Key Points
-
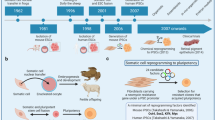
In the highly competitive industry of drug discovery, pharmaceutical and biotechnology companies must rely on improved high throughput technologies to advance programs from target confidence to pre-clinical evaluation.
-
Embryonic stem cells have recently become highly visible with the identification of pluripotent cell lines derived from human blastocysts or primordial germ cells. Much of the focus of these cells has centered on future opportunities in regenerative medicine, with less emphasis on the near term opportunities in cell-based systems across the drug discovery timeline.
-
Mouse embryonic stem cells have been used extensively over the last 10-15 years by drug discoverers in the development of genetically modified mice for target validation, target selectivity, model development and toxicity evaluation.
-
Advances in the isolation and purification of in vitro differentiated murine ES cells combined with highly sophisticated methods of genetic engineering have afforded opportunities in drug discovery directly in murine ES cells. The strength of an ES cell in vitro approach is time to delivery of results, cost an
Abstract
The completed sequencing of the human genome has identified numerous potential drug targets, which are expected to deliver the next generation of new medicines. However, for drug companies to realize this opportunity, they must rely on improved prognostic applications of high-throughput technologies, from target identification to preclinical compound evaluation. Reducing the timelines and attrition rate of new therapeutics for clinical evaluation requires cell-based methods for testing the efficacy and safety of new compounds. Drug discoverers are beginning to use stem cells as a new resource for increasing confidence in the mechanism of action of new targets and the safety of modulating their activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Harris, S. Transgenic knockouts as part of high-throughput, evidence based target selection and validation strategies. Drug Discov. Today 6, 628–636 (2001)
Abuin, A., Holt, K. H., Platt, K. A., Sands, A. T. & Zambrowicz, B. P. Full-speed mammalian genetics; in vivo target validation in the drug discovery process. Trends Biotechnol. 20, 261–268 (2002). References 1 and 2 are excellent reviews on the application of genetically modified mice in drug discovery.
Crawley, J. N. Behavioral phenotyping of transgenic and knockout mice: experimental design and evaluation of general health, sensory functions, motor abilities and specific behavioral tests. Brain Res. 835, 18–26 (1999).
van der Staay, F. J. & Steckler, T. Behavioral phenotyping of mouse mutants. Behav. Brain Res. 125, 3–12 (2001).
Doevendans, P. A., Daemen, M. J., de Muinck, E. D. & Smits, J. F. Cardiovascular phenotyping in mice. Cardiovasc. Res. 39, 34–49 (1998).
Weissleder, R. Scaling down imaging: molecular mapping of cancer in mice. Nature Rev. Cancer 2, 11–18 (2002).
Massoud, T. F. & Gambhir, S. S. Molecular imaging in living subjects: seeing fundamental biological processes in a new light. Genes Dev. 17, 545–580 (2003).
Zambrowicz, B. P. & Sands, A. T. Knockouts of the 100 best-selling drugs — will they model the next 100? Nature Rev. Drug Discov. 2, 38–51 (2003). An important review aligning genetically modified mice with therapeutic target evaluation. The authors describe the phenotypes associated with KO mice for targets of many of the most successful medicines and conclude that this is a cost-effective strategy for defining novel target mechanisms for the next generation of medicines.
Zhang, S. H., Reddick, R. L., Piedrahita, J. A. & Maeda, N. Spontaneous hypercholesterolemia and arterial lesions in mice lacking apolipoprotein E. Science 258, 468–471 (1992).
Plump, A. S. et al. Severe hypercholesterolemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell 71, 343–353 (1992).
Beierschmitt, W. P. et al. Induction of hepatic microsomal drug-metabolizing enzymes by inhibitors of 5-lipoxygenase (5-LO) studies in rats and 5-LO knockout mice. Toxicol. Sci. 63, 15–21 (2001).
Davis, J. A. et al. An Alzheimer's disease-linked PS1 variant rescues the developmental abnormalities of PS1-deficient embryos. Neuron 20, 603–609 (1998).
Grilli, M. et al. Presenilin-1 regulates the neuronal threshold to excitotoxicity both physiologically and pathologically. Proc. Natl Acad. Sci. USA 97, 12822–12827 (2000).
