignon JS, Siminovitch KA . Review: identification of PTP1C mutation as the genetic defect in motheaten and viable motheaten mice: a step toward defining the roles of protein tyrosine phosphatases in the regulation of hemopoietic cell differentiation and function. Clin Immunol Immunopathol 1994; 73: 168–179.
Berg KL, Siminovitch KA, Stanley ER . SHP-1 regulation of p62DOK tyrosine phosphorylation in macrophages. J Biol Chem 1999; 274: 35855–35865.
Holtz MS, Slovak ML, Zhang F, Sawyers CL, Forman SJ, Bhatia R . Imatinib mesylate (STI571) inhibits growth of primitive malignant progenitors in chronic myelogenous leukemia through reversal of abnormally increased proliferation. Blood 2002; 99: 3792–3800.
Strife A, Wisniewski D, Liu C, Lambek CL, Darzynkiewicz Z, Silver RT et al. Direct Evidence that Bcr-Abl tyrosine kinase activity disrupts normal synergistic interactions between kit ligand and cytokines in primary primitive progenitor cells. Mol Cancer Res, 2003; 1: 176–185.
Wisniewski D, Lambek C, Liu C, Strife A, Veach D, Nagar B et al. Characterization of potent inhibitors of the Bcr-Abl and the c-kit receptor tyrosine kinases. Cancer Res 2002; 15: 4244–4255.
Bishop JM . Review: molecular themes in oncogenesis. Cell 1991; 64: 235–248.
Pawson T, Scott JD . Signaling through scaffold, anchoring, and adaptor proteins. Science 1997; 278: 2075–2080.
Toker A, Cantley LC . Signalling through the lipid products of phosphoinositide-3-OH kinase. Nature 1997; 387: 673–676.
Evans JP, Wickremasinghe RG, Hoffbrand AV . Preliminary communication: detection of tyrosine protein kinase substrates in fresh leukemia cells and normal blood cells using an immunoblotting technique. Leukemia 1987; 1: 782–785.
Huhn RD, Posner MR, Rayter SI, Foulkes JG, Frackelton Jr AR . Cell lines and peripheral blood leukocytes derived from individuals with chronic myelogenous leukemia display virtually identical proteins phosphorylated on tyrosine residues. Proc Natl Acad Sci USA 1987; 84: 4408–4412.
Naldini L, Stacchini A, Cirillo DM, Aglietta M, Gavosto F, Comoglio PM . Phosphotyrosine antibodies identify the p210c-abl tyrosine kinase and proteins phosphorylated on tyrosine in human chronic myelogenous leukemia cells. Mol Cell Biol 1986; 6: 1803–1811.
Ogawa R, Ohtsuka M, Watanabe Y . Complement-mediated lysis of K562 human leukemic cells by antibodies to phosphotyrosine and identification of cell surface proteins phosphorylated on tyrosine. Cancer Res 1986; 46: 2507–2510.
Druker B, Okuda K, Matulonis U, Salgia R, Roberts T, Griffin JD . Rapid communication: tyrosine phosphorylation of rasGAP and associated proteins in chronic myelogenous leukemia cell lines. Blood 1992; 79: 2215–2220.
Lu D, Liu J, Campbell M, Guo JQ, Heisterkamp N, Groffen J et al. Tyrosine phosphorylation of P160 BCR by P210 BCR-ABL. Blood 1993; 82: 1257–1263.
Gotoh A, Miyazaqa K, Ohyashiki K, Tauchi T, Boswell HS, Broxmeyer HE et al. Tyrosine phosphorylation and activation of focal adhesion kinase (p125FAK) by BCR-ABL oncoprotein. Exp Hematol 1995; 23: 1153–1159.
Andoniou CE, Thien CBF, Langdon WY . Tumour induction by activated abl involves tyrosine phosphorylation of the product of the cbl oncogene. EMBO J 1994; 13: 4515–4523.
de Jong R, ten Hoeve J, Heisterkamp N, Groffen J . Communication: Crkl is complexed with tyrosine-phosphorylated Cbl in Ph-positive leukemia. J Biol Chem 1995; 270: 21468–21471.
Matsuguchi T, Inhorn RC, Carlesso N, Xu G, Druker B, Griffin JD . Tyrosine phosphorylation of p95Vav in myeloid cells is regulated by GM-CSF, IL-3 and Steel factor and is constitutively increased by p210BCR/ABL. EMBO J 1995; 14: 257–265.
Ernst TJ, Slattery KE, Griffin JD . p210Bcr/Abl and p160v-Abl induce an increase in the tyrosine phosphorylation of p93c-Fes. J Biol Chem 1994; 269: 5764–5769.
Salgia R, Brunkhorst B, Pisick E, Li J-L, Lo SH, Chen LB et al. Increased tyrosine phosphorylation of focal adhesion proteins in myeloid cell lines expressing p210BCR/ABL. Oncogene 1995; 11: 1149–1155.
Tauchi T, Boswell HS, Leibowitz D, Broxmeyer HE . Coupling between p210bcr-abl and Shc and Grb2 adaptor proteins in hematopoietic cells permits growth factor receptor-independent link to ras activation pathway. J Exp Med 1994; 179: 167–175.
Tauchi T, Feng G-S, Marshall MS, Shen R, Mantel C, Pawson T, Broxmeyer HE . The ubiquitously expressed Syp phosphatase interacts with c-kit and Grb2 in hematopoietic cells. J Biol Chem 1994; 269: 25206–25211.
McGlade J, Cheng A, Pelicci G, Pelicci PG, Pawson T . Shc proteins are phosphorylated and regulated by the v-Src and v-Fps protein-tyrosine kinases. Proc Natl Acad Sci USA 1992; 89: 8869–8873.
Pelicci G, Lanfrancone L, Grignani F, McGlade J, Cavallo F, Forni G et al. A novel transforming protein (SHC) with an SH2 domain is implicated in mitogenic signal transduction. Cell 1992; 70: 93–104.
Pelicci G, Lanfrancone L, Salcini AE, Romano A, Mele S, Borrello MG et al. Constitutive phosphorylation of Shc proteins in human tumors. Oncogene 1995; 11: 899–907.
Puil L, Liu J, Gish G, Mbamalu G, Bowtell D, Pelicci PG et al. Bcr-Abl oncoproteins bind directly to activators of the Ras signalling pathway. EMBO J 1994; 13: 764–773.
Nichols GL, Raines MA, Vera JC, Lacomis L, Tempst P, Golde DW . Blood 1994; 84: 2912–2918.
Oda T, Heaney C, Hagopian JR, Okuda K, Griffin JD, Druker B . J Biol Chem 1994; 269: 22925–22928.
ten Hoeve J, Arlinghaus RG, Guo JQ, Heisterkamp N, Groffen J . Blood 1994; 84: 1731–1736.
Uemura N, Salgia R, Li JL, Pisick E, Sattler M, Griffin JD . The BCR/ABL oncogene alters interaction of the adapter proteins CRKL and CRK with cellular proteins. Leukemia 1997; 11: 376–385.
Sattler M, Salgia R, Shrikhande G, Verma S, Choi J-L, Rohrschneider LR et al. The phosphatidylinositol polyphosphate 5-phosphatase SHIP and the protein tyrosine phosphatase SHP-2 form a complex in hematopoietic cells which can be regulated by BCR/ABL and growth factors. Oncogene 1997; 15: 2379–2384.
Di Cristofano A, Carpino N, Dunant N, Friedland G, Kobayashi R, Strife A et al. Communication: molecular cloning and characterization of p56dok-2 defines a new family of RasGAP-binding proteins. J Biol Chem 1998; 273: 4827–4830.
Di Cristofano A, Pesce B, Cordon-Cardo C, Pandolfi PP . Pten is essential for embryonic development and tumour suppression. Nat Genet 1998; 19: 348–355.
Dunant NM, Wisniewski D, Strife A, Clarkson B, Resh MD . The phosphatidylinositol polyphosphate 5-phosphatase SHIP1 associates with the Dok1 phosphoprotein in Bcr-Abl transformed cells. Cell Signal 2000; 12: 317–326.
Cleveland JL, Dean M, Rosenberg N, Wang JY, Rapp UR . Tyrosine kinase oncogenes abrogate interleukin-3 dependence of murine myeloid cells through signaling pathways involving c-myc: conditional regulation of c-myc transcription by temperature-sensitive v-abl. Mol Cell Biol 1989; 9: 5685–5695.
Cohen L, Mohr R, Chen YY, Huang M, Kato R, Dorin D et al. Transcriptional activation of a ras-like gene (kir) by oncogenic tyrosine kinases. Proc Natl Acad Sci USA 1994; 91: 12448–12452.
Deininger MWN, Vieira S, Mendiola R, Schultheis B, Goldman JM, Melo JV . BCR-ABL tyrosine kinase activity regulates the expression of multiple genes implicated in the pathogenesis of chronic myeloid leukemia. Cancer Res 2000b; 60: 2049–2055.
Watari K, Tojo A, Nagamura-Inoue T, Tsujimura H, Nagamura F, Tani K et al. A new melanoma antigen, PRAME, is identified as a BCR./ABL-inducible gene in TF-1 cells. Blood 1997; 90(Suppl 1): 197a.
Xie Z, Chang S, McQueen T, Andreeff M . Effect of BCR/ABL transfection on the gene expression profile of human hematopoietic M07e cells: Analysis by DNA expression array. Blood 1998; 92(Suppl 1): 91a.
Advani AS, Dressman HK, Quiroz M, Taylor GA, Pendergast AM . Elevated expression of numerous interferon inducible genes in primary bone marrow cells expressing P185 Bcr-Abl versus P210 Bcr-Abl by DNA microarray analysis. Blood 2002; 100: 584a (Abstract 2296).
Liang X, Wisniewski D, Strife A, Shivakrupa, Clarkson B, Resh MD . Phosphatidylinositol 3-kinase and Src family kinases are required for phosphorylation and membrane recruitment of Dok-1 in c-kit signaling. (Published online ahead of print February 1 2002 as 101074/jbcM200277200). J Biol Chem 2002; 277: 13732–13738.
Strife A, Clarkson B . Biology of chronic myelogenous leukemia: is discordant maturation the primary defect? Seminars in Hematology 1988; 25: 1–19.
Deininger MWN, Vieira SAD, Parada Y, Banerji L, Lam EW-F, Peters G et al. Direct relation between BCR-ABL tyrosine kinase activity and Cyclin D2 expression in lymphoblasts. Cancer Res 2001; 61: 8005–8013.
Jena N, Deng M, Sicinska E, Sicinski P, Daley GQ . Critical role for Cyclin D2 in BCR/ABL-induced proliferation of hematopoietic cells. Cancer Res 2002; 62: 535–541.
Jonuleit T, van der Kuip H, Miething C, Michels H, Hallek M, Duyster J et al. Bcr-Abl kinase down-regulates cyclin-dependent kinase inhibitor p27 in human and murine cell lines. Blood 2000; 96: 1933–1939.
Parada Y, Banerji L, Glassford J, Lea NC, Collado M, Rivas C et al. BCR-ABL and Interleukin 3 promote haematopoietic cell proliferation and survival through modulation of Cyclin D2 and p27Kip1 expression. J Biol Chem 2001; 276: 23572–23580.
Wang Y, Miller AL, Moosseker MS, Koleske AJ . The Abl-related gene (Arg) nonreceptor tyrosine kinase uses two F-actin-binding domains to bundle F-actin. Proc Natl Acad Sci 2001; 98: 14865–14870.
Faderl S, Talpaz M, Estrov Z, Kantarjian HM . Chronic myelogenous leukemia: biology and therapy. Ann Intern Med 1999; 131: 207–219.
Kantarjian H, Melo JV, Tura S, Giralt S, Talpaz M . Chronic myelogenous leukemia: disease biology and current and future therapeutic strategies. Hematology (Am Soc Hematol Educ Program) 2000; 90–109.
Sawyers CL . Review article: Medical Progress: Chronic myeloid leukemia. N Eng J Med 1999; 340: 1330–1340.
Silver RT, Woolf SH, Hehlmann R, Appelbaum FR, Anderson J, Bennett C et al. An evidence-based analysis of the effect of busulfan, hydroxyurea, interferon, and allogeneic Bone Marrow Transplant in treating the chronic phase of chronic myeloid leukemia developed for the American Society of Hematology. Blood 1999; 94: 1517–1536.
Court Brown WM, Doll R . Adult leukemia. Br Med J 1960; 1: 1753.
Boise Jr JD, Day NE, Anderson A . Second cancers following radiation treatment for cervical cancer. J Natl Cancer Inst 1985; 74: 955.
Ichimaru M, Ishimaru T, Belsky JL . Incidence of leukemia in atomic bomb survivors belonging to a fixed cohort in Hiroshima and Nagasaki, 1950–71. Radiation dose, years after exposure, age at exposure, and type of leukemia. J Radiat Res (Tokyo) 1978; 19: 262–282.
Maloney WC . Radiation leukemia revisited. Blood 1987; 70: 905.
Dowling MD, Haghbin M, Gee T, Wakonig-Vaartaja T, Clarkson B . Attempt to induce true remissions in chronic myelocytic leukemia (CML). Cancer Chemother Rep 1973; 57: 102.
Kantarjian H, Talpaz M, Spinolo J . High doses of cyclophosphamide, BCNU, and etoposide induce cytogenetic responses in most patients with advanced stages of Philadelphia chromosome (Ph1)-positive chronic myelogenous leukemia. Blood 1989; 74: 273a.
Clarkson B, Strife A . Discordant maturation in chronic myelogenous leukemia. In: Deisseroth AB, Arlinghaus RB (eds). Chronic Myelogenous Leukemia – Molecular Approaches to Research and Therapy. New York, NY: Marcel Dekker, Inc., 1991, pp 3–90.
Pedersen B . Annotation: functional and biochemical phenotype in relation to cellular age of differentiated neutrophils in chronic myeloid leukemia. Br J Haematol 1982; 51: 339–344.
Donohue DM . Quantitative measurement of hematopoietic cells of the marrow. J Clin Invest 1958; 37: 1564.
Ellis RE . The distribution of active bone marrow in the adult. Phys Med Biol 1961; 5: 255.
