Abstract
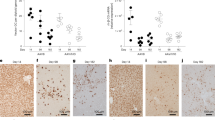
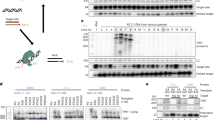
A major limitation of adenovirus-mediated gene therapy for inherited diseases is the instability of transgene expression in vivo, which originates at least in part from the loss of the linear, extrachromosomal vector genomes. Herein we describe the production of a gene-deleted adenovirus–transposon vector that stably maintains virus-encoded transgenes in vivo through integration into host cell chromosomes. This system utilizes a donor transposon vector that undergoes Flp-mediated recombination and excision of its therapeutic payload in the presence of the Flp and Sleeping Beauty recombinases. Systemic in vivo delivery of this system resulted in efficient generation of transposon circles and stable transposase-mediated integration in mouse liver. Somatic integration was sufficient to maintain therapeutic levels of human coagulation Factor IX for more than six months in mice undergoing extensive liver proliferation. These vectors combine the versatility of adenoviral vectors with the integration capabilities of a eukaryotic DNA transposon and should prove useful in the treatment of genetic diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Kozarsky, K.F. & Wilson, J.M. Gene therapy: adenovirus vectors. Curr. Opin. Genet. Dev. 3, 499–503 (1993).
Yang, Y. et al. Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy. Proc. Natl. Acad. Sci. USA 91, 4407–4411 (1994).
Yang, Y., Li, Q., Ertl, H.C. & Wilson, J.M. Cellular and humoral immune responses to viral antigens create barriers to lung-directed gene therapy with recombinant adenoviruses. J. Virol. 69, 2004–2015 (1995).
Yang, Y., Jooss, K.U., Su, Q., Ertl, H.C. & Wilson, J.M. Immune responses to viral antigens versus transgene product in the elimination of recombinant adenovirus-infected hepatocytes in vivo. Gene Ther. 3, 137–144 (1996).
Parks, R.J. et al. A helper-dependent adenovirus vector system: removal of helper virus by Cre-mediated excision of the viral packaging signal. Proc. Natl. Acad. Sci. USA 93, 13565–13570 (1996).
Schiedner, G. et al. Genomic DNA transfer with a high-capacity adenovirus vector results in improved in vivo gene expression and decreased toxicity. Nat. Genet. 18, 180–183 (1998).
Balague, C. et al. Sustained high-level expression of full-length human Factor VIII and restoration of clotting activity in hemophilic mice using a minimal adenovirus vector. Blood 95, 820–828 (2000).
Clemens, P.R. et al. In vivo muscle gene transfer of full-length dystrophin with an adenoviral vector that lacks all viral genes. Gene Ther. 3, 965–972 (1996).
Morral, N. et al. High doses of a helper-dependent adenoviral vector yield supraphysiological levels of α1-antitrypsin with negligible toxicity. Hum. Gene Ther. 9, 2709–2716 (1998).
Morral, N. et al. Administration of helper-dependent adenoviral vectors and sequential delivery of different vector serotype for long-term liver-directed gene transfer in baboons. Proc. Natl. Acad. Sci. USA 96, 12816–12821 (1999).
Ehrhardt, A. & Kay, M.A. A new adenoviral helper-dependent vector results in long-term therapeutic levels of human coagulation Factor IX at low doses in vivo. Blood 99, 1–8 (2002).
Kim, I.H., Jozkowicz, A., Piedra, P.A., Oka, K. & Chan, L. Lifetime correction of genetic deficiency in mice with a single injection of helper-dependent adenoviral vector. Proc. Natl. Acad. Sci. USA 98, 13282–13287 (2001).
Harui, A., Suzuki, S., Kochanek, S. & Mitani, K. Frequency and stability of chromosomal integration of adenovirus vectors. J. Virol. 73, 6141–6146 (1999).
Hillgenberg, M., Tonnies, H. & Strauss, M. Chromosomal integration pattern of a helper-dependent minimal adenovirus vector with a selectable marker inserted into a 27.4-kilobase genomic stuffer. J. Virol. 75, 9896–9908 (2001).
Plasterk, R.H., Izsvak, Z. & Ivics, Z. Resident aliens: the Tc1/mariner superfamily of transposable elements. Trends Genet. 15, 326–332 (1999).