Wang, Y., Schnegelsberg, P. N., Dausman, J. & Jaenisch, R. Functional redundancy of the muscle-specific transcription factor Myf5 and myogenin. Nature 379, 823–825 (1996).
Prosser, H. M. et al. Targeted replacement of rodent CCR2 with the human orthologue CCR2B: a mouse model for in vivo analysis of human target-selective small molecule MCP-1 receptor antagonists. Drug Discov. Res. 55, 197–209 (2002).
Shiao, L. L., Cascieri, M. A., Trumbauer, M., Chen, H. & Sullivan, K. A. Generation of mice expressing the human glucagons receptor with a direct replacement vector. Transgenic Res. 8, 295–302 (1999).
Zambrowicz, B. P. et al. Wnk1 kinase deficiency lowers blood pressure in mice: A gene-trap screen to identify potential targets for therapeutic intervention. Proc. Natl Acad. Sci. USA 10 November 2003 (doi:10.1073pnas.2336103100).
Vivian, J. L., Chen, Y., Yee, D., Schneider, E. & Magnuson, T. An allelic series of mutations in Smad2 and Smad4 identified in genotype-based screen of N-ethyl-N-nitrosourea-mutagenized mouse embryonic stem cells. Proc. Natl Acad. Sci. USA 99, 15542–15547 (2002).
Doetschman, T. C., Eistetter, H., Katz, M., Schmidt, W. & Kemler, R. The in vitro development of blastocyst-derived embryonic stem cell lines: formation of a visceral yolk sac, blood islands and myocardium. J. Embryol. Exp. Morphol. 87, 27–45 (1985). The first report of in vitro differentiation in murine embryonic stem cells. The authors describe the spontaneous differentiation potential of the ES cells and offer a forward-looking perspective of this observation.
Williams, R. L. et al. Myeloid leukemia inhibitory factor maintains the developmental potential of embryonic stem cells. Nature 336, 684–687 (1988).
Wobus, A. M., Grosse, R. & Schoneich, J. Specific effects of nerve growth factor on the differentiation pattern of mouse embryonic stem cells in vitro. Biomed. Biocem. Acta. 47, 965–973 (1988).
Robbins, J., Gulick, J., Sanchez, A., Howles, P. & Doetschman, T. Mouse embryonic stem cells express the cardiac myosin heavy chain genes during development in vitro. J. Biol. Chem. 265, 11905–11909 (1990).
Maltsev, V. A., Rohwedel, J., Herscheler, J. & Wobus A. M. Embryonic stem cells differentiate in vitro into cardiomyocytes representing sinusoidal, atrial and ventricular cell types. Mech. Dev. 44, 41–50 (1993).
Schmitt, R. M., Bruyns, E. & Snodgrass, H. R. Hematopoietic development of embryonic stem cells in vitro: cytokine and receptor gene expression. Genes Dev. 5, 728–740 (1991).
Wiles, M. V. & Keller, G. Multiple hematopoietic lineages develop from embryonic stem (ES) cells in culture. Development 111, 259–267 (1991).
Chen, U. Differentiation of mouse embryonic stem cells into lympho-hematopoietic lineages in vitro. Dev. Immunol. 2, 29–50 (1992).
Keller, G., Kennedy, M., Papayannopoulou, T. & Wiles, M. V. Hematopoeitic commitment during embryonic stem cell differentiation in culture. Mol. Cell. Biol. 13, 473–486 (1993).
Strubing, C. et al. Differentiation of pluripotent embryonic stem cells into neuronal lineages in vitro gives rise to mature inhibitory and excitatory neurons. Mech. Dev. 53, 275–287 (1995).
Fraichard, A. et al. In vitro differentiation of embryonic stem cells into glial and functional neurons. J. Cell Sci. 108, 3181–3188 (1995).
Desbaillets, I., Ziegler, U., Groscurth, P. & Gasserman, M. Embryoid body: an in vitro model of mouse embryogenesis. Exp. Physiol. 85, 645–651 (2000).
Dani, C. et al. Differentiation of embryonic stem cells into adipocytes in vitro. J. Cell Sci. 110, 1279–1285 (1997).