Finch CA . Kinetics of the formed elements of human blood. Blood 1977; 50: 699.
Nelp WB, Bower RE . The quantitative distribution of the erythron and the RE cell in the bone marrow organ of man. Blood 1969; 34: 276.
Jandl JH (ed) . Chapter 22 – Chronic myeloproliferative syndromes. In: Blood – Textbook of Hematology. Boston: Little, Brown and Company, 1987, pp 671–690.
Clarkson BD, Fried J, Chou T-C, Strife A, Ferguson R, Sullivan S et al. Duration of the dormant state in an established cell line of human hematopoietic cells. Cancer Res 1977; 37: 4506–4522.
Clarkson B, Rubinow SI . Growth kinetics in human leukemia. In: Drewinko B, Humphrey R (eds). Growth Kinetics and Biochemical Regulations of Normal and Malignant Cells. Baltimore: Williams & Wilkins Publ. Co., 1977, pp 591–628.
Ogawa M, Fried J, Sakai Y, Strife A, Clarkson BD . Studies of cellular proliferation in human leukemia. VI. The proliferative activity, generation time, and emergence time of neutrophilic granulocytes in chronic granulocytic leukemia. Cancer 1970; 25: 1031–1049.
Buckle A-M, Mottram R, Pierce A, Lucas GS, Russell N, Miyan JA et al. The effect of Bcr-Abl protein tyrosine kinase on maturation and proliferation of primitive haematopoietic cells. Mol Med 2000; 6: 892–902.
Passegue E, Jochum W, Schorpp-Kistner M, Mhle-Steinlein U, Wagner EF . Chronic myeloid leukemia with increased granulocyte progenitors in mice lacking JunB expression in the myeloid lineage. Cell 2001; 104: 21–32.
Dao MA, Verfaillie CM . STI571 precedes the action of proteasome inhibitors on cell cycle: tracking the molecular mechanism of p27Kip1 degradation in p210bcr/abl hematopoietic cells. Blood 2002; 100: 580a (Abstract 2278).
Baggiolini M, Dewald B, Moser B . Human chemokines: an update. Ann Rev Immunol 1997; 15: 675–705.
Broxmeyer HE, Kim CH . Regulation of hematopoiesis in a sea of chemokine family members with a plethora of redundant activities. Exp Hematol 1999; 27: 1113–1123.
Ogawa M . Review article. Differentiation and proliferation of hematopoietic stem cells. Blood 1993; 81: 2844–2853.
Orkin SH, Porcher C, Fujiwara Y, Visvader J, Wang LC . Intersections between blood cell development and leukemia genes. Cancer Res 1999; 59(Suppl): 1784s–1787s.
Papayannopoulou T . Hematopoietic stem/progenitor cell mobilization A continuing quest for etiologic mechanisms. Ann NY Acad Sci 1999; 872: 187–197.
Williams DA . Editorial. Ex vivo expansion of hematopoietic stem and progenitor cells – Robbing Peter to pay Paul? Blood 1993; 81: 3169–3172.
Darte JM, Dacie JV, McSorley JGA . Pelger-like leucocytes in chronic myeloid leukemia. Acta Haematol 1952; 12: 117.
Langenhuijsen MMAC . Neutrophils with ring-shaped nuclei in myeloprolifera-tive disease. Br J Haematol 1984; 58: 277.
Matsuo T, Tomonaga M, Kuriyama K, Yao E, Nonaka H, Jinnai I et al. Prognostic significance of the morphological dysplastic changes in chronic myelogenouss leukemia. Leuk Res 1986; 10: 331–337.
Wintrobe MM, Lee GR, Boggs DR, Bithell TC, Foerster J, Athens JW et al. (eds). Chronic myeloid leukemia, Chapter 64. In: Clinical Hematology, 8th edn. Philadelphia: Lea & Febiger Publishing Co., 1981, pp. 1565–1595.
Lingg G, Schmalzl F, Breton-Gorius J, Tabilio A, Schaefer HE, Geissler D et al. Megakaryoblastic micromegakaryocytic crisis in chronic myeloid leukemia. Blut 1985; 51: 275–285.
Doan CA, Reinhart HL . The basophil granulocyte, basophilocytosis and myeloid leukemia basophil and ‘mixed granule’ types: an experimental clinical and pathological study with the report of a new syndrome. Am J Clin Pathol 1941; 11: 1–33.
Mlynek M-L, Leder L-D . Lineage infidelity in chronic myeloid leukemia: Demonstration and significance of hybridoid leukocytes. Virchows Arch B Cell Pathol 1986; 51: 107–114.
Anderson D . Ultrastructure of normal and leukaemic leukocytes in human peripheral blood. J Ultrastruct Res 1966; 9(Suppl): 1–42.
Asano M, Kawahara I . Ultramicroscopic characteristics of bone marrow cells in human chronic myeloid leukemia. Med J Shinshu Univ Jpn 1968; 13: 109–126.
Bessis M . Ultrastructure of normal and leukaemic granulocytes. In: Zarafonetis CJS (ed). Proceedings of the International Conference on Leukaemia-Lymphoma. Philadelphia: Lea & Febiger, 1986, p 281.
Cawley JC, Hayhoe FGJ (eds). Ultrastructure of Haemic Cells: A Cytological Atlas of Normal and Leukaemic Blood and Bone Marrow. London: W.B. Saunders Company Ltd., 1973.
Kakefuda T . Electron microscopy of normal and leukaemic cells. In: Amromin GD (ed). Pathology of Leukaemia. New York: Hoeber, 1968, pp 82–124.
Tanaka Y, Goodman JR . Electron Microscopy of Human Blood Cells. New York: Harper and Row, 1972.
Vainchenker W, Guichard J, Deschamps JF . Megakaryocyte cultures in the chronic phase and in the blast crisis of chronic myeloid leukaemia: studies on the differentiation of the megakaryocyte progenitors and on the maturation of megakaryocytes in vitro. Br J Haematol 1982; 51: 131–146.
Kuto F, Nagaoka T, Watanabe Y, Hayashi M, Horasawa Y, Hirasawa Y et al. Chronic myelocytic leukemia: ultrastructural histopathology of bone marrow from patients in the chronic phase. Ultrastruct Pathol 1984; 6: 307–317.
Nagaoka T, Kuto F, Watanabe Y, Fujino Y, Hirasawa Y, Tokuhiro H . Bone marrow sinus and cell egress in human leukaemia: A morphometric study of core biopsies using wide-field electron microscopy. Br J Haematol 1986; 63: 737–747.
Tavassoli M . The marrow-blood barrier. Br J Haematol 1979; 41: 297–302.
Matsuo T . In vitro modulation of alkaline phosphatase activity in neutrophils from patients with chronic myelogenous leukemia by monocyte-derived activity. Blood 1986; 67: 492–497.
Pedersen B, Hayhoe FGJ . Relation between phagocytic activity and alkaline phosphatase content of neutrophils in chronic myeloid leukaemia. Br J Haematol 1971; 21: 257–260.
Perillie PE, Finch S . The local exudative cellular response in leukemia. J Clin Invest 1960; 39: 1353–1357.
Perillie PE, Finch SC . Alkaline phosphatase activity of exudative leukocytes in acute leukemia. Blood 1961; 18: 572–580.
Broxmeyer HD, Mendelsohn N, Moore MAS . Abnormal granulocyte feedback regulation of colony forming and colony stimulating activity-producing cells from patients with chronic myelogenous leukemia. Leukemia Res 1977; 1: 3–12.
Odeberg H, Olofsson T, Olsson I . Granulocyte function in chronic granulocytic leukaemia. I. Bacterial and metabolic capabilities during phagocytosis in isolated granulocytes. Br J Haematol 1975; 29: 427–441.
Baker MA, Taub RN, Kanani A . Increased activity of a specific sialyltransferase in chronic myelogenous leukemia. Blood 1985; 66: 1068–1071.
Nojiri H, Takaku F, Ohta M . Changes in glycosphingolipid composition during differentiation of human leukemic granulocytes in chronic myelogenous leukemia compared with in vitro granulocytic differentiation of human promyelocytic leukemia cell line HL-60. Cancer Res 1985; 45: 6100–6106.
Bednarek JM, Knight RD, Taylor G, Evans WH . Progressive loss of phenotypic proteins in mature granulocytes before the onset of blast crisis in human chronic myelogenous leukemia. J Natl Cancer Inst 1988; 80: 251–257.
Evans WH, Bednarek JM, Alvarez VL . Direct analysis of differentiation proteins in normal and leukemic human granulocytosis by high-performance liquid chromatography. J Natl Cancer Inst 1985; 75: 227–235.
Olofsson T, Odeberg H, Olsson I . Granulocyte function in chronic granulocytic leukemia. II. Bactericidal capacity phagocytic rate, oxygen consumption, and granule protein composition in isolated granulocytes. Blood 1976; 48: 581–593.
Zingde SM, Mungikar AM, Chhajlani V, Advani SH, Gothoskar BP . Plasma membranes from normal and chronic myeloid leukemic granulocytes: isolation and two-dimensional polyacrylamide gel electrophoretic analysis. Cancer Biochem Biophys 1985; 7: 333–342.
Banerjee TK, Senn H, Holland JF . Comparative studies on localized leukocyte mobilization in patients with chronic myelocytic leukemia. Cancer 1972; 29: 637–644.
Senn HJ, Rhomberg WU, Jungi WF . Strung der leukozytren Abwehrfunktion als paraneoplastiches Syndrom bei Hmoblastosen. Schweiz Med Wochenschr 1971; 101: 466–470.
Anklesaria PN, Advani SH, Bhisey AN . Defective chemotaxis and adherence in granulocytes from chronic myeloid leukemia (CML) patients. Leukemia Res 1985; 9: 641–648.
Brandt L, Mitelman F, Panani A, Lenner HC . Extremely long duration of chronic myeloid leukaemia with Ph1 negative and Ph1 positive bone marrow cells. Scand J Haematol 1976; 16: 321–325.
Corberand J, Laharraque P Deharrard B, Nyuyen F, Pris J . Phagocytosis in myeloproliferative disorders. Am J Clin Pathol 1980; 74: 301–305.
El-Maallem H, Fletcher J . Defective hydrogen-peroxidase production in chronic granulocytic neutrophils. Br J Haematol 1979; 41: 49–55.
Goldman JM, Catovsky D . Function of the phagocytic leukocytes in leukemia. Br J Haematol 1972; 23(Suppl): 223–230.
Penny R, Galton DAG . Studies on neutrophil function. II. Pathological aspects. Br J Haematol 1966; 12: 633–645.
Penny R, Galton DAG, Scott JT, Eisen V . Studies on neutrophile function. I. Physiological and pharmacological aspects. Br J Haematol 1966; 12: 623–632.
Rosner F, Valmont J, Kozinn PJ, Caroline L . Leukocyte function in patients with leukemia. Cancer 1970; 25: 835–842.
Tornyos K . Phagocytic activity of cells of the inflammatory exudate in human leukemia. Cancer Res 1967; 27: 1756–1760.
Whittaker JA, Khurshid M, Hughes HR . Neutrophil function in chronic granulocytic leukemia before and after busulphan treatment. Br J Haematol 1974; 28: 541–549.
Anklesaria PN, Bhisey AN . Studies on electrophoretic mobility of leucocytes from chronic myeloid leukemia patients. Indian J Exp Biol 1985; 23: 609–612.
Bhisey AN, Rao SGA, Advani SH, Ray V . Agglutination of granulocytes from chronic myeloid leukaemia by concanavalin. Acta Haematol (Basel) 1980; 63: 211–216.
Zingde SM, Advani SH, Gothoskar BP . Plasma membrane proteins from human normal and chronic myeloid leukemic granulocytes: identification and partial characterization of the concanavalin A-binding and detergent resistant proteins. Blut 1987a; 55: 89–100.
Zingde SM, Anklesaria PN, Advani SH, Bhisey AN, Gothoskar BP . Differential endocytosis of fluorescein isothiocyanate-concanavalin A by normal and chronic myeloid leukemic granulocytes. Blut 1987b; 55: 81–88.
Brandt L . Adhesiveness to glass and phagocytic activity of neutrophilic leukocytes in myeloproliferative diseases. Scand J Haematol 1965; 2: 126–136.
Taub RN, Baker MA, Madyastha KR . Masking of neutrophil surface lectin-binding sites in chronic myelogenous leukemia (CML). Blood 1980; 55: 294–298.
Eaves AC, Cashman JD, Gaboury LA, Kalousek DK, Eaves CJ . Unregulated proliferation of primitive chronic myeloid leukemia progenitors in the presence of normal marrow-adherent cells. Proc Natl Acad Sci USA 1986; 83: 5306–5310.
Gordon MY, Dowding CR, Riley GP, Goldman JM, Greaves MF . Altered adhesive interactions with marrow stroma of hematopoietic progenitor cells in chronic myeloid leukemia. Nature 1987; 328: 342–344.
Verfaillie CM, McCarthy JB, McGalve PB . Mechanisms underlying abnormal trafficking of malignant progenitors in chronic myelogenous leukemia. Decreased adhesion to stroma and fibronectin but increased adhesion to the basement membrane components laminin and collagen Type IV. J Clin Invest 1992; 90: 1232–1241.
Dowding C, Guo AP, Maisin D, Gordon MY, Goldman JM . The effects of interferon-alpha on the proliferation of CML progenitor cells in vitro are not related to the precise position of the M-BCR breakpoint. Br J Haematol 1991; 77: 165–171.
Bhatia R, McGlave PB, Verfaillie CM . Treatment of marrow stroma with interferon-alpha restores normal beta 1 integrin-dependent adhesion of chronic myelogenous leukemia hematopoietic progenitors. Role of MIP-1 alpha. J Clin Invest 1995; 96: 931–939.
Guba SC, Emerson SG . Hematopoietic regulation of stem cell dynamics in chronic myelogenous leukemia. In: Deisseroth AB, Arlinghaus RB (eds). Chronic Myelogenous Leukemia: Molecular Approaches to Research and Therapy. New York: Marcel Dekker, 1991, pp 337–347.
Abboud CN, Lichtman MA . Chapter 4: structure of the marrow in Part II General Hematology. In: Beutler E, Lichtman MA, Coller BS, Kipps TJ (eds). Williams Hematology, 5th edn. New York: McGraw-Hill, Inc. Health Professions Division, 1995, pp 25–38.