Fischer, S.E., Wienholds, E. & Plasterk, R.H. Regulated transposition of a fish transposon in the mouse germ line. Proc. Natl. Acad. Sci. USA 98, 6759–6764 (2001).
Ivics, Z., Hackett, P.B., Plasterk, R.H. & Izsvak, Z. Molecular reconstruction of Sleeping Beauty, a Tc1-like transposon from fish, and its transposition in human cells. Cell 91, 501–510 (1997).
Yant, S.R. et al. Somatic integration and long-term transgene expression in normal and haemophilic mice using a DNA transposon system. Nat. Genet. 25, 35–41 (2000).
Nakai, H. et al. Extrachromosomal recombinant adeno-associated virus vector genomes are primarily responsible for stable liver transduction in vivo. J. Virol. 75, 6969–6976 (2001).
Vos, J.C., De Baere, I. & Plasterk, R.H. Transposase is the only nematode protein required for in vitro transposition of Tc1. Genes Dev. 10, 755–761 (1996).
Ng, P., Beauchamp, C., Evelegh, C., Parks, R. & Graham, F.L. Development of a FLP/frt system for generating helper-dependent adenoviral vectors. Mol. Ther. 3, 809–815 (2001).
Rodriguez, C.I. et al. High-efficiency deleter mice show that FLPe is an alternative to Cre-loxP. Nat. Genet. 25, 139–140 (2000).
Plasterk, R.H. The Tc1/mariner transposon family. Curr. Top. Microbiol. Immunol. 204, 125–143 (1996).
Miller, D.G., Rutledge, E.A. & Russell, D.W. Chromosomal effects of adeno-associated virus vector integration. Nat. Genet. 30, 147–148 (2002).
Nakai, H., Iwaki, Y., Kay, M.A. & Couto, L.B. Isolation of recombinant adeno-associated virus vector–cellular DNA junctions from mouse liver. J. Virol. 73, 5438–5447 (1999).
Chalmers, R., Guhathakurta, A., Benjamin, H. & Kleckner, N. IHF modulation of Tn10 transposition: sensory transduction of supercoiling status via a proposed protein/DNA molecular spring. Cell 93, 897–908 (1998).
Crellin, P. & Chalmers, R. Protein–DNA contacts and conformational changes in the Tn10 transpososome during assembly and activation for cleavage. EMBO J. 20, 3882–3891 (2001).
Wang, Z. & Harshey, R.M. Crucial role for DNA supercoiling in Mu transposition: a kinetic study. Proc. Natl. Acad. Sci. USA 91, 699–703 (1994).
Soifer, H. et al. Stable integration of transgenes delivered by a retrotransposon–adenovirus hybrid vector. Hum. Gene Ther. 12, 1417–1428 (2001).
Lieber, A., Steinwaerder, D.S., Carlson, C.A. & Kay, M.A. Integrating adenovirus-adeno-associated virus hybrid vectors devoid of all viral genes. J. Virol. 73, 9314–9324 (1999).
Recchia, A. et al. Site-specific integration mediated by a hybrid adenovirus/adeno-associated virus vector. Proc. Natl. Acad. Sci. USA 96, 2615–2620 (1999).
Zheng, C., Baum, B.J., Iadarola, M.J. & O'Connell, B.C. Genomic integration and gene expression by a modified adenoviral vector. Nat. Biotechnol. 18, 176–180 (2000).
Murphy, S.J., Chong, H., Bell, S., Diaz, R.M. & Vile, R.G. Novel integrating adenoviral/retroviral hybrid vector for gene therapy. Hum. Gene Ther. 13, 745–760 (2002).
Soifer, H. et al. A novel, helper-dependent, adenovirus–retrovirus hybrid vector: stable transduction by a two-stage mechanism. Mol. Ther. 5, 599–608 (2002).
Shayakhmetov, D.M. et al. A high-capacity, capsid-modified hybrid adenovirus/adeno-associated virus vector for stable transduction of human hematopoietic cells. J. Virol. 76, 1135–1143 (2002).
Feng, M. et al. Stable in vivo gene transduction via a novel adenoviral/retroviral chimeric vector. Nat. Biotechnol. 15, 866–870 (1997).