Kramer, J., Hegeret, C., Guan, K., Wobus, A. M., Muller, P. K. & Rohedel, J. Embryonic stem cell-derived chondrogenic differentiation in vitro: activation by BMP-2 and BMP-4. Mech. Dev. 92, 193–205 (2000).
Hegert, C. et al. Differentiation plasticity of chondrocytes derived from mouse embryonic stem cells. J. Cell Sci. 115, 4617–4628 (2002).
Boheler, K. R., Czyz, J., Tweedie, D., Yang, H. T., Anisimov, S. V. & Wobus, A. M. Differentiation of pluripotent embryonic stem cells into cardiomyocytes. Circ. Res. 91, 189–201. (2002).
Angelov, D. N. et al. Temporospatial relationships between macroglia and microglia during in vitro differentiation of murine stem cells. Dev. Neurosci. 20, 42–51 (1998).
Brustle, O. et al. Embryonic stem cell-derived glial precursors: a source of myelinating transplants. Science 285, 754–756 (1999).
Lumelsky, N. et al. Differentiation of embryonic stem cells to insulin-secreting structures similar to pancreatic islets. Science 292, 1389–1394 (2001).
Wichterle, H., Lieberam, I., Porter, J. A. & Jessell, T. M. Directed differentiation of embryonic stem cells into motor neurons. Cell 110, 385–397 (2002).
Yamane, T. et al. Development of osteoclasts from embryonic stem cells through a pathway that is c-fms but not c-kit dependent. Blood 90, 3516–3523 (1997).
Berthier, R. et al. The MS-5 murine stromal cell line and hematopoietic growth factor synergize to support the megakaryocyte differentiation of embryonic stem cells. Exp. Hematol. 25, 481–490 (1997).
Stevens, L. C. The development of transplantable teratocarcinomas from intratesticular grafts of pre- and postimplantation embryos. Dev. Biol. 21, 364–382 (1970).
Damjanov, I., Damjanov, J. & Solter, D. in Teratocarcinomas and Embryonic Stem Cells: A Practical Approach (ed Robertson, E. J.) 1–18 (IRL Oxford, Washington, 1987).
Klug, M. G., Soonpaa, M. H., Koh, G. Y. & Fields, L. J. Genetically selected cardiomyocytes from differentiating embryonic stem cells form stable intracardiac grafts. J. Clin. Invest. 98, 216–224 (1996). References 41–43 clearly establish lineage selection as an important method in the derivation and purification of some in vitro differentiated ES cells.
Mountford, P. et al. Dicistronic targeting constructs: reporters and modifiers of mammalian gene expression. Proc. Natl Acad. Sci. USA 91, 4303–4307 (1994).
Li, M., Pevny, L., Lovell-Badge, R. & Smith, A. Generation of purified neural precursors from embryonic stem cells by lineage selection. Curr. Biol. 8, 971–974 (1998).
Kolossov, E. et al. Functional characteristics of ES cell-derived cardiac precursor cells identified by tissue-specific expression of green fluorescent protein. J. Cell Biol. 143, 2045–2056 (1998).
Allen, M. et al. Deficiency of the stress kinase p38α results in embryonic lethality: characterization of the kinase dependence of stress responses of enzyme-deficient embryonic stem cells. J. Exp. Med. 191, 859–870 (2000). This paper describes how murine ES cell in vitro differentiation was used to overcome the embryonic lethality of the target gene KO mouse and detail the role of the p38 kinase in inflammatory signalling. The results were important to drug discovery investigators and the potential impact of in vitro -based stem-cell technology was highlighted.
Kim, J. -H. et al. Dopaminergic neurons derived embryonic stem cells function in an animal model of Parkinson's disease. Nature 418, 50–56 (2002).
Lee, M. A. et al. Overexpression of midbrain-specific transcription factor Nurr1 modifies susceptibility of mouse neural stem cells to neurotoxins. Neurosci. Lett. 333, 74–78 (2002).
Blyszczuk, P. et al. Expression of Pax4 in embryonic stem cells promotes differentiation of nestin positive progenitor and insulin-producing cells. Proc. Natl Acad. Sci. USA 100, 998–1003 (2003).
Bagutti, C., Wobus, A. M., Fassler, R. & Watt, F. M. Differentiation of embryonic stem cells into keratinocytes: comparisons of wild-type and β1-integrin deficient cells. Dev. Biol, 179, 184–196 (1996).
