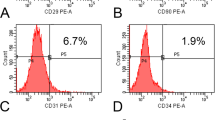
Abstract
In adults and children over two years of age, large cranial defects do not reossify successfully, posing a substantial biomedical burden. The osteogenic potential of bone marrow stromal (BMS) cells has been documented. This study investigates the in vivo osteogenic capability of adipose-derived adult stromal (ADAS) cells, BMS cells, calvarial-derived osteoblasts and dura mater cells to heal critical-size mouse calvarial defects. Implanted, apatite-coated, PLGA scaffolds seeded with ADAS or BMS cells produced significant intramembranous bone formation by 2 weeks and areas of complete bony bridging by 12 weeks as shown by X-ray analysis, histology and live micromolecular imaging. The contribution of implanted cells to new bone formation was 84–99% by chromosomal detection. These data show that ADAS cells heal critical-size skeletal defects without genetic manipulation or the addition of exogenous growth factors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Krebsbach, P.H. et al. Bone formation in vivo: comparison of osteogenesis by transplanted mouse and human marrow stromal fibroblasts. Transplantation 63, 1059–1069 (1997).
Krebsbach, P.H., Mankani, M.H., Satomura, K., Kuznetsov, S.A. & Robey, P.G. Repair of craniotomy defects using bone marrow stromal cells. Transplantation 66, 1272–1278 (1998).
Mendes, S.C. et al. Bone tissue-engineered implants using human bone marrow stromal cells: effect of culture conditions and donor age. Tissue Eng. 8, 911–920 (2002).
Peng, H. et al. Synergistic enhancement of bone formation and healing by stem cell-expressed VEGF and bone morphogenetic protein-4. J. Clin. Invest. 110, 751–759 (2002).
Vacanti, J.P., Langer, R., Upton, J. & Marler, J.J. Transplantation of cells in matrices for tissue regeneration. Adv. Drug Deliv. Rev. 33, 165–182 (1998).
Wright, V. et al. BMP4-expressing muscle-derived stem cells differentiate into osteogenic lineage and improve bone healing in immunocompetent mice. Mol. Ther. 6, 169–178 (2002).
Durham, S.R., McComb, J.G. & Levy, M.L. Correction of large (>25 cm(2)) cranial defects with “reinforced” hydroxyapatite cement: technique and complications. Neurosurgery 52, 842–845; discussion 845 (2003).
Harris, S.E. et al. Effects of transforming growth factor beta on bone nodule formation and expression of bone morphogenetic protein 2, osteocalcin, osteopontin, alkaline phosphatase, and type I collagen mRNA in long-term cultures of fetal rat calvarial osteoblasts. J. Bone Miner. Res. 9, 855–863 (1994).
Dong, J. et al. In vivo evaluation of a novel porous hydroxyapatite to sustain osteogenesis of transplanted bone marrow-derived osteoblastic cells. J. Biomed. Mater. Res. 57, 208–216 (2001).
Lee, J.Y. et al. Effect of bone morphogenetic protein-2-expressing muscle-derived cells on healing of critical-sized bone defects in mice. J. Bone Joint Surg. Am. 83-A, 1032–1039 (2001).
Shea, L.D., Wang, D., Franceschi, R.T. & Mooney, D.J. Engineered bone development from a pre-osteoblast cell line on three-dimensional scaffolds. Tissue Eng. 6, 605–617 (2000).
Muller-Mai, C.M., Stupp, S.I., Voigt, C. & Gross, U. Nanoapatite and organoapatite implants in bone: histology and ultrastructure of the interface. J. Biomed. Mater. Res. 29, 9–18 (1995).
Reddi, A. Bioceramics, cells and signals in tissue engineering. Bioceramics, Proceedings of the International Symposium on Ceramics in Medicine 11, 9–11 (1998).
Hench, L.L. Bioceramics. J. Am. Ceramic Soc. 81, 1705–1728 (1998).
Bourgeois, B. et al. Calcium-deficient apatite: a first in vivo study concerning bone ingrowth. J. Biomed. Mater. Res. 65A, 402–408 (2003).
Neo, M. et al. A comparative study of ultrastructures of the interfaces between four kinds of surface-active ceramic and bone. J. Biomed. Mater. Res. 26, 1419–1432 (1992).
van Blitterswijk, C.A., Hesseling, S.C., Grote, J.J., Koerten, H.K. & de Groot, K. The biocompatibility of hydroxyapatite ceramic: a study of retrieved human middle ear implants. J. Biomed. Mater. Res. 24, 433–453 (1990).
Le Huec, J.C., Schaeverbeke, T., Clement, D., Faber, J. & Le Rebeller, A. Influence of porosity on the mechanical resistance of hydroxyapatite ceramics under compressive stress. Biomaterials 16, 113–118 (1995).