Lichtman MA . The ultrastructure of the hematopoietic environment of marrow. A review. Exp Hematol 1981; 9: 391–410.
Singer JW, Keating A, Wight TN . The human haematopoietic microenvironment. In: Hoffbrand AV (ed). Recent Advances in Haematology, Vol. 4. Edinburgh: Churchill Livingstone, 1985, pp 1.
Maniatis AK, Amsel S, Mitus WJ, Coleman N . Chromosome pattern of bone marrow fibroblasts in patients with chronic granulocytic leukaemia. Nature 1969; 222: 1278–1279.
Bhatia R, McGlave PB, Dewald GW, Blazar BR, Verfaillie CM . Abnormal function of the bone marrow microenvironment in chronic myelogenous leukemia: role of malignant stromal macrophages. Blood 1995; 85: 3636–3645.
Nirsimloo N, Gordon MY . Progenitor cells in the blood and marrow of patients with chronic phase chronic myeloid leukaemia respond differently to macrophage inflammatory protein-1 alpha. Leukemia Res 1995; 19: 319–323.
Singer JW, Andrews III DF, Nemunaitis JJ . Marrow stromal cells, growth factors, and the c-abl proto-oncogene. In: Deisseroth AB, Arlinghaus RB (eds). Chronic myelogenous leukemia: Molecular Approaches to Research and Therapy. New York: Marcel Dekker, 1991, pp 309–335.
Taub RN, Hindenburg AA, Baker MA . Brief communication: regeneration of membrane sialic acid after neuraminidase treatment of leukemic granulocytes. Leukemia Res 1985; 19: 507–510.
Baker MA, Kanani A, Hindeburg A, Taub RN . Changes in the granulocyte membrane following chemotherapy for chronic myelogenous leukemia. Br J Haematol 1986; 62: 431–438.
Kanani A, May C, Sutherland DR, Hindenburg A, Taub R N, Mills GB et al. Altered rise of FMLP stimulated free cytosolic calcium [Ca2+ in CML granulocytes. Blood 1987; 70(Suppl 1): 90a (Abstract 214).
Yuo A, Kitagawa S, Okabe T, Urabe A, Komatsu Y, Itoh S et al. Recombinant human granulocyte colony-stimulating factor repairs the abnormalities of neutrophils in patients with myelodysplastic syndromes and chronic myelogenous leukemia. Blood 1987; 70: 404–411.
Oseas R, Yang H-H, Baehner RL, Boxer LA . Lactoferrin: a promoter of polymorphonuclear leukocyte adhesiveness. Blood 1981; 57: 939–945.
Bhatia R, Munthe HA, Williams AD, Zhang F, Forman SJ, Slovak ML . Chronic myelogenous leukemia primitive hematopoietic progenitors demonstrate increased sensitivity to growth factor-induced proliferation and maturation. Exp Hematol 2000; 28: 1401–1412.
Brummendorf TH, Holyoake TL, Rufer N, Barnett MJ, Schulzer M, Eaves CJ et al. Plenary paper: prognostic implications of differences in telomere length between normal and malignant cells from patients with chronic myeloid leukemia measured by flow cytometry. Blood 2000; 95: 1883–1890.
Kiger AA, Jones DL, Schulz C, Rogers MB, Fuller MT . Stem cell self-renewal specified by Jak-Stat activation in response to a support cell cue. Science 2001; 294: 2542–2545.
Tran J, Brenner TJ, DiNardo S . Somatic control over the germline stem cell lineage during Drosophila spermatogenesis. Nature 2000; 407: 754–757.
Tulina N, Matunis E . Control of stem cell self-renewal in Drosophila spermatogenesis by JAK-STAT signaling. Science 2001; 294: 2546–2549.
Matsuda T, Nakamura T, Nakao K, Arai T, Katsuki M, Heike T, Yokota T . STAT3 activation is sufficient to maintain an undifferenti-ated state of mouse embryonic stem cells. EMBO J 1999; 18: 4261–4269.
Iscove NN, Nawa K . Hematopoietic stem cells expand during serial transplantation in vivo without apparent exhaustion. Curr Biol 1997; 7: 805–808.
Jordan C, Lemischka IR . Clonal systemic analysis of long-term hematopoiesis in the mouse. Genes Dev 1990; 4: 220–232.
Metcalf D . Stem cells, pre-progenitor cells and lineage-committed cells: are our dogmas correct? Ann NY Acad Sci 1999; 872: 289–304.
Cui YF, Lord BI, Woolford LB, Testa NG . The relative spatial distribution of in vitro-CFCs in the bone marrow, responding to specific growth factors. Cell Prolif 1996; 5: 243–257.
Clarkson BD, Fried J . Changing concepts of treatment in acute leukemia. Med Clin North Am 1971; 55: 561–600.
Fokas AS, Keller JB, Clarkson BD . Mathematical model of granulocytopoiesis and chronic myelogenous leukemia. Cancer Res 1991; 51: 2084–2091.
Stryckmans P, Cronkite EP, Fache F, Fliedner TM, Ramos J . Deoxyribonucleic acid synthesis time of erythropoietic and granulopoietic cells in human beings. Nature 1966; 211: 717–720.
Vincent PC, Cronkite EP, Greenberg ML, Kirsten C, Schiffer LM, Stryckmans PA . Leukocyte kinetics in chronic myeloid leukemia. I. DNA synthesis time in blood and marrow myelocytes. Blood 1969; 33: 843–850.
Baccarini M, Killmann SA . Cytokinetic studies in chronic myeloid leukaemia: evidence for early presence of abnormal myeloblasts. Scand J Haematol 1972; 9: 283–292.
Clarkson B, Strife A, Fried J, Gulati S . Cytokinetics and cancer treatment. In: Freireich EJ, Frei E (eds). Proceedings of the General Motors Cancer Research Foundation (monograph). Philadelphia: JB Lippincott, 1986a, pp 131–190.
Cronkite EP, Vincent PC . Granulocytopoiesis. In: Stohlman F (ed). Hemopoietic Cellular Proliferation. New York: Grune & Stratton Publ Co, 1970, pp 211–228.
Greenberg ML, Chanana AD, Cronkite EP, Giacomelli G, Rai KR, Schiffer LM et al. The generation time of human leukemic myeloblasts. Lab Invest 1972; 26: 245–252.
Killmann SA, Cronkite EP, Robertson JS, Fliedner TM, Bond VP . Estimation of phases of the life cycle of leukemic cells from labeling in human beings in vivo with tritiated thymidine. Lab Invest 1963; 12: 671–684.
Killmann SA . Acute leukemia: the kinetics of leukemic blast cells in man: an analytical review. Semin Hematol 1968; 1: 38–102.
Moore MAS, Williams N, Metcalf D . In vitro colony formation by normal and leukemic human hematopoietic cells: characterization of the colony forming cells. J Natl Cancer Inst 1973; 50: 603–623.
Rondanelli GE, Magliulo E, Giraldi A, Cario EP . The chronology of the mitotic cycle of human granulocytopoietic cells. Blood 1967; 30: 557–568.
Schmid JR, Kiely JM, Tauxe WN, Owen Jr CA . Cell proliferation in leukemia during relapse and remission. I. DNA and RNA synthesis of leukemic cells in the bone marrow in vitro. Acta Haematol 1966; 36: 313–322.
Stryckmans P, Debusscher L, Peltzer T, Socquet M . Variations of the proliferative activity of leukemic myeloblasts related to the stage of the disease. In: Bessis M, Brecher G (eds). Unclassifiable Leukemias. New York: Springer-Verlag Publishing Co., 1975, pp 239.
Vincent PC . Cell kinetics of the leukemias. In: Gunz F, Baikie AG (eds). Leukemia, 3rd edn. New York: Grune & Stratton Publishing Co., 1974, pp. 189–221.
Stryckmans P, Debusscher L, Socquet M . Regulation of bone marrow myeloblast proliferation in chronic myeloid leukemia. Cancer Res 1976; 36: 3034–3038.
Stryckmans P, Debusscher L, Collard E . Cell kinetics in chronic granulocytic leukaemia (CGL). Clin Haematol 1977; 6: 21–40.
Cheng T, Rodrigues N, Dombkowski D, Stier S, Scadden DT . Stem cell repopulation efficiency but not pool size is governed by p27kip1. Nat Med 2000; 6: 1235–1240.
Eaves C, Cashman J, Eaves A . Defective regulation of leukemic hematopoiesis in chronic myeloid leukemia. Leukemia Res 1998; 22: 1085–1096.
Gesbert F, Sellers WR, Signoretti S, Loda M, Griffin JD . BCR/ABL regulates expression of the cyclin-dependent kinase inhibitor p27Kip1 through the phosphatidylinositol 3-kinase/AKT pathway. J Biol Chem 2000; 275: 39223–39230.
Traycoff CY, Halstead B, Rice S, McMahel J, Srour EF,, Cornetta K . Chronic myelogenous leukaemia CD34+ cells exit G0/G1 phases of cell cycle more rapidly than normal marrow CD34+ cells. Br J Haematol 1998; 203: 759–767.
Clarkson B, Ota K, O'Connor A, Karnofsky DA . Production of granulocytes by the spleen in chronic granulocytic leukemia (CGL). J Clin Invest 1963; 42: 924.
Athens JW, Raab SO, Haab OP, Boggs DR, Ashenbrucker H, Cartwright GE et al. Leukokinetic studies: X. Blood granulocyte kinetics in chronic myelocytic leukemia. J Clin Invest 1965; 44: 765–777.
Chervenick PA, Boggs DR . Granulocyte kinetics in chronic myelocytic leukemia. Ser Haematol 1968; 1: 24–37.
Galbraith PR . Studies on the longevity, sequestration and release of the leukocytes in chronic myelogenous leukemia. J Assoc Med Can 1966; 95: 511–521.
Galbraith PR, Abu-Zahra HT . Granulopoiesis in chronic granulocytic leukaemia. Br J Haematol 1972; 22: 135–143.
Mauer AM, Jarrold T . Granulocyte kinetic studies in patients with proliferative disorders of the bone marrow. Blood 1963; 22: 125–138.
Morley AA . A neutrophil cycle in healthy individuals. Lancet 1966; 2: 1220–1222.
Morley AA, King-Smith EA, Stohlman Jr F . The oscillatory nature of hemopoiesis. In: Stohlman Jr F (ed). Hemopoietic Cellular Proliferation. New York: Grune & Stratton, 1970. pp 3–14.
King-Smith EA, Morley A . Computer simulation of granulopoiesis-normal and impaired granulopoiesis. Blood 1970; 36: 254–262.
Morley A, Stohlman Jr F . Cyclophosphamide-induced cyclical neutropenia. An animal model of a human periodic disease. N Engl J Med 1970; 282: 643–646.
Morley AA, Carew JP, Baikie AG . Familial cyclical neutropenia. Br J Haematol 1967; 13: 719–738.
Jakubowski AA, Souza L, Kelly F, Fain K, Budman D, Clarkson B et al. Effects of human granulocyte colony-stimulating factor in a patient with idiopathic neutropenia. N Engl J Med 1989; 320: 38–42.
Haurie C, Dale DC, Mackey MC . Occurrence of periodic oscillations in the differential blood counts of congenital, idiopathic, and cyclical neutropenic patients before and during treatment with G-CSF. Exp Hematol 1999a; 27: 401–409.
Fortin P, Mackey MC . Periodic chronic myelogenous leukaemia: spectral analysis of blood cell counts and aetiological implications. Br J Haematol 1999; 104: 336–345.
Gatti RA, Robinson WA, Deinard AS, Nesbit M, McCullough JJ, Ballow M et al. Cyclic leukocytosis in chronic myelogenous leukemia: new perspectives on pathogenesis and therapy. Blood 1973; 41: 771–782.
Morley AA, Baikie AG, Galton DAG . Cyclic leucocytosis as evidence for retention of normal homoeostatic control in chronic granulocytic leukaemia. Lancet 1967; 2: 1320–1323.
Shadduck RK, Winkelstein A, Nunna NG . Cyclic leukemic cell production in CML. Cancer 1972; 29: 399–401.
Vodopick H, Rupp EM, Edwards CL, Goswitz FA, Beauchamp JJ . Spontaneous cyclic leukocytosis and thromobocytosis in chronic granulocytic leukemia. N Engl J Med 1972; 286: 284–290.
Kennedy BJ . Cyclic leukocyte oscillations in chronic myelogenous leukemia during hydroxyurea therapy. Blood 1970; 35: 751–760.
Haurie C, Person R, Dale DC, Mackey MC . Hematopoietic dynamics in grey collies. Exp Hematol 1999b; 27: 1139–1148.
Haurie C, Dale DC, Rudnicki R, Mackey MC . Modeling complex neutrophil dynamics in the grey collie. J Theoret Biol 2000; 204: 505–519.
Duvall CP, Perry S . The use of 51-chromium in the study of leukocyte kinetics in chronic myelocytic leukemia. J Lab Clin Med 1968; 71: 614–628.
Cronkite EP . Normal human granulocytopoiesis. In: The Proliferation and Spread of Neoplastic Cells. Baltimore: Williams & Wilkins Publishing Co, 1968, pp 281–294.
Perry S, Moxley III JH, Weiss GH, Zelen M . Studies of leukocyte kinetics by liquid scintillation counting in normal individuals and in patients with chronic myelocytic leukemia. J Clin Invest 1966; 45: 1388–1399.
Scott JL, McMillan R, Davidson JG, Marino JV . Leukocyte labeling with 51chromium. II. Leukocyte kinetics in chronic myeloid leukemia. Blood 1971; 38: 162–173.
Moxley JH, Perry S, Weiss GH, Zelen M . Return of leucocytes to the bone marrow in chronic myelocytic leukaemia. Nature 1965; 208: 1281–1282.
Uchida N . Leukokinetic studies in peripheral blood. II. Granulocyte kinetics in chronic myelocytic leukemia. Acta Haematol Japon 1971; 34: 186–204.
Kerr JFR, Wylie AH, Currie AR . Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 1972; 26: 239–257.