Fairchild, P. J. et al. Directed differentiation of dendritic cells from mouse embryonic stem cells. Curr. Biol. 10, 1515–1518 (2000).
Nakano, T., Kodama, H. & Honjo, T. In vitro development of primitive and definitive erythryocytes from different precursors. Science 272, 722–724 (1996).
Risau, W. et al. Vasculogenesis and angiogenesis in embryonic-stem-cell-derived embryoid bodies. Development 102, 471–478 (1988).
Rohwedel, J. et al. Muscle cell differentiation of embryonic stem cells reflects myogenesis in vivo: developmentally regulated expression of myogenic determination genes and functional expression of ionic currents. Dev. Biol 164, 87–101 (1994).
Buttery, L. D. et al. Differentiation of osteoblasts and in vitro bone formation from murine embryonic stem cells. J. Cell Sci. 110, 1279–1285 (1997).
Jones, E. A., Tosh, D., Wilson, D. I., Lindsay, S. & Forrester, L. M. Hepatic differentiation of murine embryonic stem cells. Exp. Cell Res. 272, 15–22 (2002).
Ali, N. N. et al. Derivation of type II alveolar epithelial cells from murine embryonic stem cells. Tissue Eng. 8, 541–550 (2002).
Hubner, K. et al. Derivation of oocytes from mouse embryonic stem cells. Science 300, 1251–1256 (2003).
Toyooka, Y., Tsunekawa, N., Akasu, R. & Noce, T. Embryonic stem cells can form germ cells in vitro. Proc. Natl Acad. Sci. USA 100, 11457–11462 (2003).
Nichols, J. et al. Formation of pluripotent stem cells in the mammalian embryo depends on POU transcription factor, Oct 4. Cell 95, 379–391 (1998).
Chambers, I. et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell 113, 643–655 (2003).
Mitsui, K. et al. The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells. Cell 113, 631–642 (2003).
Ramalho-Santos, M., Yoon, S., Matsuzaki, Y., Mulligan, R. C. & Melton, D. A. “Stemness”: transcriptional profiling of embryonic and adult stem cells. Science 298, 597–600 (2002).
Ivanova, N. B. et al. A stem cell molecular signature. Science 298, 601–604 (2002)
D'Amour, K. A. & Gage, F. H. Genetic and functional differences between multipotent neural and pluripotent embryonic stem cells. Proc. Natl Acad. Sci. USA 100, 11866–11872 (2003).
Potter, S. S., Valerius, M. T. & Brunskill, E. W. Using progenitor cells and gene chips to define genetic pathways. Methods Mol. Biol. 185, 269–284 (2002).
Hemmer, R., Wei, W., Dutriaux, A. & Sedivy, J. Somatic cell knockouts of tumor suppressor genes. Methods Mol. Biol. 223, 187–206 (2003).
Hatada, S., Nikkuni, K., Bentley, S. A., Kirby, S. & Smithies, O. Gene correction in hematopoietic progenitor cells by homologous recombination. Proc. Natl Acad. Sci. USA 97, 13807–13811 (2000).
Abuin, A & Bradley, A. Recycling selectable markers in mouse embryonic stem cells. Mol. Cell. Biol. 16, 1851–1856 (1996).
Mortenson, R. M., Conner, D. A., Chao, S., Geisterfer-Lowrance, A. A. & Seidman, J. G. Production of homozygous mutant ES cells with single targeting construct. Mol. Cell. Biol. 12, 2391–2395 (1992).
Langa, F. et al. Teratocarcinomas induced by embryonic stem (ES) cells lacking vimentin: an approach to study the role of vimentin in tumorigenesis. J. Cell Sci. 113, 3463–3472 (2000).
Ishikawa, H., Ryseck, R. P. & Bravo, R. Characterization of ES cells deficient for the p105 precursor (NF-κB1): role of p50 NLS. Oncogene 13, 255–263 (1996).
Fassler, R. et al. Differentiation and integrity of cardiac muscle cells are impaired in the absence of β1 integrin. J. Cell Sci. 109, 2989–2999 (1996).