Sarkar, B.K. Fatigue of brittle materials—a critical appraisal. Bull. Mater. Sci. 18, 755–772 (1995).
Frazza, E.J. & Schmitt, E.E. A new absorbable suture. J. Biomed. Mater. Res. 5, 43–58 (1971).
Rosa, A.L., Beloti, M.M., Van Noort, R., Hatton, P.V. & Devlin, A.J. Surface topography of hydroxyapatite affects ROS17/2.8 cells response. Pesqui. Odontol. Bras. 16, 209–215 (2002).
Okumura, A. et al. Substrate affects the initial attachment and subsequent behavior of human osteoblastic cells (Saos-2). Biomaterials 22, 2263–2271 (2001).
Zhao, F. et al. [Tissue engineering study on chitosan-gelatin/hydroxyapatite composite scaffolds–osteoblasts culture]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 16, 130–133 (2002).
Steiner, C., Elixhauser, A. & Schnaier, J. The healthcare cost and utilization project: an overview. Eff. Clin. Pract. 5, 143–151 (2002).
Hong, L. et al. Bone regeneration at rabbit skull defects treated with transforming growth factor-beta1 incorporated into hydrogels with different levels of biodegradability. J. Neurosurg. 92, 315–325. (2000).
Buttery, L.D. et al. Differentiation of osteoblasts and in vitro bone formation from murine embryonic stem cells. Tissue Eng. 7, 89–99 (2001).
Gundle, R., Joyner, C.J. & Triffitt, J.T. Human bone tissue formation in diffusion chamber culture in vivo by bone-derived cells and marrow stromal fibroblastic cells. Bone 16, 597–601 (1995).
Haynesworth, S.E., Goshima, J., Goldberg, V.M. & Caplan, A.I. Characterization of cells with osteogenic potential from human marrow. Bone 13, 81–88 (1992).
Johnstone, B., Hering, T.M., Caplan, A.I., Goldberg, V.M. & Yoo, J.U. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp. Cell. Res. 238, 265–272 (1998).
Wakitani, S., Saito, T. & Caplan, A.I. Myogenic cells derived from rat bone marrow mesenchymal stem cells exposed to 5-azacytidine. Muscle Nerve 18, 1417–1426 (1995).
Jaiswal, N., Haynesworth, S.E., Caplan, A.I. & Bruder, S.P. Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro . J. Cell Biochem. 64, 295–312 (1997).
Toquet, J. et al. Osteogenic potential in vitro of human bone marrow cells cultured on macroporous biphasic calcium phosphate ceramic. J. Biomed. Mater. Res. 44, 98–108 (1999).
Im, G.I., Kim, D.Y., Shin, J.H., Hyun, C.W. & Cho, W.H. Repair of cartilage defect in the rabbit with cultured mesenchymal stem cells from bone marrow. J. Bone Joint Surg. Br. 83, 289–294 (2001).
Halvorsen, Y.D. et al. Extracellular matrix mineralization and osteoblast gene expression by human adipose tissue-derived stromal cells. Tissue Eng. 7, 729–741 (2001).
Tholpady, S.S., Katz, A.J. & Ogle, R.C. Mesenchymal stem cells from rat visceral fat exhibit multipotential differentiation in vitro . Anat. Rec. 272A, 398–402 (2003).
Zuk, P.A. et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 7, 211–228 (2001).
Zuk, P.A. et al. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 13, 4279–4295 (2002).
Mizuno, H. et al. Myogenic differentiation by human processed lipoaspirate cells. Plast. Reconstr. Surg. 109, 199–209 (2002).
Ashjian, P.H. et al. In vitro differentiation of human processed lipoaspirate cells into early neural progenitors. Plast. Reconstr. Surg. 111, 1922–1931 (2003).
Wickham, M.Q., Erickson, G.R., Gimble, J.M., Vail, T.P. & Guilak, F. Multipotent stromal cells derived from the infrapatellar fat pad of the knee. Clin. Orthop. 412, 196–212 (2003).
Halvorsen, Y.D. et al. Thiazolidinediones and glucocorticoids synergistically induce differentiation of human adipose tissue stromal cells: biochemical, cellular, and molecular analysis. Metabolism 50, 407–413 (2001).
Erickson, G.R. et al. Chondrogenic potential of adipose tissue-derived stromal cells in vitro and in vivo . Biochem. Biophys. Res. Commun. 290, 763–769 (2002).
Safford, K.M. et al. Neurogenic differentiation of murine and human adipose-derived stromal cells. Biochem. Biophys. Res. Commun. 294, 371–379 (2002).
