Abstract
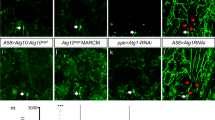
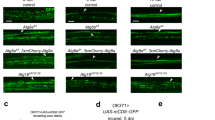
The cellular machinery promoting phagocytosis of corpses of apoptotic cells is well conserved from worms to mammals. An important component is the Caenorhabditis elegans engulfment receptor CED-1 (ref. 1) and its Drosophila orthologue, Draper2. The CED-1/Draper signalling pathway is also essential for the phagocytosis of other types of ‘modified self’ including necrotic cells3, developmentally pruned axons4,5 and dendrites6, and axons undergoing Wallerian degeneration7. Here we show that Drosophila Shark, a non-receptor tyrosine kinase similar to mammalian Syk and Zap-70, binds Draper through an immunoreceptor tyrosine-based activation motif (ITAM) in the Draper intracellular domain. We show that Shark activity is essential for Draper-mediated signalling events in vivo, including the recruitment of glial membranes to severed axons and the phagocytosis of axonal debris and neuronal cell corpses by glia. We also show that the Src family kinase (SFK) Src42A can markedly increase Draper phosphorylation and is essential for glial phagocytic activity. We propose that ligand-dependent Draper receptor activation initiates the Src42A-dependent tyrosine phosphorylation of Draper, the association of Shark and the activation of the Draper pathway. These Draper–Src42A–Shark interactions are strikingly similar to mammalian immunoreceptor–SFK–Syk signalling events in mammalian myeloid and lymphoid cells8,9. Thus, Draper seems to be an ancient immunoreceptor with an extracellular domain tuned to modified self, and an intracellular domain promoting phagocytosis through an ITAM-domain–SFK–Syk-mediated signalling cascade.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zhou, Z., Hartwieg, E. & Horvitz, H. R. CED-1 is a transmembrane receptor that mediates cell corpse engulfment in C. elegans . Cell 104, 43–56 (2001)
Freeman, M. R., Delrow, J., Kim, J., Johnson, E. & Doe, C. Q. Unwrapping glial biology. Gcm target genes regulating glial development, diversification, and function. Neuron 38, 567–580 (2003)
Chung, S., Gumienny, T. L., Hengartner, M. O. & Driscoll, M. A common set of engulfment genes mediates removal of both apoptotic and necrotic cell corpses in C. elegans . Nature Cell Biol. 2, 931–937 (2000)
Awasaki, T. et al. Essential role of the apoptotic cell engulfment genes draper and ced-6 in programmed axon pruning during Drosophila metamorphosis. Neuron 50, 855–867 (2006)
Hoopfer, E. D. et al. Wlds protection distinguishes axon degeneration following injury from naturally occurring developmental pruning. Neuron 50, 883–895 (2006)
Williams, D. W., Kondo, S., Krzyzanowska, A., Hiromi, Y. & Truman, J. W. Local caspase activity directs engulfment of dendrites during pruning. Nature Neurosci. 9, 1234–1236 (2006)
MacDonald, J. M. et al. The Drosophila cell corpse engulfment receptor Draper mediates glial clearance of severed axons. Neuron 50, 869–881 (2006)
Fodor, S., Jakus, Z. & Mocsai, A. ITAM-based signaling beyond the adaptive immune response. Immunol. Lett. 104, 29–37 (2006)
Underhill, D. M. & Goodridge, H. S. The many faces of ITAMs. Trends Immunol. 28, 66–73 (2007)
Hengartner, M. O. Programmed cell death in the nematode C. elegans . Recent Prog. Horm. Res. 54, 213–222 (1999)
Fadok, V. A., Bratton, D. L. & Henson, P. M. Phagocyte receptors for apoptotic cells: recognition, uptake, and consequences. J. Clin. Invest. 108, 957–962 (2001)
Henson, P. M., Bratton, D. L. & Fadok, V. A. Apoptotic cell removal. Curr. Biol. 11, R795–R805 (2001)
Savill, J., Dransfield, I., Gregory, C. & Haslett, C. A blast from the past: clearance of apoptotic cells regulates immune responses. Nature Rev. Immunol. 2, 965–975 (2002)
Su, H. P. et al. Interaction of CED-6/GULP, an adapter protein involved in engulfment of apoptotic cells with CED-1 and CD91/low density lipoprotein receptor-related protein (LRP). J. Biol. Chem. 277, 11772–11779 (2002)
Liu, Q. A. & Hengartner, M. O. Candidate adaptor protein CED-6 promotes the engulfment of apoptotic cells in C. elegans. . Cell 93, 961–972 (1998)