Gowen, L. C., Johnson, B. L., Latour, A. M., Sulik, K. K. & Koller, B. H. BRCA1 deficiency results in early embryonic lethality characterized by neuroepithelial abnormalities. Nature Genet. 12, 191–194 (1996).
Snouwaert, J. N. et al. BRCA1 deficient embryonic stem cells display a decreased homologous recombination frequency and an increased non-homologous recombination that is corrected by expression of BRCA1 transgene. Oncogene 18, 7900–7907 (1999).
Minamino, T. et al. MEKK1 suppresses oxidative stress-induced apoptosis of embryonic stem cell-derived cardiac myocytes. Proc. Natl Acad. Sci. USA 96, 15127–15132 (1999).
Takeshima, H. et al. Embryonic lethality and abnormal cardiac myocytes in mice lacking ryanodine receptor type 2. EMBO J. 17, 3309–3316 (1998).
Yang, H. T. et al. The ryanodine receptor modulates the spontaneous beating rate of cardiomyocytes during development. Proc. Natl Acad. Sci. USA 99, 9225–9230 (2002).
Zhang, X., Morham, S. G., Langenbach, R., Baggs, R. B. & Young, D. A. Lack of cyclooxygenase-2 inhibits growth in teratocarcinomas in mice. Exp Cell Res. 254, 232–240 (2000).
Moore, R., Radice, G. L., Dominis, M. & Kemler, R. The generation and in vivo differentiation of murine embryonal stem cells genetically null for either N-cadherin or P-cadherin. Int. J. Dev. Biol. 43, 831–834 (1999).
Hertzberg, R. P. & Pope, A. J. High-throughput screening: new technology for the 21st century. Curr. Opinion Chem. Biol. 4, 445–451 (2000).
Laschinski, G., Vogel, R. & Spielman, H. Cytotoxicity test using blastocyst-derived euploid embryonal stem cells: a new approach to in vitro teratogenesis screening. Reprod. Toxicol. 5, 57–64 (1991).
Spielman, H., Pohl, I., Doring, B., Liebsch, M. & Moldenhauer, F. The embryonic stem cell test, an in vitro embryotoxicity test using two permanent mouse cell lines 3T3 fibroblasts and embryonic stem cells. Toxicol. In Vitro 10, 119–127 (1997).
Balls, M. & Hellstein, E. Statement on the scientific validity of the embryonic stem cell test (EST) — an in vitro test for embryotoxicity. Altern. Lab. Anim. 30, 265–268 (2002).
Rudnicki, M. A. & McBurney, M. W. in Teratocarcinomas and Embryonic Stem Cells: A Practical Approach (ed Robertson, E. J.) 19–49 (IRL Oxford, Washington DC, 1987).
Brown, N. Selection of test chemicals for the ECVAM international validation study on in vitro embryotoxicity test. Altern. Lab. Anim. 30, 177–198, (2002).
Spielman, H. et al. Preliminary results of the ECVAM validation study on three in vitro embryotoxicity tests. Altern. Lab. Anim. 29, 301–330 (2001). References 83–85, and 87 and 88, describe the validated use of murine ES cells as an alternative to animal testing in teratology and embryotoxicity testing. This embryonic stem cell test demonstrated excellent alignment in predicting embryotoxicity with known teratogens in vivo.
Bremer, S. et al. Establishment of an in vitro reporter assay for the development of cardiac toxicity. Toxicol. In Vitro 15, 215–223 (2001).
Paparella, M., Kolossov, E., Fleischmann, B. K., Hescheler, J. & Bremer, S. The use of quantitative image analysis in the assessment of in vitro embryotoxicity endpoints based on a novel embryonic stem cell clone with endoderm-related GFP expression. Toxicol. In Vitro 16, 589–597 (2002).
Bremer, S., Pellizzer, C., Coecke, S., Paparella, M. & Catalani, P. Detection of the embryotoxic potential of cyclophosphamide by using a combined system of metabolic competent cells and embryonic stem cells. Altern. Lab. Anim. 30, 77–85 (2002).
Suda, Y., Suzuki, M., Ikawa, Y. & Aizawa, S. Mouse embryonic stem cells exhibit indefinite proliferation potential. J. Cell Physiol. 133, 197–201 (1987).
