Abstract
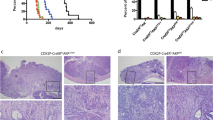
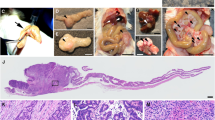
The retinoblastoma gene (Rb) is mutated at significant frequency in various human epithelial tumors, including colorectal cancer, and is strongly associated with metastatic disease. However, sole inactivation of Rb in the mouse has so far failed to yield epithelial cancers. Here, we specifically inactivate Rb and/or p53 in the urogenital epithelium and the intestine. We find that the loss of both tumor suppressors is unable to yield tumors in the transitional epithelium lining the bladder, kidneys and ureters. Instead, these mice develop highly metastatic tumors of neuroendocrine, not epithelial, origin within the urogenital tract to give prostate cancer in the males and vaginal tumors in the females. Additionally, we discovered that the sole inactivation of Rb in the intestine was sufficient to induce formation of metastatic colorectal adenocarcinomas. These tumors closely mirror the human disease in regard to the age of onset, histological appearance, invasiveness and metastatic potential. Like most human colorectal carcinomas, our murine Rb-deficient tumors demonstrate genomic instability and they show activation of β-catenin. Deregulation of the Wnt/β-catenin pathway is specific to the intestinal tumors, as genomic instability but not activation of β-catenin was observed in the neuroendocrine tumors. To date, attempts to generate genetically engineered mouse models of colorectal cancer tumors have yielded mostly cancer of the small intestine, which rarely occurs in humans. Our system provides the opportunity to accurately model and study colorectal cancer in the mouse via a single gene mutation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Burkhart DL, Sage J . Cellular mechanisms of tumour suppression by the retinoblastoma gene. Nat Rev Cancer 2008; 8: 671–682.
Clarke AR, Maandag ER, van Roon M, van der Lugt NM, van der Valk M, Hooper ML et al. Requirement for a functional Rb-1 gene in murine development. Nature 1992; 359: 328–330.
Williams BO, Schmitt EM, Remington L, Bronson RT, Albert DM, Weinberg RA et al. Extensive contribution of Rb-deficient cells to adult chimeric mice with limited histopathological consequences. Embo J 1994; 13: 4251–4259.
Maandag EC, van der Valk M, Vlaar M, Feltkamp C, O'Brien J, van Roon M et al. Developmental rescue of an embryonic-lethal mutation in the retinoblastoma gene in chimeric mice. EMBO J 1994; 13: 4260–4268.
Henley SA, Dick FA . The retinoblastoma family of proteins and their regulatory functions in the mammalian cell division cycle. Cell Div 2012; 7: 10.
Parisi T, Bronson RT, Lees JA . Inhibition of pituitary tumors in Rb mutant chimeras through E2f4 loss reveals a key suppressive role for the pRB/E2F pathway in urothelium and ganglionic carcinogenesis. Oncogene 2009; 28: 500–508.
Lee EY, Cam H, Ziebold U, Rayman JB, Lees JA, Dynlacht BD . E2F4 loss suppresses tumorigenesis in Rb mutant mice. Cancer Cell 2002; 2: 463–472.
Humbert PO, Rogers C, Ganiatsas S, Landsberg RL, Trimarchi JM, Dandapani S et al. E2F4 is essential for normal erythrocyte maturation and neonatal viability. Mol Cell 2000; 6: 281–291.
Wu XR . Urothelial tumorigenesis: a tale of divergent pathways. Nat Rev Cancer 2005; 5: 713–725.
Dinney CP, McConkey DJ, Millikan RE, Wu X, Bar-Eli M, Adam L et al. Focus on bladder cancer. Cancer Cell 2004; 6: 111–116.
Rosenberg JE, Hahn WC . Bladder cancer: modeling and translation. Genes Dev 2009; 23: 655–659.
He F, Mo L, Zheng XY, Hu C, Lepor H, Lee EY et al. Deficiency of pRb family proteins and p53 in invasive orothelial carcinogenesis. Cancer Res 2009; 69: 9413–9421.
Saam JR, Gordon JI . Inducible gene knockouts in the small intestinal and colonic epithelium. J Biol Chem 1999; 274: 38071–38082.
