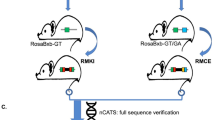
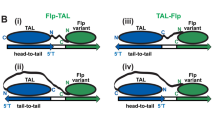
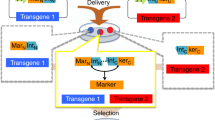
Abstract
Currently two site-specific recombinases are available for engineering the mouse genome: Cre from P1 phage1,2 and Flp from yeast3,4. Both enzymes catalyze recombination between two 34–base pair recognition sites, lox and FRT, respectively, resulting in excision, inversion, or translocation of DNA sequences depending upon the location and the orientation of the recognition sites5,6. Furthermore, strategies have been designed to achieve site-specific insertion or cassette exchange7,8,9,10. The problem with both recombinase systems is that when they insert a circular DNA into the genome (trans event), two cis-positioned recognition sites are created, which are immediate substrates for excision. To stabilize the trans event, functional mutant recognition sites had to be identified8,9,10,11,12. None of the systems, however, allowed efficient selection-free identification of insertion or cassette exchange. Recently, an integrase from Streptomyces phage φC31 has been shown to function in Schizosaccharomyces pombe13 and mammalian14,15 cells. This enzyme recombines between two heterotypic sites: attB and attP. The product sites of the recombination event (attL and attR) are not substrates for the integrase16. Therefore, the φC31 integrase is ideal to facilitate site-specific insertions into the mammalian genome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lakso, M. et al. Targeted oncogene activation by site-specific recombination in transgenic mice. Proc. Natl. Acad. Sci. USA 89, 6232–6236 (1992).
Orban, P.C., Chui, D. & Marth, J.D. Tissue- and site-specific DNA recombination in transgenic mice. Proc. Natl. Acad. Sci. USA 89, 6861–6865 (1992).
Dymecki, S.M. Flp recombinase promotes site-specific DNA recombination in embryonic stem cells and transgenic mice. Proc. Natl. Acad. Sci. USA 93, 6191–6196 (1996).
Rodriguez, C.I. et al. High-efficiency deleter mice show that FLPe is an alternative to Cre-loxP. Nat. Genet. 25, 139–140 (2000).
Lewandoski, M. Conditional control of gene expression in the mouse. Nat. Rev. Genet. 2, 743–755 (2001).
Nagy, A. Cre recombinase: the universal reagent for genome tailoring. Genesis 26, 99–109 (2000).
Koch, K.S. et al. Site-specific integration of targeted DNA into animal cell genomes. Gene 249, 135–144 (2000).
Kolb, A.F. Selection-marker-free modification of the murine β-casein gene using a lox2272 [correction of lox2722] site. Anal. Biochem. 290, 260–271 (2001).
Araki, K., Araki, M. & Yamamura, K. Targeted integration of DNA using mutant lox sites in embryonic stem cells. Nucleic Acids Res. 25, 868–872 (1997).
Schlake, T. & Bode, J. Use of mutated FLP recognition target (FRT) sites for the exchange of expression cassettes at defined chromosomal loci. Biochemistry 33, 12746–12751 (1994).
Lee, G. & Saito, I. Role of nucleotide sequences of loxP spacer region in Cre-mediated recombination. Gene 216, 55–65 (1998).
Seibler, J., Schubeler, D., Fiering, S., Groudine, M. & Bode, J. DNA cassette exchange in ES cells mediated by Flp recombinase: an efficient strategy for repeated modification of tagged loci by marker-free constructs. Biochemistry 37, 6229–6234 (1998).
Thomason, L.C., Calendar, R. & Ow, D.W. Gene insertion and replacement in Schizosaccharomyces pombe mediated by the Streptomyces bacteriophage fC31 site-specific recombination system. Mol. Genet. Genomics 265, 1031–1038 (2001).
Thyagarajan, B., Olivares, E.C., Hollis, R.P., Ginsburg, D.S. & Calos, M.P. Site-specific genomic integration in mammalian cells mediated by phage fC31 integrase. Mol. Cell Biol. 21, 3926–3934 (2001).
Groth, A.C., Olivares, E.C., Thyagarajan, B. & Calos, M.P. A phage integrase directs efficient site-specific integration in human cells. Proc. Natl. Acad. Sci. USA 97, 5995–6000 (2000).
Thorpe, H.M., Wilson, S.E. & Smith, M.C. Control of directionality in the site-specific recombination system of the Streptomyces phage fC31. Mol. Microbiol. 38, 232–241 (2000).
Nagy, A., Rossant, J., Nagy, R., Abramow-Newerly, W. & Roder, J.C. Derivation of completely cell culture-derived mice from early-passage embryonic stem cells. Proc. Natl. Acad. Sci. USA 90, 8424–8428 (1993).
Reddy, S., DeGregori, J.V., von Melchner, H. & Ruley, H.E. Retrovirus promoter-trap vector to induce lacZ gene fusions in mammalian cells. J. Virol. 65, 1507–1515 (1991).
Schaft, J., Ashery-Padan, R., van der Hoeven, F., Gruss, P. & Stewart, A.F. Efficient FLP recombination in mouse ES cells and oocytes. Genesis 31, 6–10 (2001).
Tucker, K.L. et al. Germ-line passage is required for establishment of methylation and expression patterns of imprinted but not of nonimprinted genes. Genes Dev. 10, 1008–1020 (1996).
Nagy, A., Gertsenstein, M., Vintersten, K. & Behringer, R. Manipulating the Mouse Embryos. A Laboratory Manual, 3rd edn. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2003).
Sambrook, J., Fritsch, E.F. & Maniatis, T. Molecular Cloning. A Laboratory Manual. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 1989).
Acknowledgements
We thank Francis Stewart for the pCAAGSFlpeIRESpuro plasmid, and Nikolett Kabacs for technical assistance, and Sandra Tondat and Sarang Kulkarni for generating chimeric mice. We thank Nishma Kassam and Richard Behringer for discussion and excellent proofreading of the manuscript. This work was supported by funds from NIH (grant no. 5 R01 DA14784 02) and a grant from the Multiple Sclerosis Society of Canada. A.N. is a senior scientist of CIHR.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Belteki, G., Gertsenstein, M., Ow, D. et al. Site-specific cassette exchange and germline transmission with mouse ES cells expressing φC31 integrase. Nat Biotechnol 21, 321–324 (2003). https://doi.org/10.1038/nbt787
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt787
This article is cited by
-
Double knock-in pig models with elements of binary Tet-On and phiC31 integrase systems for controllable and switchable gene expression
Science China Life Sciences (2022)
-
Genetic Manipulations in Dermatophytes
Mycopathologia (2017)
-
An efficient strategy for producing a stable, replaceable, highly efficient transgene expression system in silkworm, Bombyx mori
Scientific Reports (2015)
-
Cytoprotection by the NO-Donor SNAP Against Ischemia/Reoxygenation Injury in Mouse Embryonic Stem Cell-Derived Cardiomyocytes
Molecular Biotechnology (2014)
-
Genome editing and animal models
Chinese Science Bulletin (2014)