Koury MJ . Minireview: programmed cell death (apoptosis) in hematopoiesis. Exp Hematol 1992; 20: 391–394.
Lockshin RA, Williams CM . Programmed cell death. II. Endocrine potentiation of the breakdown of the intersegmental muscles of silkmoths. J Insect Physiol 1964; 10: 643–649.
Squier MKT, Sehnert AJ, Cohen JJ . Apoptosis in leukocytes. J Leukocyte Biol 1995; 57: 2–10.
Daley GQ, Baltimore D . Transformation of an interleukin 3-dependent hematopoietic cell line by the chronic myelogenous leukemia-specific p210bcr/abl protein. Proc Nat Acad Sci USA 1988; 85: 9312–9316.
Cortez D, Kadlec L, Pendergast AM . Structural and signaling requirements for BCR-ABL-mediated transformation and inhibition of apoptosis. Mol Cell Biol 1995; 15: 5531–5541.
Hariharan IK, Adams JM, Cory S . bcr-abl oncogene renders myeloid cell line factor independent: potential autocrine mechanisms in chronic myeloid leukemia. Oncogene Res 1988; 3: 387–399.
Kabarowski JHS, Allen PB, Wiedemann LM . A temperature sensitive p210 BCR-ABL mutant defines the primary consequences of BCR-ABL tyrosine kinase expression in growth factor dependent cells. EMBO J 1994; 13: 5887–5895.
Laneuville P, Timm M, Hudson AT . bcr/abl expression in 32D cl3(G) cells inhibits apoptosis induced by protein tyrosine kinase inhibitors. Cancer Res 1994; 54: 1360–1366.
Li L, Keating MJ, Plunkett W, Yang LY . Fludarabine-mediated repair inhibition of cisplatin-induced DNA lesions in human chronic myelogenous leukemia-blast crisis K562 cells: induction of synergistic cytotoxicity independent of reversal of apoptosis resistance. Mol Pharmacol 1997; 52: 798–806.
McGahon A, Bissonnette R, Schmitt M, Cotter KM, Green DR, Cotter TG . Rapid Communication. BCR-ABL maintains resistance of chronic myelogenous leukemia cells to apoptotic cell death. Blood 1994; 83: 1179–1187.
Munker R, Marini F, Jiang S, Savary C, Owen-Schaub L, Andreeff M . Expression of CD95(FAS) by gene transfer does not sensitize K562 to Fas-killing. Hematol Cell Ther 1997; 39: 75–78.
Riordan FA, Bravery CA, Mengubas K, Ray N, Borthwick NJ, Akbar AN et al. Herbimycin A accelerates the induction of apoptosis following etoposide treatment or gamma-irradiation of bcr/abl-positive leukaemia cells. Oncogene 1998; 16: 1533–1542.
Scheid S, Heinzinger M, Waller CF, Lange W . Bcl-2 mRNA-targeted ribozymes: effects on programmed cell death in chronic myelogenous leukemia cell lines. Ann Hematol 1998; 76: 117–125.
Smetsers TFCM, Skorski T, van de Locht LTF, Wessels HMC, Pennings AHM, de Witte T et al. Antisense BCR-ABL oligonucleotides induce apoptosis in the Philadelphia chromosome-positive cell line BV-173. Leukemia 1994; 8: 129–140.
Evans CA, Owen-Lynch PJ, Whetton AD, Dive C . Advances in brief. Activation of the Abelson tyrosine kinase activity is associated with suppression of apoptosis in hemopoietic cells. Cancer Res 1993; 53: 1735–1738.
Bedi A, Zehnbauer BA, Barber JP, Sharkis SJ, Jones RJ . Rapid communication: inhibition of apoptosis by BCR-ABL in chronic myeloid leukemia. Blood 1994; 83: 2038–2044.
Gora-Tybor J, Deininger MW, Goldman JM, Melo JV . The susceptibility of Philadelphia chromosome positive cells to FAS-mediated apoptosis is not linked to the tyrosine kinase activity of BCR-ABL. Br J Haematol 1998; 103: 716–720.
Handa H, Hegde UP, Kotelnikov VM, Mundle SD, Dong LM, Burke P et al. Bcl-2 and c-myc expression, cell cycle kinetics and apoptosis during the progression of chronic myelogenous leukemia from diagnosis to blastic phase. Leukemia Res 1997; 21: 479–489.
Horita M, Andreu EJ, Benito A, Arbona C, Sanz C, Benet I et al. Blockade of the Bcr-Abl kinase activity induces apoptosis of chronic myelogenous leukemia cells by suppressing signal transducer and activator of transcription 5-dependent expression of Bcl-xL . J Exp Med 2000; 191: 977–984.
Selleri C, Sato T, Del Vecchio L, Luciano L, Barrett AJ, Rotoli B et al. Involvement of Fas-mediated apoptosis in the inhibitory effects of interferon-alpha in chronic myelogenous leukemia. Blood 1997; 89: 957–964.
Selleri C, Maciejewski JP, Pane F, Luciano L, Raiola AM, Mostarda I et al. Fas-mediated modulation of Bcr/Abl in chronic myelogenous leukemia results in differential effects on apoptosis. Blood 1998; 92: 981–989.
Stuppia L, Calabrese G, Peila R, Guanciali-Franchi P, Morizio E, Spadano A, Palka G . p53 loss and point mutations are associated with suppression of apoptosis and progression of CML into myeloid blastic crisis. Cancer Genet Cytogenet 1997; 98: 28–35.
Amarante-Mendes GP, McGahon AJ, Nishioka WK, Afar DE, Witte ON, Green DR . Bcl-2-independent Bcr-Abl-mediated resistance to apoptosis: protection is correlated with upregulation of Bcl-xL . Oncogene 1998; 16: 1383–1390.
Fang G, Kim CN, Perkins CL . CGP57148B (STI571) induces differentiation and apoptosis and sensitizes Bcr-Abl-positive human leukemia cells to apoptosis due to antileukemic drugs. Blood 2000; 96: 2246–2253.
Ray S, Bullock G, Nunez G, Tang C, Ibrado AM, Huang Y et al. Enforced expression of Bcl-xS induces differentiation and sensitizes CML-blast crisis K562 cells to Ara-C mediated differentiation and apoptosis. Cell Growth Differ 1996; 7: 1617–1623.
Albrecht T, Schwab R, Henkes M, Peschel C, Huber C, Aulitzky WE . Primary proliferating immature myeloid cells from CML patients are not resistant to induction apoptosis by DNA damage and growth factor withdrawal. Br J Haematol 1996; 95: 501–507.
Amos TAS, Lewis JL, Grand FH, Gooding RP, Goldman JM, Gordon MY . Apoptosis in chronic myeloid leukaemia: normal responses by progenitor cells to growth factor deprivation, X-irradiation and glucocorticoids. Br J Haematol 1995; 91: 387–393.
Gordon MY, Dazzi F, Marley SB, Lewis JL, Nguyen D, Grand FH et al. Cell biology of CML cells. Leukemia 1999; 1(Suppl): S65–S71.
Roger R, Isaad C, Pallardy M, Leglise M-C, Turhan AG, Bertoglio J et al. BCR-ABL does not prevent apoptotic death induced by human natural killer or lymphokine activated killer cells. Blood 1996; 87: 1113–1122.
Vigneri P, Wang JY . Induction of apoptosis in chronic myelogenous leukemia cells through nuclear entrapment of BCR-ABL tyrosine kinase. Nature Med 2001; 7: 228–234 (Comment pp 156–157).
Wang JYJ . Regulation of cell death by the Abl tyrosine kinase. Oncogene 2000; 19: 5643–5650.
Marley SB, Deininger MW, Davidson RJ, Goldman JM, Gordon MY . The tyrosine kinase inhibitor STI571, like interferon-α, preferentially reduces the capacity for amplification of granulocyte-macrophage progenitors from patients with chronic myeloid leukemia. Exp Hematol 2000; 28: 551–557.
Strife A, Perez A, Lambek C, Wisniewski D, Bruno S, Darzynkiewicz Z et al. Characterization of lineage-negative blast subpopulations derived from normal and chronic myelogenous leukemia bone marrows and determination of their responsiveness to human c-kit ligand. Cancer Res 1993; 53: 401–409.
Strife A, Perez A, Lambek C, Wisniewski D, Bruno S, Darzynkiewicz Z et al. Differences in the composition and in the efficiency of red cell production of normal and CML erythroid progenitor populations are highlighted by response to human c-kit ligand. Leukemia Res 1993; 17: 799–807.
Strife A, Lambek C, Wisniewski D, Arlin Z, Thaler H, Clarkson B . Proliferative potential of subpopulations of granulocyte–macrophage progenitor cells in normal subjects and chronic myelogenous leukemia patients. Blood 1983; 62: 389–397.
Strife A, Lambek C, Wisniewski D, Gulati S, Gasson JC, Golde DW et al. Activities of four purified growth factors on highly enriched human hematopoietic progenitor cells. Blood 1987; 69: 1508–1523.
Clarkson B, Ohkita T, Ota K, Fried J . Studies of cellular proliferation in human leukemia I: estimation of growth rates of leukemic and normal hematopoietic cells in two adults with acute leukemia given single injections of tritiated thymidine. J Clin Invest 1967a; 46: 506–529.
Clarkson BD . Review of recent studies of cellular proliferation in acute leukemia. In: Perry S (ed). Human Tumor Cell Kinetics. Bethesda, Maryland: National Cancer Institute Monograph 30/584, 1969a, pp 81–120.
Clarkson BD, Fried J, Ogawa M . Discussion: magnitude of proliferating fraction and rate of proliferation of populations of leukemic cells in man. In: Recent Results in Cancer Research, Monograph 17: Symposium on Normal and Malignant Cell Growth, New York: Springer-Verlag, 1969b, pp 175–185.
Clarkson B, Fried J, Strife A, Sakai Y, Ota K, Ohkita T . Studies of cellular proliferation in human leukemia III: behavior of leukemic cells in three adults with acute leukemia given continuous infusions of 3H Thymidine for 8 or 10 days. Cancer 1970; 25: 1237–1260.
Clarkson B, Strife A, Fried J, Sakai Y, Ota K, Ohkita T et al. Studies of cellular proliferation in human leukemia IV: behavior of normal hematopoietic cells in 3 adults with acute leukemia given continuous infusions of 3H-Thymidine for 8 or 10 days. Cancer 1970; 26: 1–19.
Yen A, Fried J, Kitahara T, Strife A, Clarkson BD . The kinetic significance of cell size: I. Variation of cell cycle parameters with size measured at mitosis. Exp Cell Res 1975; 95: 295–302.
Yen A, Fried J, Kitahara T, Strife A, Clarkson BD . The kinetic significance of cell size: II. Size distributions of resting and proliferating cells during interphase. Exp Cell Res 1975; 95: 303–310.
Holyoake TL, Jiang X, Drummond MW, Eaves AC, Eaves CJ . Review: elucidating critical mechanisms of deregulated stem cell turnover in the chronic phase of chronic myeloid leukemia. Leukemia 2002; 16: 549–558.
Wisniewski D, Strife A, Clarkson B . Co-detection of chimeric BCR/ABL (target) and β-actin (control) messenger RNA in individual CFU-GM colonies derived from CML patients using the polymerase chain reaction. Leukemia Res 1991; 15: 867–874.
Brandt J, Briddell RA, Srour EF, Leemhuis TB, Hoffman R . Role of c-kit ligand in the expansion of human hematopoietic progenitor cells. Blood 1992; 79: 634–641.
Galli SJ, Zsebo KM, Geissler EN . The kit ligand, stem cell factor. Adv Immunol 1994; 55: 1–96.
McNiece IK, Langley KE, Zsebo KM . Recombinant human stem cell factor synergises with GM-CSF, G-CSF, IL-3 and epo to stimulate human progenitor cells of the myeloid and erythroid lineage. Exp Hematol 1991; 19: 226–231.
Eaves AC, Eaves CJ . Abnormalities in the erythroid progenitor compartments in patients with chronic myelogenous leukemia (CML). Expe Hematol 1979; 7: 65–75.
Singer JW, Adamson JW, Arlin ZA, Kempin SJ, Clarkson BD, Fialkow PJ . Chronic myelogenous leukemia. In vitro studies of hematopoietic regulation in a patient undergoing intensive chemotherapy. J Clin Invest 1981; 67: 1593–1598.
Wognum AW, Krystal G, Eaves CJ, Eaves AC, Lansdorp PM . Increased erythropoietin-receptor expression on CD34-positive bone marrow cells from patients with chronic myeloid leukemia. Blood 1992; 79: 642–649.
Marley SB, Lewis JL, Goldman JM, Gordon MY . Abnormal kinetics of colony formation by erythroid burst-forming units (BFU-E) in chronic myeloid leukaemia. Br J Haematol 1996; 93: 878–883.
Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM et al. Efficacy and safety of a specific inhibitor of the Bcr-Abl tyrosine kinase in chronic myeloid leukemia. N Engl J Med 2001a; 344: 1031–1037.
Nagar B, Bornmann W, Pellicena P, Schindler T, Young M, Veach D et al. Crystal structure of the c-abl tyrosine kinase domain in complex with PD173955 and STI-571. Cancer Res 2002; 15: 4236–4243.
Veach DR, Swendeman S, Nagar B, Wisniewski D, Strife A, Lambek CL et al. Towards picomolar inhibition of Bcr-Abl: Synthesis and evaluation of a focused library of pyrido-[23-d]-pyrimidine tyrosine kinase inhibitors guided by X-ray crystallography and molecular modeling. Proc Am Assoc Cancer Res 2002; 43: 847 (Abstract 4199).
Trumpp-Kallmeyer S, Rubin JR, Humblet C, Hamby JM, Showalter HDH . Development of a binding model to protein tyrosine kinases for substituted pyrido[2,3-d]pyrimidine inhibitors. J Med Chem 1998; 41: 1752–1763.
Berman E, Jhanwar S, McBride M, Strife A, Wisniewski D, Lambek C et al. Characterization of two novel sublines established from a human megakaryoblastic leukemic cell line transfected with p210bcr-abl. Leukemia Res 2000; 24: 289–297.