Lawrence MS, Stojanov P, Mermel CH, Robinson JT, Garraway LA, Golub TR et al. Discovery and saturation analysis of cancer genes across 21 tumour types. Nature 2014; 505: 495–501.
Zack TI, Schumacher SE, Carter SL, Cherniack AD, Saksena G, Tabak B et al. Pan-cancer patterns of somatic copy number alteration. Nat Genet 2013; 45: 1134–1140.
Yoo LI, Liu DW, Le Vu S, Bronson RT, Wu H, Yuan J . Pten deficiency activates distinct downstream signaling pathways in a tissue-specific manner. Cancer Res 2006; 66: 1929–1939.
Sharma A, Yeow WS, Ertel A, Coleman I, Clegg N, Thangavel C et al. The retinoblastoma tumor suppressor controls androgen signaling and human prostate cancer progression. J Clin Invest 2010; 120: 4478–4492.
Humphrey PA . Histological variants of prostatic carcinoma and their significance. Histopathology 2012; 60: 59–74.
Zhou Z, Flesken-Nikitin A, Corney DC, Wang W, Goodrich DW, Roy-Burman P et al. Synergy of p53 and Rb deficiency in a conditional mouse model for metastatic prostate cancer. Cancer Res 2006; 66: 7889–7898.
Haigis K, Sage J, Glickman J, Shafer S, Jacks T . The related retinoblastoma (pRb) and p130 proteins cooperate to regulate homeostasis in the intestinal epithelium. J Biol Chem 2006; 281: 638–647.
Kucherlapati MH, Nguyen AA, Bronson RT, Kucherlapati RS . Inactivation of conditional Rb by Villin-Cre leads to aggressive tumors outside the gastrointestinal tract. Cancer Res 2006; 66: 3576–3583.
Nandan MO, Yang VW . Genetic and chemical models of colorectal cancer in mice. Curr Colorectal Cancer Rep 2010; 6: 51–59.
Schwitalla S, Ziegler PK, Horst D, Becker V, Kerle I, Begus-Nahrmann Y et al. Loss of p53 in enterocytes generates an inflammatory microenvironment enabling invasion and lymph node metastasis of carcinogen-induced colorectal tumors. Cancer Cell 2013; 23: 93–106.
Markowitz SD, Bertagnolli MM . Molecular origins of cancer: Molecular basis of colorectal cancer. N Engl J Med 2009; 361: 2449–2460.
Moran A, Ortega P, de Juan C, Fernandez-Marcelo T, Frias C, Sanchez-Pernaute A et al. Differential colorectal carcinogenesis: Molecular basis and clinical relevance. World J Gastrointest Oncol 2010; 2: 151–158.
Coschi CH, Martens AL, Ritchie K, Francis SM, Chakrabarti S, Berube NG et al. Mitotic chromosome condensation mediated by the retinoblastoma protein is tumor-suppressive. Genes Dev 2010; 24: 1351–1363.
Manning AL, Longworth MS, Dyson NJ . Loss of pRB causes centromere dysfunction and chromosomal instability. Genes Dev 2010; 24: 1364–1376.
Manning AL, Yazinski SA, Nicolay B, Bryll A, Zou L, Dyson NJ . Suppression of genome instability in pRB-deficient cells by enhancement of chromosome cohesion. Mol Cell 2014; 53: 993–1004.
Burgess AW, Faux MC, Layton MJ, Ramsay RG . Wnt signaling and colon tumorigenesis—a view from the periphery. Exp Cell Res 2011; 317: 2748–2758.
Horst D, Reu S, Kriegl L, Engel J, Kirchner T, Jung A . The intratumoral distribution of nuclear beta-catenin is a prognostic marker in colon cancer. Cancer 2009; 115: 2063–2070.
Blache P, van de Wetering M, Duluc I, Domon C, Berta P, Freund JN et al. SOX9 is an intestine crypt transcription factor, is regulated by the Wnt pathway, and represses the CDX2 and MUC2 genes. J Cell Biol 2004; 166: 37–47.
Zhang ZT, Pak J, Shapiro E, Sun TT, Wu XR . Urothelium-specific expression of an oncogene in transgenic mice induced the formation of carcinoma in situ and invasive transitional cell carcinoma. Cancer Res 1999; 59: 3512–3517.