Luo, G., Ivics, Z., Izsvak, Z. & Bradley, A. Chromosomal transposition of a Tc1/mariner-like element in mouse embryonic stem cells. Proc. Natl. Acad. Sci. USA 95, 10769–10773 (1998).
Peters, A.H. et al. Absence of germline infection in male mice following intraventricular injection of adenovirus. Mol. Ther. 4, 603–613 (2001).
Kanatsu-Shinohara, M. et al. Adenovirus-mediated gene delivery and in vitro microinsemination produce offspring from infertile male mice. Proc. Natl. Acad. Sci. USA 99, 1383–1388 (2002).
Tallone, T. et al. A mouse model for adenovirus gene delivery. Proc. Natl. Acad. Sci. USA 98, 7910–7915 (2001).
Steinwaerder, D.S. & Lieber, A. Insulation from viral transcriptional regulatory elements improves inducible transgene expression from adenovirus vectors in vitro and in vivo. Gene Ther. 7, 556–567 (2000).
Groth, A.C., Olivares, E.C., Thyagarajan, B. & Calos, M.P. A phage integrase directs efficient site-specific integration in human cells. Proc. Natl. Acad. Sci. USA 97, 5995–6000 (2000).
Thyagarajan, B., Olivares, E.C., Hollis, R.P., Ginsburg, D.S. & Calos, M.P. Site-specific genomic integration in mammalian cells mediated by phage phiC31 integrase. Mol. Cell. Biol. 21, 3926–3934 (2001).
Burr, A.W., Carpenter, M.R., Hines, J.E., Gullick, W.J. & Burt, A.D. Intrahepatic distribution of transforming growth factor-α (TGFα) during liver regeneration following carbon tetrachloride-induced necrosis. J. Pathol. 170, 95–100 (1993).
Liu, F., Song, Y. & Liu, D. Hydrodynamics-based transfection in animals by systemic administration of plasmid DNA. Gene Ther. 6, 1258–1266 (1999).
Zhang, G., Budker, V. & Wolff, J.A. High levels of foreign gene expression in hepatocytes after tail vein injections of naked plasmid DNA. Hum. Gene Ther. 10, 1735–1737 (1999).
McGrory, W.J., Bautista, D.S. & Graham, F.L. A simple technique for the rescue of early region I mutations into infectious human adenovirus type 5. Virology 163, 614–617 (1988).
Kay, M.A., Graham, F., Leland, F. & Woo, S.L. Therapeutic serum concentrations of human α1-antitrypsin after adenoviral-mediated gene transfer into mouse hepatocytes. Hepatology 21, 815–819 (1995).
Izsvak, Z., Ivics, Z. & Plasterk, R.H. Sleeping Beauty, a wide host-range transposon vector for genetic transformation in vertebrates. J. Mol. Biol. 302, 93–102 (2000).
Acknowledgements
We thank K. Ohashi for helpful discussions, J. Chamberlain for providing the C7-Cre cells, and S. Dymecki for providing Flp/FRT-based plasmids. This work was supported by NIH grant DK49022 (M.A.K.). A.E. is the recipient of a Judith Pool National Hemophilia Fellowship. J.G.M. was funded by grants from the Carlsberg Foundation and the Danish Medical Research Council.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Yant, S., Ehrhardt, A., Mikkelsen, J. et al. Transposition from a gutless adeno-transposon vector stabilizes transgene expression in vivo. Nat Biotechnol 20, 999–1005 (2002). https://doi.org/10.1038/nbt738
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt738
This article is cited by
-
Programming CAR-T cells to kill cancer
Nature Biomedical Engineering (2018)
-
Retroviral vectors and transposons for stable gene therapy: advances, current challenges and perspectives
Journal of Translational Medicine (2016)
-
Transcriptionally regulated and nontoxic delivery of the hyperactive Sleeping Beauty Transposase
Molecular Therapy - Methods & Clinical Development (2016)
-
A High-Capacity Adenoviral Hybrid Vector System Utilizing the Hyperactive Sleeping Beauty Transposase SB100X for Enhanced Integration
Molecular Therapy - Nucleic Acids (2016)
-
Hybrid Nonviral/Viral Vector Systems for Improved piggyBac DNA Transposon In Vivo Delivery
Molecular Therapy (2015)