Moore S, Haylock DN, Lovesque JP, Mcdiarmid LA, Samuels LM, To LB et al. Stem cell factor as a single agent induces selective proliferation of the Philadelphia chromosome positive fraction of CML CD34+ cells. Blood 1998; 92: 2461–2470.
Moore S, Mcdiarmid LA, Hughes TP . Stem cell factor and chronic myeloid leukemia Cd34+ cells. Leukemia Lymphoma 2000; 38: 211–220.
Pierce A, Spooncer E, Ainsworth S, Whetton AD . BCR-ABL alters the proliferation and differentiation response of multipotent hematopoietic cells to stem cell factor. Oncogene 2002; 21: 3068–3075.
Metcalf D . Review Article. Hematopoietic regulators: redundancy or subtlety? Blood 1993; 82: 3515–3523.
Muta K, Krantz SB, Bondurant MC, Dai C-H . Stem cell factor retards differentiation of normal human erythroid progenitor cells while stimulating proliferation. Blood 1995; 86: 572–580.
Cervantes F, Robertson JE, Rozman C, Baccarani M, Tura S, Gómez GA et al. Long-term survivors in chronic granulocytic leukemia: a study by the International CGL Prognosis Study Group. Br J Haematol 1994; 87: 293–300.
Hehlmann R, Heimpel H, The German CML Study Group. Randomized comparison of busulfan and hydroxyurea in chronic myelogenous leukemia: prolongation of survival by hydroxyurea. Blood 1993; 82: 398–407.
Monfardini S, Gee T, Fried J, Clarkson B . Survival in chronic myelogenous leukemia: influence of treatment and extent of disease at diagnosis. Cancer 1973; 31: 492–501.
Selleri L, Emilia G, Temperani P . Philadelphia-positive chronic myelogenous leukemia with typical bcr/abl molecular features and atypical, prolonged survival. Leukemia 1989; 3: 538.
Sokal JE, Cox EB, Baccarani M, Tura S, Gomez GA, Robertson JE et al. Prognostic discrimination in ‘good-risk’ chronic granulocytic leukemia. Blood 1984; 63: 789–799.
Sokal JE, Baccarani M, Tura S, Fiacchini M, Cervantes F, Rozman C et al. Prognostic discrimination among younger patients with chronic granulocytic leukemia: relevance to Bone Marrow Transplant. Blood 1985; 66: 1352–1357.
Italian Cooperative Study Group on Chronic Myeloid Leukemia. Prospective confirmation of a prognostic classification for Ph+ chronic myeloid leukaemia. Br J Haematol 1988; 69: 463–466.
Kantarjian HM, Smith TL, McCredie KL . Chronic myelogenous leukemia: a multivariate analysis of the associations of patient characteristics and therapy with survival. Blood 1985; 66: 1326.
Simon W, Segel GB, Lichtman MA . Upper and lower time limits in the decision to recommend marrow transplantation for patients with chronic myelogenous leuykaemia. Br J Haematol 1988; 70: 31–36.
Minot JB, Buckman TE, Isaacs R . Chronic myelogenous leukemia: age, incidence, duration and benefit derived from irradations. JAMA 1924; 82: 1489.
Rushing D, Goldman A, Gibbs G, Howe R, Kennedy BJ . Hydroxyurea versus busulfan in the treatment of chronic myelogenous leukemia. Am J Clin Oncol 1982; 5: 307–313.
Arlin ZA, Mertelsmann R, Berman E, Gee T, Kurland E, Chaganti RSK et al. 13-cis-retinoic acid does not increase the true remission rate and the duration of true remission (induced by cytotoxic chemotherapy) in patients with chronic phase chronic myelogenous leukemia. J Clin Oncol 1985; 3: 473–476.
Clarkson B . Editorial: chronic myelogenous leukemia: is aggressive treatment indicated. J Clin Oncol 1985a; 3: 135–139.
Hester JP, Waddell CC, Coltman CA . Response of chronic myelogenous leukemia patients to COAP-splenectomy. A Southwest Oncology Group Study. Cancer 1984; 54: 1977–1982.
Kantarjian HM, Vellekoop L, McCredie KB, Keating MJ, Hester J, Smith T et al. Intensive combination chemotherapy (ROAP 10) and splenectomy in the management of chronic myelogenous leukemia. J Clin Oncol 1985; 3: 192–200.
Kantarjian HM, Talpaz M, Andersson B, Khouri I, Giralt S, Rios MB et al. High doses of cyclophosphamide, etoposide and total body irradiation followed by autologous stem cell transplantation in the management of patients with chronic myelogenous leukemia. Bone Marrow Transplant 1994; 14: 57–61.
Agiletta M, Piacibello W, Stacchini A, Sanavio F, Gavosto F . In-vitro effect of retinoic acid on normal and chronic myeloid leukemia granulopoiesis. Leukemia Res 1985; 9: 879–883.
Lilly M, Tompkins C, Brown C, Pettit G, Kraft A . Differentiation and growth modulation of chronic myelogenous leukemia cells by bryostatin. Cancer Res 1990; 50: 5520–5525.
Januszewicz E, Rabizadah ER, Maimon Z, Zaizov R, Shaklai M . Inhibition by retinoic acid of myeloid progenitors in chronic myeloid leukemia and myeloproliferative disease: increased sensitivity in blastic phase of chronic myeloid leukemia. Pathology 1988; 20: 1–6.
Wiemann MC, Khorsand J, Poisson L, Frackleton AR, Calabresi P . Evaluation of differentiation-inducing drugs against human chronic myeloid leukemic cells in vivo. Proc Am Assoc Cancer Res 1988; 29: A99676.
Gold EJ, Mertelsmann RH, Itri LM, Gee T, Arlin Z, Kempin S et al. Phase I clinical trial of 13-cis-retinoic acid in myelodysplastic syndrome. Cancer Treat Rep 1983; 67: 981–986.
Hellstrom E, Robert KH, Gahrton G . Therapeutic effects of low-dose cytosine arabinoside, α-interferon, 1-α-hydroxy vitamin D3, and retinoic acid in acute leukemia and myelodysplastic syndromes. Eur J Haematol 1988; 40: 449–459.
Wiernik P, Dutcher J, Paietta E, Hittelman W, Vyas R, Strack M et al. Treatment of promyelocytic blast crisis of chronic myelogenous leukemia with all trans-retinoic acid. Leukemia 1991; 5: 504–509.
Castaigne S, Chomienne C, Daniel M-T, Ballerini P, Berger R, Fenaux P et al. All-trans-retinoic acid as a differentiation therapy for acute promyelocytic leukemia I: clinical results. Blood 1990; 76: 1704–1709.
Chen Z-X, Xue Y-Q, Zhang R, Tao R-F, Xia X-M, Li C et al. A clinical and experimental study on all-trans retinoic acid-treated acute promyelocytic leukemia patients. Blood 1991; 78: 1413–1419.
Frankel SR, Eardley A, Lauwers G, Weiss M, Warrell Jr RP . The ‘retinoic acid syndrome’ in acute promyelocytic leukemia. Ann Intern Med 1992a; 117: 292–296.
Frankel SR, Miller Jr WH, Dmitrovsky E . Retinoic acid and its rearranged receptor in the etiology and treatment of acute promyelocytic leukemia. Oncology 1992b; 6: 74–83.
Warrell Jr RP, Frankel SR, Miller Jr WH, Scheinberg DA, Itri LM, Hittelman WN et al. Differentiation therapy of acute promyelocytic leukemia with tretinoin (all-trans retinoic acid). N Engl J Med 1991; 324: 1385–1393.
Warrell Jr RP, de Thé H, Wang Z-Y, Degos L . Advances in biology and treatment of acute promyelocytic leukemia. N Engl J Med 1993; 329: 177–189.
Allan NC, Richards SM, Shepherd PC . UK Medical Research Council randomised, multicentre trial of interferon-alpha n1 for chronic myeloid leukaemia: improved survival irrespective of cytogenetic response. Lancet 1995; 345: 1392–1397.
Chronic Myeloid Leukemia Trialists' Collaborative Group. Interferon alpha versus chemotherapy for chronic myeloid leukemia: a meta-analysis of seven randomized trials. J Natl Cancer Inst 1997; 89: 1616–1620.
Gale RP, Hehlmann R, Zhang MJ, Hasford J, Goldman JM, Heimpel H et al. Survival with bone marrow transplantation versus hydroxyurea or interferon for chronic myelogenous leukemia. The German CML Study Group. Blood 1998; 91: 1810–1819.
Ozer H, George SL, Schiffer CA . Prolonged subcutaneous administration of recombinant-α 2b interferon in patients with previously untreated Philadelphia chromosome-positive chronic-phase chronic myelogenous leukemia: effect on remission duration and survival: Cancer and Leukemia Group B Study 8583. Blood 1993; 82: 2975.
Tura S, Baccarani M, The Italian Cooperative Study Group on Chronic Myeloid Leukemia. Alpha-interferon in the treatment of chronic myeloid leukemia. Blood 1995; 85: 2999–3002.
Wetzler M, Kantarjian H, Kurzrock R, Talpaz M . (Review) Interferon-alpha therapy for chronic myelogenous leukemia. Am J Med 1995; 99: 402–411.
Hasford J, Pfirrmann M, Helhmann R, Allan NC, Baccarani M, Kluin-Nelemans JC et al. A new prognostic score for survival of patients with chronic myeloid leukemia treated with interferon alfa. Writing Committee for the Collaborative CML Prognostic Factors Project Group. J Nat Cancer Inst 1998; 90: 850–858.
Kloke O, Niederle N, Opalka B, Hawig I, Seeber S, Becher R . Prognostic impact of interferon alpha-induced cytogenetic remission in chronic myelogenous leukaemia: long-term follow-up. Eur J Haematol 1996; 56: 78–81.
Sawyers CL . Genetic interactions between abl and p53: implications for cell growth and transformation. Proceedings of the International Symposium on Chronic Myelogenous Leukemia, Biarritz, France, July 7–9, 1999 (Abstract, p 28).
Sawyers CL . The cell cycle: tyrosine kinase inhibitors in chronic myeloid leukemia. Cancer J Sci Am 1999; 5: 63–69.
Eberle F, Toiron Y, Camerlo J, Lafage M, Sainty D, Arnoulet C et al. Persistence of BCR/ABL mRNA-expressing bone-marrow cells in patients with chronic myelogenous leukemia in complete cytogenetic remission induced by interferon-alpha therapy. Leukemia Lymphoma 1995; 18: 153–157.
Hochhaus A, Lin F, Reiter A . Variable numbers of BCR-ABL transcripts persist in CML patients who achieve complete cytogenetic remission with interferon-alpha. Br J Haematol 1995; 91: 126–131.
Lee M-S, Kantarjian H, Talpaz M, Freireich EJ, Deisseroth A, Trujillo JM et al. Detection of minimal residual disease by polymerase chain reaction in Philadelphia chromosome-positive chronic myelogenous leukemia following interferon therapy. Blood 1992; 79: 1920–1923.
Opalka B, Wandl UB, Becher R, Kloke O, Nagel-Hiemke NM, Moritz T et al. Minimal residual disease in patients with chronic myelogenous leukemia undergoing long-term treatment with recombinant interferon alpha-2b alone or in combination with interferon gamma. Blood 1991; 78: 2188–2193.
Pasternak G, Schultheis B, Heissig B, Horner S, Sick C, Helhmann R . Does long-term culture favor normal clonogenic cells from interferon-treated patients with chronic myelogenous leukemia? Leukemia 1999; 13(Suppl 1): S55 S64.
Galvani D, Cawley C . Mechanism of action of alpha interferon in chronic granulocytic leukaemia: evidence for preferential inhibition of late progenitors. Br J Haematol 1989; 73: 475–479.
Van Etten RA . The molecular pathogenesis of the Philadelphia-positive leukemias: implications for diagnosis and therapy, Chapter 14. In: Freireich EJ, Kantarjian H (eds). Leukemia: Advances, Research and Treatment. Dordrecht, MA: Kluwer Academic Publishers, 1993, pp 295–325.
Choudhury A, Gajewski JL, Liang JC, Popat U, Claxton DF, Kliche KL et al. Use of leukemic dendritic cells for the generation of antileukemic cellular cytotoxicity against Philadelphia chromosome-positive chronic myelogenous leukemia. Blood 1997; 89: 1133–1142.
Chen C-K, Uchida H, Miyakawa Y, Ikeda Y, Kizaki M . Induction of apoptosis by cooperative interaction of ICSBP and PU.1 on the regulation of bcl-2 gene expression in interferon-α-treated CML cells. Blood 2002; 100: 582a (Abstract 2286).
Guilhot F, Chastang C, Michallet M . Interferon alfa-2b combined with cytarabine versus interferon alone in chronic myelogenous leukemia. French Chronic Myeloid Leukemia Study Group. N Engl J Med 1997; 337: 223–229.
Maloisel F, Guerci A, Guyotat D, Ifrah N, Michallet M, Reiffers J et al. Results of a phase II trial of oral cytarabine ocfosfate (YNK01) and interferon alpha-2b for the treatment of chronic myelogenous leukemia patients in chronic phase. France Intergroupe des Leucemies Myeloides Chroniques. Leukemia 2002; 16: 573–580.
Brunstein CG, McGlave PB . The biology and treatment of chronic myelogenous leukemia. Oncology 2001; 1: 23–32.
Horowitz MM, Rowlings PA, Passweg JR . Allogeneic bone marrow transplantation for CML: a report from the International Bone Marrow Transplant Registry. Bone Marrow Transplant 1996; 17(Suppl 3): S5–S6.
van Rhee F, Szydlo RM, Hermans J . Long-term results after allogeneic Bone Marrow Transplant for chronic myelogenous leukemia in chronic phase: a report from the Chronic Leukemia Working Party of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant 1997; 20: 553–560.
Zuffa E, Bandini G, Bonini A, Santucci MA, Martinelli G, Rosti G et al. Prior treatment with alpha-interferon does not adversely affect the outcome of allogeneic BMT in chronic phase chronic myeloid leukemia. Haematologica 1998; 83: 231–236.
Clift RA, Anasetti C . Allografting for chronic myeloid leukaemia. Baillieres Clin Haematol 1997; 10: 319–336.
Lin F, Goldman JM, Cross NC . A comparison of the sensitivity of blood and bone marrow for the detection of minimal residual disease in chronic myeloid leukaemia. Br J Haematol 1994; 86: 683–685.