Ellis, R. E., Jacobson, D. M. & Horvitz, H. R. Genes required for the engulfment of cell corpses during programmed cell death in Caenorhabditis elegans . Genetics 129, 79–94 (1991)
Kinchen, J. M. et al. Two pathways converge at CED-10 to mediate actin rearrangement and corpse removal in C. elegans . Nature 434, 93–99 (2005)
Yu, X., Odera, S., Chuang, C. H., Lu, N. & Zhou, Z. C. elegans Dynamin mediates the signaling of phagocytic receptor CED-1 for the engulfment and degradation of apoptotic cells. Dev. Cell 10, 743–757 (2006)
Watts, R. J., Schuldiner, O., Perrino, J., Larsen, C. & Luo, L. Glia engulf degenerating axons during developmental axon pruning. Curr. Biol. 14, 678–684 (2004)
Awasaki, T. & Ito, K. Engulfing action of glial cells is required for programmed axon pruning during Drosophila metamorphosis. Curr. Biol. 14, 668–677 (2004)
Aldskogius, H. & Kozlova, E. N. Central neuron-glial and glial-glial interactions following axon injury. Prog. Neurobiol. 55, 1–26 (1998)
Ferrante, A. W., Reinke, R. & Stanley, E. R. Shark, a Src homology 2, ankyrin repeat, tyrosine kinase, is expressed on the apical surfaces of ectodermal epithelia. Proc. Natl Acad. Sci. USA 92, 1911–1915 (1995)
Biswas, R., Stein, D. & Stanley, E. R. Drosophila Dok is required for embryonic dorsal closure. Development 133, 217–227 (2006)
Berton, G., Mocsai, A. & Lowell, C. A. Src and Syk kinases: key regulators of phagocytic cell activation. Trends Immunol. 26, 208–214 (2005)
Fernandez, R. et al. The Drosophila shark tyrosine kinase is required for embryonic dorsal closure. Genes Dev. 14, 604–614 (2000)
Monroe, J. G. ITAM-mediated tonic signalling through pre-BCR and BCR complexes. Nature Rev. Immunol. 6, 283–294 (2006)
Pitcher, L. A. & van Oers, N. S. T-cell receptor signal transmission: who gives an ITAM? Trends Immunol. 24, 554–560 (2003)
Cox, D. & Greenberg, S. Phagocytic signaling strategies: Fcγ receptor-mediated phagocytosis as a model system. Semin. Immunol. 13, 339–345 (2001)
Couto, A., Alenius, M. & Dickson, B. J. Molecular, anatomical, and functional organization of the Drosophila olfactory system. Curr. Biol. 15, 1535–1547 (2005)
Keegan, K. & Cooper, J. A. Use of the two hybrid system to detect the association of the protein-tyrosine-phosphatase, SHPTP2, with another SH2-containing protein, Grb7. Oncogene 12, 1537–1544 (1996)
Lioubin, M. N. et al. p150Ship, a signal transduction molecule with inositol polyphosphate-5-phosphatase activity. Genes Dev. 10, 1084–1095 (1996)
Li, W., Yeung, Y. G. & Stanley, E. R. Tyrosine phosphorylation of a common 57-kDa protein in growth factor- stimulated and -transformed cells. J. Biol. Chem. 266, 6808–6814 (1991)
Acknowledgements
This work was supported by National Institutes of Health grants (1R01NS053538 to M.R.F., and 1RO1GM55293 and 1RO1CA26504 to E.R.S.), by an Albert Einstein Cancer Center Grant (PO3-13330 to E.R.S.), a Smith Family New Investigator Award (to M.R.F.) from the Smith Family Foundation, and a grant from the Christopher and Dana Reeves Foundation (to M.R.F.). M.R.F. is an Alfred P. Sloan Research Fellow.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary information
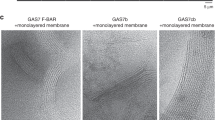
The file contains Supplementary Figure 1 showing RNAi knockdown of Src64B or Btk29A does not affect glial responses to axon injury or clearance of severed axons from the CNS. These experiments show that RNAi knockdown in glia of Src64b or btk29A, the only other Src kinases in Drosophila besides Src42a, do not affect glial responses to axonal injury, nor the ability of glia to phagocytose degenerating axonal debris. (PDF 3080 kb)
Rights and permissions
About this article
Cite this article
Ziegenfuss, J., Biswas, R., Avery, M. et al. Draper-dependent glial phagocytic activity is mediated by Src and Syk family kinase signalling. Nature 453, 935–939 (2008). https://doi.org/10.1038/nature06901
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06901
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.