Dragoo, J.L. et al. Bone induction by BMP-2 transduced stem cells derived from human fat. J. Orthop. Res. 21, 622–629 (2003).
Lee, J.A. et al. Biological alchemy: engineering bone and fat from fat-derived stem cells. Ann. Plast. Surg. 50, 610–617 (2003).
Cowan, C.M., Quarto, N., Warren, S.M., Salim, A. & Longaker, M.T. Age-related changes in the biomolecular mechanisms of calvarial osteoblast biology affect FGF-2 signaling and osteogenesis. J. Biol. Chem. 278, 32005–32013 (2003).
Fong, K.D. et al. New strategies for craniofacial repair and replacement: a brief review. J. Craniofac. Surg. 14, 333–339 (2003).
Mulliken, J.B. & Glowacki, J. Induced osteogenesis for repair and construction in the craniofacial region. Plast. Reconstr. Surg. 65, 553–560 (1980).
Bostrom, R. & Mikos, A. (eds.) Tissue Engineering of Bone, vol. 1 (Birkhauser, Boston, 1997).
Lieberman, J.R., Daluiski, A. & Einhorn, T.A. The role of growth factors in the repair of bone. Biology and clinical applications. J. Bone Joint Surg. Am. 84-A, 1032–1044 (2002).
Bradley, J.P. et al. Studies in cranial suture biology: in vitro cranial suture fusion. Cleft Palate Craniofac. J. 33, 150–156 (1996).
Bradley, J.P., Levine, J.P., McCarthy, J.G. & Longaker, M.T. Studies in cranial suture biology: regional dura mater determines in vitro cranial suture fusion. Plast. Reconstr. Surg. 100, 1091–1099 (1997).
Opperman, L.A., Passarelli, R.W., Morgan, E.P., Reintjes, M. & Ogle, R.C. Cranial sutures require tissue interactions with dura mater to resist osseous obliteration in vitro . J. Bone Miner. Res. 10, 1978–1987 (1995).
Opperman, L.A. et al. Dura mater secretes soluble heparin-binding factors required for cranial suture morphogenesis. In vitro cell dev. biol. 32, 150–158 (1996).
Mehrara, B.J. et al. Basic fibroblast growth factor and transforming growth factor beta-1 expression in the developing dura mater correlates with calvarial bone formation. Plast. Reconstr. Surg. 104, 435–444 (1999).
Greenwald, J.A. et al. Regional differentiation of cranial suture-associated dura mater in vivo and in vitro: implications for suture fusion and patency. J. Bone Miner. Res. 15, 2413–2430 (2000).
Jiang, Y. et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418, 41–49 (2002).
Medvinsky, A. & Smith, A. Stem cells: Fusion brings down barriers. Nature 422, 823–825 (2003).
Wang, X. et al. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature 422, 897–901 (2003).
Vassilopoulos, G., Wang, P.R. & Russell, D.W. Transplanted bone marrow regenerates liver by cell fusion. Nature 422, 901–904 (2003).
Wiedmann-Al-Ahmad, M., Gutwald, R., Lauer, G., Hubner, U. & Schmelzeisen, R. How to optimize seeding and culturing of human osteoblast-like cells on various biomaterials. Biomaterials 23, 3319–3328 (2002).
Acknowledgements
This work has been supported by a grant from the National Institutes of Health (R01DE-14526) and the Oak foundation to M.T.L. The authors would like to thank Randall P. Nacamuli for his scientific and editorial assistance. We also acknowledge Koji Iwata and Michael Goris for their assistance with small animal imaging.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Cowan, C., Shi, YY., Aalami, O. et al. Adipose-derived adult stromal cells heal critical-size mouse calvarial defects. Nat Biotechnol 22, 560–567 (2004). https://doi.org/10.1038/nbt958
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt958
This article is cited by
-
Beta-naphthoflavone increases the differentiation of osteoblasts and suppresses adipogenesis in human adipose derived stem cells involving STAT3 pathway
Molecular & Cellular Toxicology (2023)
-
Adipose-derived stromal/stem cells are verified to be potential seed candidates for bio-root regeneration in three-dimensional culture
Stem Cell Research & Therapy (2022)
-
Ultra-low binder content 3D printed calcium phosphate graphene scaffolds as resorbable, osteoinductive matrices that support bone formation in vivo
Scientific Reports (2022)
-
Characterization and functional analysis of the adipose tissue-derived stromal vascular fraction of pediatric patients with osteogenesis imperfecta
Scientific Reports (2022)
-
Cryopreserved human adipose-derived stromal vascular fraction maintains fracture healing potential via angiogenesis and osteogenesis in an immunodeficient rat model
Stem Cell Research & Therapy (2021)