Lin F, Chase A, Bungey J, Goldman JM, Cross NC . Correlation between the proportion of Philadelphia chromosome-positive metaphase cells and levels of BCR-ABL mRNA in chronic myeloid leukaemia. Genes Chromosomes Cancer 1995; 13: 110–114.
Xu WM, Piao XH, Addy L, Jamal M, Minden MD, Messner HA . Minimal residual disease in bone marrow transplant recipients with chronic myeloid leukemia. Bone Marrow Transplant 1994; 14: 299–306.
Miyamura K, Tahara T, Tanimoto M . Long persistent bcr-abl positive transcript detected by polymerase chain reaction after marrow transplant for chronic myelogenous leukemia without clinical relapse: a study of 64 patients. Blood 1993; 81: 1089–1093.
Radich JP, Gehly G, Gooley T . Polymerase chain reaction detection of the BCR-ABL fusion transcript after allogeneic marrow transplantation for chronic myeloid leukemia: results and implications in 346 patients. Blood 1995; 85: 2632–2638.
Cullis JO, Jiang YZ, Schwarer AP, Hughes TP, Barrett AJ, Goldman JM . Donor leukocyte infusions for chronic myeloid leukemia in relapse after allogeneic bone marrow transplantation. Blood 1992; 79: 1379–1381.
Kolb HJ, Schattenberg A, Goldman JM, Hertenstein B, Jackobsen N, Arcese W et al. Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. Blood 1995; 86: 2041–2050.
Porter DL, Roth MS, McGarigle C, Ferrara JLM, Antin JH . Induction of graft-versus-host disease as immunotherapy for relapsed chronic myeloid leukemia. N Engl J Med 1994; 330: 100–106.
van Rhee F, Lin G, Cullis JO, Spencer A, Cross NC, Chase A et al. Relapse of chronic myeloid leukemia after allogeneic bone marrow transplant: the case for giving donor leukocyte transfusions before the onset of hematologic relapse. Blood 1994; 83: 3377–3383.
Mackinnon S, Papadopoulos EB, Carabasi MH, Reich L, Collins NH, Boulad F et al. Adoptive immunotherapy evaluating escalating doses of donor leukocytes for relapse of chronic myeloid leukemia after bone marrow transplantation: separation of graft-versus-leukemia responses from graft-versus-host disease. Blood 1995a; 86: 1261–1268.
Mackinnon S, Papadopoulos EB, Carabasi MH, Reich L, Collins NH, O'Reilly RJ . Adoptive immunotherapy using donor leukocytes following bone marrow transplantation for chronic myeloid leukemia: is T cell dose important in determining biological response? Bone Marrow Transplant 1995b; 15: 591–594.
Collins Jr RH, Shpilberg O, Drobyski WR . Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol 1997; 15: 433–444.
Drobyski WR, Hessner MJ, Klein JP . T-cell depletion plus salvage immunotherapy with donor leukocyte infusions as a strategy to treat chronic-phase chronic myelogenous leukemia patients undergoing HLA-identical sibling marrow transplantation. Blood 1999; 94: 434–441.
Sehn LH, Alyea EP, Weller E . Comparative outcomes of T-cell-depleted and non-T-cell-depeleted allogeneic Bone Marrow Transplant for chronic myelogenous leukemia: impact of donor lympocyte infusion. J Clin Oncol 1999; 17: 561–568.
Wu CJ, Chen L, Kutok JL, Canning CM, Alyea EP, Yang X-F et al. CML28 and CML66 are GVL-associated antigens that are highly expressed in proliferating cells. Blood 2002; 100: 581a (Abstract 2285).
Zorn E, Wang KL, Hochberg EP, Canning C, Alyea EP, Soiffer RJ et al. Infusion of CD4+ donor lymphocytes induces the expansion of CD8+ donor T cells with cytolytic activity directed against recipient hematopoietic cells. Clin Cancer Res 2002; 8: 2052–2060.
Lee SJ, Kuntz KM, Horowitz MM . Unrelated donor bone marrow transplantation for chronic myelogenous leukemia: a decision analysis. Ann Intern Med 1997; 127: 1080–1088.
McGlave P, Kollman C, Shu XO . The first 1000 unrelated donor transplants for CML: lessons from the National Marrow Donor Program (NMDP) experience. Blood 1996; 88(Suppl 1): 483a (abstract).
Hansen JA, Gooley TA, Martin PJ . Bone marrow transplants from unrelated donors for patients with chronic myeloid leukemia. N Engl J Med 1998; 338: 962–968.
Goldman JM . Autografting for CML: overview and perspectives. Bone Marrow Transplant 1996; 17(Suppl 3): S71–S74.
Reiffers J, Goldman J, Meloni G, Cahn JY, Faberes C, Apperley J et al. Autologous transplantation in chronic myelogenous leukemia: European results. Bone Marrow Transplant 1994; 14(Suppl 3). S51 S54.
Barnett M, Eaves C, Eaves A . Autografting with cultures marrow in chronic myeloid leukemia. Proceedings of the International Symposium on Chronic Myelogenous Leukemia, Biarritz, France July 7–9, 1999 (Abstract, p 62).
Bocchia M, Korontsvit T, Xu Q . Specific human cellular immunity to bcr-abl oncogene-derived peptides. Blood 1996; 87: 3587–3592.
Clark RE . Cytotoxic T-cells against BCR-ABL. Proceedings of the International Symposium on Chronic Myelogenous Leukemia, Biarritz, France, July 7–9, 1999 (Abstract, p 57).
de Fabritiis P, Amadori S, Petti MC . In vitro purging with BCR-ABL antisense oligodeoxynucleotides does not prevent haematologic reconstitution after autologous bone marrow transplantation. Leukemia 1995; 9: 662–664.
Gewirtz AM . Treatment of chronic myelogenous leukemia (CML) with c-myb antisense oligodeoxynucleotides. Bone Marrow Transplant 1994; 14(Suppl 3): S57–S61.
Scheinberg DA . Specific immunotherapy of chronic myelogenous leukemia: Passive and active strategies. Proceedings of the International Symposium on Chronic Myelogenous Leukemia, Biarritz, France, July 7–9, 1999 (Abstract, p 56).
Verfaillie CM, Bhatia R, Miller W . BCR/ABL-negative primitive progenitors suitable for transplantation can be selected from the marrow of most early-chronic phase but not accelerated-phase chronic myelogenous leukemia patients. Blood 1996; 87: 4770–4779.
Carlo-Stella C, Di Nicola M, Tabilio A . Generation of dendritic cells from chronic myelogenous leukemia CD34+ cells. Proceedings of the International Symposium on Chronic Myelogenous Leukemia, Biarritz, France, July 7–9, 1999 (Abstract, p 55).
Nieda M, Nicol A, Kikuchi A . Dendritic cells stimulate the expansion of bcr-abl specific CD8+ T cells with cytotoxic activity against leukemic cells from patients with chronic myeloid leukemia. Blood 1998; 91: 977–983.
Waller CF, Martens UM, Lange W . Letter to the Editor: Philadelphia chromosome-positive cells are equally distributed in AC133+ and AC133C fractions of CD34+ peripheral blood progenitor cells from patients with CML. Leukemia 1999; 13: 1466–1467.
Petzer AL, Eaves CJ, Barnett MJ, Eaves AC . Selective expansion of primitive normal hematopoietic cells in cytokine-supplemented cultures of purified cells from patients with chronic myeloid leukemia. Blood 1997; 90: 64–69.
McGlave PB, De Fabritiis P, Deisseroth A, Goldman J, Barnett M, Reiffers J et al. Autologous transplants for chronic myelogenous leukaemia: results from eight transplant groups. Lancet 1994; 343: 1486–1488.
Deisseroth AB, Zu Z, Claxton D . Genetic marking shows that Ph+ cells present in autologous transplants of chronic myelogenous leukemia (CML) contribute to relapse after autologous bone marrow in CML. Blood 1994; 83: 2068–3076.
Butturini A, Keating A, Goldman JM, Gale RP . Autotransplants in chronic myelogenous leukemia: strategies and results. Lancet 1990; 1: 1255–1257.
Carella AM, Martino OS . Autografting with mobilized hematopoietic progenitor cells in chronic myelogenous leukemia. Proceedings of the International Symposium on Chronic Myelogenous Leukemia, Biarritz, France, July 7–9, 1999 (Abstract, p 63).
Snyder DS, Wu Y, Wang JL, Rossi JJ, Swiderski P, Kaplan BE et al. Ribozyme-mediated inhibition of bcr-abl gene expression in a Philadelphia chromosome-positive cell line. Blood 1993; 82: 600–605.
Martiat P, Taj A, Vaerman JL, Phillipe M, Michaux JL, Goldman JM . Inhibition of P210BCR-ABL in B10 cells using retrovirally transduced antisense sequences against 5 sequences of the BCR/ABL gene. Blood 1991; 78: 266a.
Szczylik C, Skorski T, Nicolaides NC, Manzella L, Malaguarnera L, Venturelli D et al. Selective inhibition of leukemia cell proliferation by BCR-ABL antisense oligodeoxynucleotides. Science 1991; 253: 562–565.
Beissert T, Bianchini A, Hoelzer D, Ottmann OG, Nervi C, Ruthardt M . The size of high molecular weight complexes of ABL formed by different oligomerization interfaces may influence the sensitivity to STI571. Blood 2002; 100: 584a (Abstract 2298).
Guo XY, Cuillerot JM, Wang T, Wu Y, Arlinghaus R, Claxton D et al. Peptide containing the BCR oligomerization domain (AA 1-160) reverses the transformed phenotype of p210bcr-abl positive 32D myeloid leukemia cells. Oncogene 1998; 17: 825–833.
He Y, Wertheim JA, Xu L, Miller JR, Karnell FG, Choi JK et al. The coiled-coil domain and Tyr177 of bcr are required to induce a murine chronic myelogenous leukemia-like disease by bcr/abl. Blood 2002; 8: 2957–2968.
Liu J, Hawk K, Lin F, Arlinghaus R . Phosphoserine peptides from the serine-rich B box of the Bcr first exon inhibit the Abl and Bcr-Abl kinases. Blood 1999; 94(Suppl 1): A453.
Smith KM, Van Etten RA . Oligomerization of Bcr-Abl via the Bcr coiled-coil domain overcomes intramolecular inhibition by the SH3 domain through autophosphorylation. Blood 2002; 100: 204a (Abstract 766).
Gishizky ML, Cortez D, Pendergast AM . Mutant forms of growth factor-binding protein-2 reverse BCR-ABL-induced transformation. Proc Natl Acad Sci USA 1995; 92: 10889–10893.
Dorsey JF, Cunnick JM, Mane SM, Wu J . Regulation of the Erk2-Elk1 signaling pathway and megakaryocytic differentiation of Bcr-Abl+ K562 leukemic cells by Gab2. Blood 2002; 99: 1388–1397.
Liu Y, Jenkins B, Shin JL, Rohrschneider LR . Scaffolding protein Gab2 mediates differentiation signaling downstream of Fms receptor tyrosine kinase. Mol Cell Biol 2001; 21: 3047–3056.
Mohi G, Sattler M, Li S, Van Etten RA, Gu H, Griffin JD et al. Gab2 is required for transformation and leukemogenesis by Bcr/Abl. Blood 2002; 100: 204a (Abstract 764).
Sattler M, Mohi MG, Pride YB, Quinnan LR, Malouf NA, Podar K et al. Critical role for Gab2 in transformation by BCR/ABL. Cancer Cell 2002; 1: 479–492.
Bocchia M, Wentworth PA, Southwood S, Sidney J, McGraw K, Scheinberg DA et al. Rapid communication: specific binding of leukemia oncogene fusion protein peptides to HLA Class I molecules. Blood 1995; 85: 2680–2684.
Chen W, Peace DJ, Rovira DK, You S-G, Cheever MA . T-cell immunity to the joining region of p210BCR-ABL protein. Proc Natl Acad Sci USA 1992; 89: 1468–1472.
Kolb HJ, Beißer K, Holler E, Mittermüller J, Clemm C, Schumm M et al. Adoptive immunotherapy in human and canine chimeras. Haematol Blood Transfusion 1992; 34: 595–600.
Van Denderen J, Hermans A, Meeuwsen T, Troelstra C, Zegers N, Boersma W et al. Antibody recognition of the tumor-specific bcr-abl joining region in chronic myeloid leukemia. J Exp Med 1989; 169: 87–98.
Westermann J, Schlimper C, Richter G, Dorken B, Pezzutto A . T cell reactivity against bcr/abl fusion peptides in healthy donors and CML patients. Blood 2002; 100: 585a (Abstract 2302).
Buchdunger E, Zimmermann J, Mett H, Meyer T, Müller M, Druker BJ et al. Inhibition of the Abl protein-tyrosine kinase in vitro and in vivo by a 2-phenylaminopyrimidine derivative. Cancer Res 1996; 56: 100–104.
Kaur G, Gazit A, Levitzki A, Stowe E, Cooney DA, Sausville EA . Tyrphostin induced growth inhibition: correlation with effect on p210bcr-abl autokinase activity in K562 chronic myelogenous leukemia. Anti-Cancer Drugs 1994; 5: 213–222.
Carroll M, Ohno-Jones S, Tamura S, Buchdunger E, Zimmermann J, Lydon NB et al. CGP57148, a tyrosine kinase inhibitor, inhibits the growth of cells expressing BCR-ABL, TEL-ABL, and TEL-PDGFR fusion proteins. Blood 1997; 90: 4947–4952.
Deininger MWN, Goldman JM, Lydon N, Melo J . The tyrosine kinase inhibitor CGP57148 selectively inhibits the growth of BCR-ABL-positive cells. Blood 1997; 90: 3691–3698.
Druker BJ, Sawyers CL, Kantarjian H, Resta DJ, Reese SF, Ford JM et al. Activity of a specific inhibitor of the Bcr-Abl tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lymphoblastic leukemia with the Philadelphia chromosome. N Engl J Med 2001b; 344: 1038–1056.