Hancock, C. R., Wetherington, J. P., Lambert, N. A. & Condie, B. G. Neuronal differentiation of cryopreserved neural progenitor cells derived from mouse embryonic stem cells. Biochem. Biophys. Res. Commun. 271, 418–421 (2000).
Thomson, J. A. et al. Embryonic stem cells lines derived from human blastocysts. Science 282, 1145–1147 (1998). The first report of a human embryonic stem cell line established from a blastocyst-stage embryo.
Reubinoff, B. E., Peras, M. F., Fong, C. Y., Trounson, A. & Bongso, A. Embryonic stem cell lines from human blastocysts: somatic differntiation in vitro. Nature Biotechnol. 18, 399–404 (2000).
Shamblott, M. J. et al. Derivation of pluripotent stem cells from cultured primordial germ cells. Proc. Natl Acad. Sci. USA 95, 13726–13731 (1998). The first report of a human embryonic stem cell line established from primary germ cells of a developing embryo.
Lebkowski, J. S. et al. Human embryonic stem cells: culture, differentiation and genetic modification for regenerative medicine applications. Cancer J. 7 (Suppl. 2), S83–S93 (2001).
Henderson, J. K. et al. Preimplantation human embryos and embryonic stem cells show comparable expression of stage specific antigens. Stem Cells 20, 329–337 (2002).
Amit, M. et al. Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods in culture. Dev. Biol. 227, 271–278 (2000).
Itskovitz-Elder, J. et al. Differentiation of human embryonic stem cells into embryoid bodies comprising the three embryonic germ layers. Mol Med. 6, 88–95 (2000).
Amit, M. & Itskovitz-Elder, J. Derivation and spontaneous differentiation of human embryonic stem cells. J. Anat. 200, 225–232 (2002).
Schuldiner, M., Yanuka, O., Itskovitz-Elder, J., Melton, D. A. & Benvenisty, N. Effects of eight growth factors on the differentiation of cells derived from human embryonic stem cells. Proc. Natl Acad. Sci. USA 97, 11307–11312 (2000).
Kehat, I. et al. Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J. Clin. Invest. 108, 407–414 (2001).
Schuldiner, M. et al. Induced neuronal differentiation of human embryonic stem cells. Brain Res. 913, 201–205 (2001).
Zhang, S. -C., Werning, M., Duncan, I. D., Brustle, O. & Thomson, J. A. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nature Biotechnol. 19, 1129–1133 (2001).
Rambhatla, L., Chiu, C. P., Kundu, P., Peng, Y. & Carpenter, M. K. Generation of hepatocyte-like cells from human embryonic stem cells. Cell Transplant. 12, 1–11 (2003).
Levenberg, S., Golub, J. S., Amit, M., Itskovitz-Elder, J. & Langer, R. Endothelial cells derived from human embryonic stem cells. Proc. Natl Acad. Sci. USA 99, 4391–4396 (2002).
Eiges, R. et al. Establishment of human embryonic stem cell-transfected clones carrying a marker for undifferentiated cells. Curr. Biol. 11, 514–518 (2001).
Ma, Y., Ramezani, A., Lewis, R., Hawley, R. G. & Thomson, J. A. High-level sustained transgene expression in human embryonic stem cells using lentiviral vectors. Stem Cells 21, 111–117 (2003).
Zwaka, T. P. & Thomson, J. A. Homologous recombination in human embryonic stem cells. Nature Biotechnol. 21, 319–321 (2003). The first demonstration that human ES cells maintain the genetic machinery capable of directing homologous recombination at frequencies previously observed only in murine ES cells. The investigators established novel protocols for achieving high transfection efficiencies based on cellular differences between murine and human ES cells.
Walke, D. W. et al. In vivo drug target discovery: identifying the best targets from the genome. Curr. Opin. Biotechnol. 12, 626–631 (2001).
Hopkins, A. L. & Groom, C. R. The druggable genome. Nature Rev. Drug Discov. 1, 727–730 (2002).
Holden, C. & Vogel, G. “Show us the cells” U. S. researchers say. Science 297, 923–925 (2002).
Pittenger, M. F. et al. Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 (1999).
Jiang, Y. et al. Pluripotency of mesenchymal stem cells derived from adult bone marrow. Nature 418, 41–49 (2002).