Terry S, Beltran H . The many faces of neuroendocrine differentiation in prostate cancer progression. Front Oncol 2014; 4: 60.
Aparicio A, Den RB, Knudsen KE . Time to stratify? The retinoblastoma protein in castrate-resistant prostate cancer. Nat Rev Urol 2011; 8: 562–568.
Kaplan-Lefko PJ, Chen TM, Ittmann MM, Barrios RJ, Ayala GE, Huss WJ et al. Pathobiology of autochthonous prostate cancer in a pre-clinical transgenic mouse model. Prostate 2003; 55: 219–237.
Zeineldin M, Neufeld KL . Understanding phenotypic variation in rodent models with germline Apc mutations. Cancer Res 2013; 73: 2389–2399.
Johnson RL, Fleet JC . Animal models of colorectal cancer. Cancer Metastasis Rev 2012; 32: 39–61.
Hung KE, Maricevich MA, Richard LG, Chen WY, Richardson MP, Kunin A et al. Development of a mouse model for sporadic and metastatic colon tumors and its use in assessing drug treatment. Proc Natl Acad Sci USA 2010; 107: 1565–1570.
Yang HS, Hinds PW . pRb-mediated control of epithelial cell proliferation and Indian hedgehog expression in mouse intestinal development. BMC Dev Biol 2007; 7: 6.
Kucherlapati MH, Yang K, Fan K, Kuraguchi M, Sonkin D, Rosulek A et al. Loss of Rb1 in the gastrointestinal tract of Apc1638N mice promotes tumors of the cecum and proximal colon. Proc Natl Acad Sci USA 2008; 105: 15493–15498.
Gope R, Christensen MA, Thorson A, Lynch HT, Smyrk T, Hodgson C et al. Increased expression of the retinoblastoma gene in human colorectal carcinomas relative to normal colonic mucosa. J Natl Cancer Inst 1990; 82: 310–314.
Morris EJ, Ji JY, Yang F, Di Stefano L, Herr A, Moon NS et al. E2F1 represses beta-catenin transcription and is antagonized by both pRB and CDK8. Nature 2008; 455: 552–556.
Sage J, Miller AL, Perez-Mancera PA, Wysocki JM, Jacks T . Acute mutation of retinoblastoma gene function is sufficient for cell cycle re-entry. Nature 2003; 424: 223–228.
Jonkers J, Meuwissen R, van der Gulden H, Peterse H, van der Valk M, Berns A . Synergistic tumor suppressor activity of BRCA2 and p53 in a conditional mouse model for breast cancer. Nat Genet 2001; 29: 418–425.
Parisi T, Yuan TL, Faust AM, Caron AM, Bronson R, Lees JA . Selective requirements for E2f3 in the development and tumorigenicity of Rb-deficient chimeric tissues. Mol Cell Biol 2007; 27: 2283–2293.
Acknowledgements
We are indebted to Omer Yilmaz for scientific suggestions and professional evaluation of the pathology slides, Kevin Haigis for unpublished information and reagents, A. Amsterdam and S. Frank for critical reading of the manuscript, and the Koch Institute Swanson Biotechnology Center for technical support, particularly the Histology Core. This work was supported by NIH/NCI grants to the Koch Institute (P30-CA14051), and to JAL (P01-CA42063 and RO1-CA121921), a Ludwig Scholar at MIT.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Parisi, T., Bronson, R. & Lees, J. Inactivation of the retinoblastoma gene yields a mouse model of malignant colorectal cancer. Oncogene 34, 5890–5899 (2015). https://doi.org/10.1038/onc.2015.30
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.30
This article is cited by
-
Unmasking the biological function and regulatory mechanism of NOC2L: a novel inhibitor of histone acetyltransferase
Journal of Translational Medicine (2023)
-
SOST silencing promotes proliferation and invasion and reduces apoptosis of retinoblastoma cells by activating Wnt/β-catenin signaling pathway
Gene Therapy (2017)
-
Quantitative secretomic analysis of pancreatic cancer cells in serum-containing conditioned medium
Scientific Reports (2016)
-
Rapid tumor induction in zebrafish by TALEN-mediated somatic inactivation of the retinoblastoma1 tumor suppressor rb1
Scientific Reports (2015)