Gambacorti-Passerini C, Barni R, Marchesi E, Verga M, Rossi F, Rossi F et al. Sensitivity to the abl inhibitor STI571 in fresh leukaemic cells obtained from chronic myelogenous leukaemia patients in different stages of disease. Br J Haematol 2001; 112: 972–974.
Zimmermann J, Buchdunger E, Mett H, Meyer T, Lydon NB . Potent and selective inhibitors of the able-kinase: phenylaminopyrimidine (PAP) derivatives. Bioorg Med Chem Lett 1997; 7: 187–192.
Gambacorti-Passerini C, Barni R, leCoutre P, Zucchetti M, Cabrita G, Cleris L, Rossi F et al. Alpha 1 acidic glycoprotein (AGP) binds to the abelson inhibitor ST1571, inhibits its biological activity, and causes in vivo resistance of human leukemic cells to BCR/ABL inhibition. Proc Am Assoc Cancer Res 2000; 41(Suppl): 1 (Late-breaking Abstracts: LB-4).
Gambacorti-Passerini C, Barni R, le Coutre P, Zucchetti M, Cabrita G, Cleris L et al. Role of alpha1 acid glycoprotein in the in vivo resistance of human BCR-ABL(+) leukemic cells to the abl inhibitor STI571. J Natl Cancer Inst 2000; 92: 1641–1650.
Shah NP, Nicoll JM, Nagar B, Gorre ME, Paquette RL, Kuriyan J et al. Multiple BCR-ABl kinase domain mutations confer polyclonal resistance to the tyrosine kinase inhibitor imatinib (ST1571) in chronic phase and blast crisis chronic myeloid leukemia. Cancer Cell 2002; 2.
le Coutre P, Tassi E, Varella-Garcia M, Barni R, Mologni L, Cabrita G et al. Induction of resistance to the Abelson inhibitor STI571 in human leukemic cells through gene amplification. Blood 2000; 95: 1758–1766.
Mahon FX, Deininger MW, Schultheis B, Chabrol J, Reiffers J, Goldman JM et al. Selection and characterization of BCR-ABL positive cell lines with differential sensitivity to the tyrosine kinase inhibitor STI571: diverse mechanisms of resistance. Blood 2000; 96: 1070–1079.
Azam M, Latek RR, and Daley GQ, Mechanisms of Autoinhibition and ST1-571/Imatinib resistance revealed by mutagenesis of BCR-ABl. Cell 2003; 112: 831–843.
Marktel S, Bua M, Marin D, Chase A, Udom C, Armstrong L et al. Emergence of additional chromosomal abnormaltieis following treatment with STI571 (imatinib mesylate) for Philadelphia positive chronic myeloid leukemia in chronic phase. Blood 2001; 98: 617a (Abstract 2584).
von Bubnoff N, Schneller F, Peschel C, Duyster J . BCR-ABL gene mutations in relation to clinical resistance of Philadelphia-chromosome-positive leukaemia to STI571: a prospective study. The Lancet 2002; 359: 487–491.
Weisberg E, Griffin JD . Mechanism of resistance to the ABL tyrosine kinase inhibitor STI571 in BCR/ABL-transformed hematopoietic cell lines. Blood 2000; 95: 3498–3505.
Talpaz M, Silver RT, Druker BJ, Goldman JM, Gambacorti-Passerini C, Guilhot F et al. Imatinib induces durable hematologic and cytogenetic responses in patients with accelerated phase chronic myeloid leukemia: results of a phase 2 study. Blood 2002; 99: 1928–1937.
Sawyers CL, Hochhaus A, Feldman E, Goldman JM, Miller CB, Ottman OG et al. Imatinib induces hematologic and cytogenetic responses in patients with chronic myelogenous leukemia in myeloid blast crisis: results of a phase II study. Blood 2002; 99: 3530–3539.
Trabacchi E, Bonifazi F, Bassi S, Testoni N, Alberti D, De Vivo A et al. Imatinib (Glivec®) in patients with Ph+ chronic myeloid leukemia in accelerated/blastic phase (AP/B): results of a Phase II trial of the Italian Cooperative Study Group on CML (ICSG on CML). Blood 2002; 100: 584a (Abstract 2295).
Etienne G, Lagarde V, Reiffers J, Melo JV, Mahon FX . Effects of the tyrosine kinase inhibitor PP1 on STI571-resistant BCR-ABL positive cell lines. Blood 2001; 98: 617a (Abstract 2586).
Hoover RR, Mahon F-X, Melo JV, Daley GQ . Overcoming STI571 resistance with the farnesyl transferase inhibitor SCH66336. Blood 2001; 98: 617a (Abstract 2585).
Kano Y, Akutsu M, Tsunoda S, Mano H, Sato Y, Honma Y et al. In vitro cytotoxic effects of a tyrosine kinase inhibitor STI571 in combination with commonly used antileukemic agents. Blood 2001; 97: 1999–2007.
O'Dwyer M . Multifaceted approach to the treatment of Bcr-Abl-positive leukemias. The Oncologist 2002; 7(Suppl 1): 30–38.
O'Dwyer ME, La Rosee P, Nimmanapalli R, Bhalla KN, Druker BJ . Recent advances in Philadelphia chromosome-positive malignancies: the potential role of arsenic trioxide. Semin Hematol 2002; 39(Suppl 1): 18–21.
Peters DG, Hoover RR, Gerlach MJ, Koh EY, Zhang H, Choe K et al. Activity of the farnesyl protein transferase inhibitor SCH66336 against BCR/ABL-induced murine leukemia and primary cells from patients with chronic myeloid leukemia. Blood 2001; 97: 1401–1412.
Topaly J, Schad M, Zeller WJ, Ho AD, Fruehauf S . Strong synergism of different signal transduction inhibitors in chronic myelogenous leukemia. Blood 2001; 98: 617a (Abstract 2587).
Topaly J, Zeller WJ, Fruehauf S . Synergistic activity of the new ABL-specific tyrosine kinase inhibitor STI571 and chemotherapeutic drugs on BCR-ABL-positive chronic myelogenous leukemia cells. Leukemia 2001; 15: 342–347.
Schindler T, Bornmann W, Pellicena P, Miller T, Clarkson B, Kuriyan J . Structural mechanism for STI-571 inhibition of Abelson tyrosine kinase. Science 2000; 289: 1938–1942.
Danhauser-Riedl S, Warmuth M, Druker BJ, Emmerich B, Hallek M . Activation of Src kinases p53/56lyn and p59hck by p210bcr/abl in myeloid cells. Cancer Res 1996; 56: 3589–3596.
Lionberger JM, Wilson MB, Smithgall TE . Transformation of myeloid leukemia cells to cytokine independence by Bcr-Abl is suppressed by kinase-defective Hck. J Biol Chem 2000; 275: 18581–18585.
Warmuth M, Bergmann M, Priess A, Hauslmann K, Emmerich B, Hallek M . The Src family kinase Hck interacts with Bcr-Abl by a kinase-independent mechanism and phosphorylates the Grb2-binding site of Bcr. J Biol Chem 1997; 272: 33260–33270.
Moasser MM, Srethapakdi M, Sachar KS, Kraker AJ, Rosen N . Inhibition of src kinases by a selective tyrosine kinase inhibitor causes mitotic arrest. Cancer Res 1999; 59: 6145–6152.
Swendeman S, Nagar B, Wisniewski D, Strife A, Lambek C, Liu C et al. Crystal structures of the c-Abl tyrosine kinase domain in complex with STI-571 and a novel Bcr-Abl inhibitor, PD173955 (abstract). Proceedings: AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics: Discovery, Biology, and Clinical Applications, October 29–November 2, 2001 Miami Beach, FL. 2001 (Abstract 568, p 116).
Dorsey JF, Jove R, Kraker AJ, Wu J . The pyrido[2,3-d] pyrimidine derivative PD180970 inhibits p210Bcr-Abl tyrosine kinase and induces apoptosis of K562 leukemic cells. Cancer Res 2000; 60: 3127–3131.
Buchdunger E, Cioffi C, Cozens R, Druker BJ, Lydon NB, Mueller M et al. Preclinical evaluation of CGP57148, a potent protein-tyrosine kinase inhibitor as a therapeutic agent for Ph+ leukemias and solid tumors. Proc Am Assoc Cancer Res 1998; 39: 559 (Abstract 3801).
Druker BJ, Tamura S, Buchdunger E, Ohno S, Segal GM, Fanning S et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat Med 1996; 2: 561–566.
Li S, Hu Y . Src kinase inhibitor CGP76030 synergizes with STI 571 in the treatment of B-cell acute lymphoblastic leukemia induced by the BCR/ABL oncogene in mice. Blood 2002; 100: (Abstract 2283).
Wilson MB, Schreiner SJ, Choi H-J, Kamens J, Smithgall TE . Selective pyrrolo-pyrimidine inhibitors reveal a necessary role for Src family kinases in Bcr-Abl signal transduction and oncogenesis. Oncogene 2002; 21: 8075–8088.
Wilson MB, Schreiner SJ, Choi H-J, Kamens J, Smithgall TE . The pyrrolo-pyrimidine Src kinase inhibitor A-419259 induces growth arrest and apoptosis in CML cells. Blood 2002; 100: 580a (Abstract 2281).
La Rosee P, Corbin AS, Stoffregen EP, Deininger MW, Druker BJ . Activity of the Bcr-Abl kinase inhibitor PD180970 against clinically relevant Bcr-Abl isoforms that cause resistance to imatinib mesylate (Gleevec, STI571). Cancer Res 2002; 62: 7149–7153.
Graham SM, Jorgensen HG, Allan E, Pearson C, Alcorn MJ, Richmond L et al. Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. Blood 2002; 99: 319–325.
Johnson FM, Donato NJ, Talpaz M . Gleevec induces cell cycle arrest, apoptosis, and activation of MAPK in head and neck squamous carcinoma cells. Proc Am Assoc Cancer Res 2002; 43: 846 (Abstract 4192).
Clarkson BD . The survival value of the dormant state in neoplastic and normal cell populations. In: Clarkson B, Baserga R (eds). Control of Proliferation in Animal Cells. New York: Cold Spring Harbor Laboratory, 1974, pp 945–972.
Clarkson B . Consideration of cell kinetic principles to strategy of treating leukemia. In: Cancer Chemotherapy – Fundamental Concepts and Recent Advances. Year Book Medical Publishers, Inc., 1975, pp 19–50.
Macken CA, Perelson AS . Stem Cell Proliferation and Differentiation, a Multitype Branching Process Model, Lecture Notes in Biomathematics, Vol. 76. New York: Springer-Verlag, 1988.
Wichmann HE, Loeffler M . Mathematical Modeling Of Stem Cell Proliferation. Boca Raton, FL: CRC Press, 1985.
Gishizky ML, Witte ON . Initiation of deregulated growth of multipotent progenitor cells by bcr-abl in vitro. Science 1992; 256: 836–839.
Kabarowski JHS, Witte ON . Consequences of BCR-ABL expression within the hematopoietic stem cell in chronic myeloid leukemia. Stem Cells 2000; 18: 399–408.
Lund-Johansen F, Houck D, Hoffman R, Davis K, Olweus J . Primitive human hematopoietic progenitor cells express receptors for granulocyte-macro-phage colony-stimulating factor. Exp Hematol 1999; 27: 762–772.
Quesenberry PJ, Colvin GA, Lambert J-F . Perspective: the chiaroscuro stem cell: a unified stem cell theory. Blood 2002; 100: 4266–4271.
Ramsfjell V, Bryder D, Bjorgvinsdottir H, Kornfalt S, Nilsson L, Borge OJ et al. Distinct requirements for optimal growth and in vitro expansion of human CD34+CD38− bone marrow long-term culture-initiating cells (LTC-IC), extended LTC-IC, and murine in vivo long-term reconstituting stem cells. Blood 1999; 94: 4093–4102.
Zandstra PW, Conneally E, Petzer AL, Piret JM, Eaves CJ . Cytokine manipulation of primitive human hematopoietic cell self-renewal. Proc Natl Acad Sci USA 1997; 94: 4698–4703.
Ziegler BL, Valtieri M, Porada GA, De Maria R, M_ller R, Masella B et al. KDR receptor: a key marker defining hematopoietic stem cells. Science 1999; 285: 1553–1558.
Kawashima I, Zanjani ED, Almaida-Porada G, Flake AW, Zeng H, Ogawa M . CD34+ human marrow cells that express low levels of kit protein are enriched for long-term marrow-engrafting cells. Blood 1996; 87: 4136–4142.
Luens KM, Travis MA, Chen BP, Hill BL, Scollay R, Murray LJ . Thrombopoietin, kit ligand, and flk2/flt3 ligand together induce increased numbers of primitive hematopoietic progenitors from human CD34+Thy-1+Lin− cells with preserved ability to engraft SCID-hu bone. Blood 1998; 91: 1206–1215.
Goodell MA, Rosenzweig M, Kim H, Marks DF, DeMaria MA, Paradis G et al. Dye efflux studies suggest that hematopoietic stem cells expressing low or undetectable levels of CD34 antigen exist in multiple species. Nat Med 1997; 3: 1337–1345.
Ishida A, Zeng H, Ogawa M . Expression of lineage markers by CD34(+) hematopoietic stem cells of adult mice. Exp Hematol 2002; 30: 361–365.
Ogawa M, Tajima F, Ito T, Sato T, Laver JH, Deguchi T . CD34 expression by murine hematopoietic stem cells. Developmental changes and kinetic alterations. Ann NY Acad Sci 2001; 938: 139–145.
Sato T, Laver JH, Ogawa M . Reversible expression of CD34 by murine hematopoietic stem cells. Blood 1999; 94: 2548–2554.
Wisniewski D, Platsoucas C, Strife A, Lambek C, Clarkson B . Enrichment of hematopoietic progenitor cells (CFUc and BFUe) from human peripheral blood. Exp Hematol 1982; 10: 817–829.
Wisniewski D, Strife A, Wachter M, Clarkson B . Regulation of human peripheral blood erythroid burst-forming unit growth by T lymphocytes and T lymphocyte subpopulations defined by OKT4 and OKT8 monoclonal antibodies. Blood 1985; 65: 456–463.
Wisniewski D, Knowles R, Wachter M, Strife A, Clarkson B . Expression of two natural killer cell antigens, H-25 and H-366, by human immature myeloid cells and by erythroid and granulocytic/monocytic colony-forming units. Blood 1987; 69: 419–429.