Broxmeyer, H. E. et al. High efficiency recovery of functional hematopoietic progenitor and stem cells from human cord blood cryopreserved for 15 years. Proc. Natl Acad. Sci. USA 100, 645–650 (2003).
Zuk, P. A. et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol. Cell 13, 4279–4295 (2002).
Roach, M. et al. Attenuation of erythropoiesis by REDK (DYRK3) in an embryonic stem cell-based in vitro differentiation model. First Annu. Mtg Int. Soc. Stem Cell Res. A86 (2003).
Acknowledgements
The author acknowledges the contributions of G. Cezar, K. Haskell, M. Roach with figures, J. Hambor and K. Neote in generating Box 1, P. Vickers for critically reading of the manuscript and T. Kelleher for encouragement. Finally without the love and support of my family this manuscript would not have been possible.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Related links
Related links
DATABASES
LocusLink
FURTHER INFORMATION
Online Mendelian Inheritance in Man
Glossary
- TARGET
-
Gene product that therapeutic approaches are directed against in the search for new medicines.
- EMBRYONIC STEM CELLS
-
(ES). Undifferentiated cells typically derived from the inner cell mass of a blastocyst-stage embryo. In culture, these cells can self-renew in the undifferentiated state or reveal their pluripotentcy on differentiation into cell types of the three embryonic germ lineages. Mouse ES cells readily differentiate into all somatic and germ lineages when incorporated into chimeric offspring through blastocyst microinjection or morula aggregation.
- KNOCKOUT MICE
-
Mice derived from gene-targeting experiments in ES cells with mutations in selected genes that result in the complete ablation of gene expression. These induced mutations are passed through the germline, allowing the derivation of novel lines of gene KO mice for functional evaluation in vivo.
- HOMOLOGOUS RECOMBINATION
-
Genetic recombination between identical (or nearly identical) double-stranded DNA sequences. This process occurs at a relatively high frequency in ES cells, allowing the engineering of gene-targeting vectors to direct pre-planned mutagenesis in ES cells.
- DIFFERENTIATION
-
Process in which a cell progresses in a linear manner to a specialized state. Stem cells can develop into any specialized cell type – for example, neuronal, muscle, hepatic and haematopoietic cells.
- EMBRYOID BODIES
-
Spherical cell clusters observed after spontaneous or induced differentiation of ES cells in culture: Embryoid bodies show differentiation that recapitulates the early stages of mammalian embryonic development, including cell types derived from endoderm, mesoderm and ectodermal lineages.
- PLURIPOTENT
-
The ability to differentiate into all cell types derived from the three embryonic germ lineages: ectoderm, mesoderm and endoderm.
- ANEUPLOID
-
Cells with abnormal number of chromosome sets.
- TERATOMA
-
A rare tumour type that typically arises in the gonads and demonstrates mixed cellular populations of all three embryonic germ layers. Investigators can assess the differentiation capacity of stem cells by injection of pluripotent cells into laboratory animals and inducing the formation of teratomas in situ.
Rights and permissions
About this article
Cite this article
McNeish, J. Embryonic stem cells in drug discovery. Nat Rev Drug Discov 3, 70–80 (2004). https://doi.org/10.1038/nrd1281
Issue Date:
DOI: https://doi.org/10.1038/nrd1281
This article is cited by
-
TRPC3 is required for the survival, pluripotency and neural differentiation of mouse embryonic stem cells (mESCs)
Science China Life Sciences (2018)
-
Culture medium refinement by dialysis for the expansion of human induced pluripotent stem cells in suspension culture
Bioprocess and Biosystems Engineering (2017)
-
Wnt/β-Catenin and MEK-ERK Signaling are Required for Fibroblast-Derived Extracellular Matrix-Mediated Endoderm Differentiation of Embryonic Stem Cells
Stem Cell Reviews and Reports (2015)
-
Developing Defined and Scalable 3D Culture Systems for Culturing Human Pluripotent Stem Cells at High Densities
Cellular and Molecular Bioengineering (2014)
-
A Review of Human Pluripotent Stem Cell-Derived Cardiomyocytes for High-Throughput Drug Discovery, Cardiotoxicity Screening, and Publication Standards
Journal of Cardiovascular Translational Research (2013)