Wisniewski D, Strife A, Atzpodien J, Clarkson BD . Effects of recombinant human tumor necrosis factor on highly enriched hematopoietic progenitor cell populations from normal human bone marrow and peripheral blood and bone marrow from patients with chronic myeloid leukemia. Cancer Res 1987; 47: 4788–4794.
Wisniewski D, Strife A, Arlin Z, Knowles R, Lambek C, Gulati S et al. Analysis of the individual and combined reactivities of monoclonal antibodies H25, H366, and MY9 with normal progenitor cells and blast cells from patients with acute myeloblastic leukemia. Leukemia 1989; 3: 446–452.
Glimm H, Eaves CJ . Direct evidence for multiple self-renewal divisions of human in vivo repopulating hematopoietic cells in short-term culture. Blood 1999; 94: 2161–2168.
Pierelli L, Scambia G, Fattorossi A, Bonanno G, Battaglia A, Rumi C et al. Functional, phenotypic and molecular characterization of cytokine low-responding circulating CD34+ haemopoietic progenitors. Br J Haematol 1998; 102: 1139–1150.
Clarkson B, Todo A, Ogawa M, Gee T, Fried J . Consideration of the cell cycle in chemotherapy of acute leukemia. In: Ultmann JE, Griem ML, Kirsten WH, Wissler RW (eds). Recent Results of Cancer Research. Current Concepts in the Management of Leukemia and Lymphoma, Vol. 36. New York: Springer-Verlag, 1971, pp 88–118.
Clarkson B . Clinical applications of cell cycle kinetics. In: Sartorelli AC, Johns DG (eds). Handbook of Experimental Pharmacology, New Series Vol. XXXVIII/1. Berlin/Heidelberg/New York: Springer Verlag, 1974, pp 156–193.
Clarkson BD, Dowling MD, Gee TS, Cunningham IB, Burchenal JH . Treatment of acute leukemia in adults. Cancer 1975; 36: 775–795.
Kolitz JE, Kempin SJ, Schluger A, Wong GY, Berman E, Jhanwar S et al. A phase II pilot trial of high-dose hydroxyurea in chronic myelogenous leukemia. Semin Hematol 1992; 19(Suppl 9: Proceedings of the Symposia on Recent Developments in HU Therapy): 27–33.
Gee TS, Yu K-P, Clarkson BD . Treatment of adult acute leukemia with arabinosylcytosine and thioguanine. Cancer 1969; 23: 1019–1032.
Clarkson BD, Arlin ZA, Gee TS, Kempin SJ, Mertelsmann RH, Higgins C et al. Acute lymphocytic leukemia in adults. In: Current Concepts in Medical Oncology II. New York: Memorial Sloan-Kettering Cancer Center, 1983, pp 103–110.
Clarkson B . The chronic leukemias. In: Wyngaarden JB, Smith Jr LH (eds). Cecil Textbook of Medicine, 17th edn. Philadelphia: WB Saunders Co., 1985b, pp 975–986.
Clarkson BD, Gee T, Mertelsmann R, Kempin SJ, Andreeff M, Berman E et al. Current status of treatment of acute leukemia in adults: an overview of the Memorial experience and review of literature. In: Davis S (ed). CRC Critical Reviews in Oncology/Hematology, Vol. 4. Boca Raton, Florida: CRC Press, 1986b, pp 221–248.
Reichert A, Heisterkamp N, Daley GQ, Groffen J . Treatment of Bcr/Abl-positive acute lymphoblastic leukemia in P190 transgenic mice with the farnesyl transferase inhibitor SCH66336. Blood 2001; 97: 1399–1403.
Yu C, Krystal G, Varticovksi L, McKinstry R, Rahmani M et al. Pharmacologic mitogen-activated protein/extracellu-lar signal-regulated kinase kinase/mitogen-activated protein kinase inhibitors interact synergistically with STI571 to induce apoptosis in Bcr/Abl-expressing human leukemia cells. Cancer Res 2002; 62: 188–199.
Sherr CJ, Roberts JM . CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 1999; 13: 1501–1512.
Thiesing JT, Ohno-Jones S, Kolibaba KS, Druker BJ . Efficacy of an Abl tyrosine kinase inhibitor in conjunction with other anti-neoplastic agents against Bcr-Abl positive cells. Blood 1999; 94(Suppl 1): 100a 101a (Abstract 440).
Priebe W, Evrony G, Fokt I, Talpaz M, Donato NJ . WP744, a novel anthracycline with enhanced apoptotic and antiproliferative activity on STI-571 resistant leukemic cells: design, synthesis, and evaluation. Proc Am Assoc Cancer Res 2002; 43: 847 (Abstract 4197).
Guzman ML, Swiderski CF, Howard DS, Grimes BA, Rossi RM, Szilvassy SJ et al. Preferential induction of apoptosis for primary human leukemic stem cells. Proc Natl Acad Sci USA 2002; 99: 16220–16225.
Puccetti E, Guller S, Orleth A . BCR-ABL mediates arsenic trioxide-induced apoptosis independently of its aberrant kinase activity. Cancer Res 2000; 60: 3409–3413.
Porosnicu M, Nimmanapalli R, Nguyen D, Worthington E, Perkins C, Bhalla KN . Co-treatment with As2O3 enhances selective cytotoxic effects of STI-571 against Bcr-Abl-positive acute leukemia cells. Leukemia 2001; 15: 772–778.
Chou T-C, O'Connor OA, Tong AP, Guan Y, Zhang Z-G, Stachel SJ et al. The synthesis, discovery, and development of a highly promising class of microtubule stabilization agents: curative effects of desoxyepothilones B and F against human tumor xenografts in nude mice. Proc Natl Acad Sci USA 2001; 98: 8113–8118.
Clarkson B, Ellis S, Little C, Gee T, Arlin Z, Mertelsmann R et al. Acute lymphoblastic leukemia in adults. Semin Oncol 1985c; 12: 160–179.
Burchenal JH . Long-term survivors in acute leukemia and Burkitt's tumor. Cancer 1968; 21: 595–599.
Burchenal JH, Clarkson B, Haghbin M, Murphy ML . Long-term survivors of acute leukemia. Excerpta Med 1978; 415: 825–828.
Haghbin M, Tan C, Clarkson B, Mike V, Murphy ML . Intensive chemotherapy in children with acute lymphoblastic leukemia (L-2 Protocol). Cancer 1974; 33: 1491–1498.
Clarkson B, Schauer P, Mertelsmann R, Gee T, Arlin Z, Kempin S et al. Results of intensive treatment of acute lymphoblastic leukemia in adults. In: Burchenal JH, Oettgen H (eds). Cancer, Achievements, Challenges and Prospects for the 1980's, Vol. 2. New York: Grune & Stratton, 1981, pp 301–317.
Clarkson B, Gaynor J, Little C, Berman E, Kempin S, Andreeff M et al. Importance of long-term follow-up in evaluating treatment regimens for adults with acute lymphoblastic leukemia. Haematol Blood Transfus 1990; 33: 397–408.
Clarkson B, Gaynor J, Little C, Berman E, Kempin S, Andreeff M et al. Clinical trials in adults with acute lymphoblastic leukemia at Memorial Sloan-Kettering Cancer Center. In: Gale RP, Hoelzer D (eds). Acute Lymphoblastic Leukemia. New York: Alan R. Liss, Inc., 1990, pp 231–252.
Gaynor J, Chapman C, Little C, McKenzie S, Miller W, Andreeff M et al. A cause-specific hazard rate analysis of prognostic factors among 199 adults with acute lymphoblastic leukemia: the Memorial Hospital experience since 1969. J Clin Oncol 1988; 6: 1014–1030.
Gee TS, Dowling MD, Haghbin M, Clarkson B . Acute lymphocytic leukemia in adults and children. Differences in responses on a single therapeutic regimen. Cancer 1976; 37: 1256–1264.
Cunningham I, Reich LM, Kempin SJ, Naval AN, Clarkson BD . Acute promyelocytic leukemia: treatment results during a decade at Memorial Hospital. Blood 1989; 73: 1116–1122.
Drapkin RL, Gee TS, Dowling MD, Arlin S, McKenzie S, Kempin S et al. Prophylactic heparin therapy in acute promyelocytic leukemia. Cancer 1978; 41: 2484–2490.
Fenaux P, Chastang C, Chevret S . A randomized comparison of all-trans-retinoic acid (ATRA) followed by chemotherapy and ATRA plus chemotherapy and the role of maintenance therapy in newly diagnosed acute promyelocytic leukemia. The European APL Group. Blood 1999; 94: 1192–1200.
Stone RM, Mayer RJ . The unique aspects of acute promyelocytic leukemia. J Clin Oncol 1990; 8: 1913–1921.
Huang M-E, Ye Y-C, Chen S-R . Use of all-trans retinoic acid in the treatment of acute promyelocytic leukemia. Blood 1988; 72: 567–572.
Fenaux P, Chomienne C, Degos L . All-trans retinoic acid and chemotherapy in the treatment of acute promyelocytic leukemia. Semin Hematol 2001; 38: 13–25.
Warrell Jr RP, Maslak P, Eardley A . Treatment of acute promyelocytic leukemia with all-trans retinoic acid: an update of the New York experience. Leukemia 1994; 8: 929–933.
Shen Z-X, Chen G-Q, Ni J-H . Use of arsenic trioxide (AS2O3) in the treatment of acute promyelocytic leukemia (APL): II. Clinical efficacy and pharmacokinetics in relapsed patients. Blood 1997; 89: 3354–3360.
Soignet SL, Maslak P, Wang Z-G . Complete remission after treatment of acute promyelocytic leukemia with arsenic trioxide. N Engl J Med 1998; 339: 1341–1348.
Sun HD, Ma L, Hu X-C . Ai-Lin 1 treated 32 cases of acute promyelocytic leukemia. Chin J Integrat Chin West Med 1992; 12: 170–172.
Soignet SL, Frankel SR, Douer D . United States multicenter study of arsenic trioxide in relapsed acute promyelocytic leukemia. J Clin Oncol 2001; 19: 3852–3860.
Slack JL, Waxman S, Tricot G, Tallman MS, Bloomfield CD . Advances in the management of acute promyelocytic leukemia and other hematologic malignancies with arsenic trioxide. The Oncologist 2002; 7(Suppl 1): 1–13.
Miller Jr WH . Molecular targets of arsenic trioxide in malignant cells. The Oncologist 2002; 7(Suppl 1): 14–19.
Perkins C, Kim CN, Fang G . Arsenic induces apoptosis of multidrug-resistant human myeloid leukemia cells that express Bcr-Abl or overexpress MDR, MRP, Bcl-2, or BCl-xL . Blood 2000; 95: 1014–1022.
Hu X-C, Zhang C, Li J-M . Use of arsenic trioxide in the treatment of chronic myelogenous leukemia: clinical efficacy in 34 patients. Program and Abstracts: Advances in Cancer Differentiation Therapy – A Meeting Combining the East and the West, Shanghai, China, October 13–14, 2000 (Abstract 14a).
Zhang R, Zhi Y-J, Tao R-F . Arsenic trioxide in treatment of chronic myelogenous leukaemia in accelerated phase. Chin Clin Oncol 2000; 5: 263–265.
Acknowledgements
We thank Pfizer Global Research and Development (Ann Arbor Laboratories, MI 48105-2430, USA), formerly Parke-Davis Pharmaceutical Research (Division of the Warner-Lambert Co., Ann Arbor, MI 48105, USA) and Dr Neal Rosen for initially providing PD173955, Dr Nicholas Lydon and Novartis Inc. (Basel, Switzerland) for initially providing STI571 (formerly known as CGP57148B), Dr Brian Druker, University of Oregon for giving us the M07e and M07e/p210bcr-abl cell lines, and the Kirin Brewery Company, Limited (Tokyo, Japan) for their generosity in supplying rhG-CSF, rhGM-CSF, rhIL-3, rhKL, and rhSCF. We also thank our collaborators: Dr William G Bornmann and Dr Darren Veach at MSKCC's Preparative Synthesis Laboratory for synthesizing PD173955, PD166326 and other compounds and helpful discussions; Dr John Kuriyan, Drs Bhushan Nagar, Thomas Schindler, Holger Sonderman, and Matthew Young at Rockefeller University, New York, and the University of California, Berkeley, for cocrystallizing the inhibitors with abl kinase and for helpful discussions. We are also grateful to Dr Richard T Silver, Emeritus Director, Clinical Oncology Chemotherapy Research, Division of Hematology and Medical Oncology, New York Presbyterian Cornell Medical Center, and his Nursing and Office Staff for their assistance in obtaining samples of CML blood and marrow, especially Ms Eugenie Balam. We are especially grateful to Su De Meritt for her extensive work in preparing the manuscript. This research was supported by National Cancer Institute (NCI) Grant CA64593 and NCI Cancer Center Support Grant CA08748, The Albert C Bostwick Foundation, The Enid A Haupt Charitable Trust, The Andrew Sage Trust, The Einard and Sue Sundin Fund, The United Leukemia Fund, The Carley H Wagner Trust, The Westvaco Corporation and MeadWestvaco.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Clarkson, B., Strife, A., Wisniewski, D. et al. Chronic myelogenous leukemia as a paradigm of early cancer and possible curative strategies. Leukemia 17, 1211–1262 (2003). https://doi.org/10.1038/sj.leu.2402912
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402912
Keywords
This article is cited by
-
Inhibition of mitochondrial folate metabolism drives differentiation through mTORC1 mediated purine sensing
Nature Communications (2024)
-
Chronic myeloid leukemia stem cells
Leukemia (2019)
-
Impact of Additional Chromosomal Aberrations Present at Diagnosis on Outcome of Adolescent and Young Adult Chronic Myeloid Leukemia Patients: A Single Center Experience
Indian Journal of Hematology and Blood Transfusion (2019)
-
Myeloproliferative neoplasms with concurrent BCR–ABL1 translocation and JAK2 V617F mutation: a multi-institutional study from the bone marrow pathology group
Modern Pathology (2018)
-
The targeting of immunosuppressive mechanisms in hematological malignancies
Leukemia (2014)