Abstract
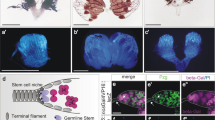
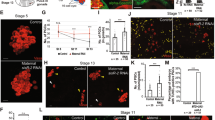
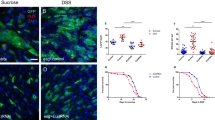
One of the key characteristics of stem cells is their capacity to divide for long periods of time in an environment where most of the cells are quiescent. Therefore, a critical question in stem cell biology is how stem cells escape cell division stop signals. Here, we report the necessity of the microRNA (miRNA) pathway1,2,3,4 for proper control of germline stem cell (GSC) division in Drosophila melanogaster. Analysis of GSCs mutant for dicer-1 (dcr-1), the double-stranded RNaseIII essential for miRNA biogenesis, revealed a marked reduction in the rate of germline cyst production. These dcr-1 mutant GSCs exhibit normal identity but are defective in cell cycle control. On the basis of cell cycle markers and genetic interactions, we conclude that dcr-1 mutant GSCs are delayed in the G1 to S transition, which is dependent on the cyclin-dependent kinase inhibitor Dacapo, suggesting that miRNAs are required for stem cells to bypass the normal G1/S checkpoint. Hence, the miRNA pathway might be part of a mechanism that makes stem cells insensitive to environmental signals that normally stop the cell cycle at the G1/S transition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
He, L. & Hannon, G. J. MicroRNAs: small RNAs with a big role in gene regulation. Nature Rev. Genet. 5, 522–531 (2004)
Nakahara, K. & Carthew, R. W. Expanding roles for miRNAs and siRNAs in cell regulation. Curr. Opin. Cell Biol. 16, 127–133 (2004)
Bartel, B. & Bartel, D. P. MicroRNAs: at the root of plant development? Plant Physiol. 132, 709–717 (2003)
Ambros, V. microRNAs: tiny regulators with great potential. Cell 107, 823–826 (2001)
Bernstein, E. et al. Dicer is essential for mouse development. Nature Genet. 35, 215–217 (2003)
Schauer, S. E., Jacobsen, S. E., Meinke, D. W. & Ray, A. DICER-LIKE1: blind men and elephants in Arabidopsis development. Trends Plant Sci. 7, 487–491 (2002)
Carmell, M. A., Xuan, Z., Zhang, M. Q. & Hannon, G. J. The Argonaute family: tentacles that reach into RNAi, developmental control, stem cell maintenance, and tumorigenesis. Genes Dev. 16, 2733–2742 (2002)
Suh, M. R. et al. Human embryonic stem cells express a unique set of microRNAs. Dev. Biol. 270, 488–498 (2004)
Houbaviy, H. B., Murray, M. F. & Sharp, P. A. Embryonic stem cell-specific MicroRNAs. Dev. Cell 5, 351–358 (2003)
Lee, Y. S. et al. Distinct roles for Drosophila Dicer-1 and Dicer-2 in the siRNA/miRNA silencing pathways. Cell 117, 69–81 (2004)
Spradling, A., Drummond-Barbosa, D. & Kai, T. Stem cells find their niche. Nature 414, 98–104 (2001)
Gilboa, L. & Lehmann, R. How different is Venus from Mars? The genetics of germ-line stem cells in Drosophila females and males. Development 131, 4895–4905 (2004)
Johnson, J., Canning, J., Kaneko, T., Pru, J. K. & Tilly, J. L. Germline stem cells and follicular renewal in the postnatal mammalian ovary. Nature 428, 145–150 (2004)
Kai, T. & Spradling, A. Differentiating germ cells can revert into functional stem cells in Drosophila melanogaster ovaries. Nature 428, 564–569 (2004)
Newfeld, S. J. et al. Mothers against dpp participates in a DDP/TGF-β responsive serine-threonine kinase signal transduction cascade. Development 124, 3167–3176 (1997)
Song, X. et al. Bmp signals from niche cells directly repress transcription of a differentiation-promoting gene, bag of marbles, in germline stem cells in the Drosophila ovary. Development 131, 1353–1364 (2004)
Shcherbata, H. R., Althauser, C., Findley, S. D. & Ruohola-Baker, H. The mitotic-to-endocycle switch in Drosophila follicle cells is executed by Notch-dependent regulation of G1/S, G2/M and M/G1 cell-cycle transitions. Development 131, 3169–3181 (2004)
Calvi, B. R. & Lilly, M. A. Fluorescent BrdU labeling and nuclear flow sorting of the Drosophila ovary. Methods Mol. Biol. 247, 203–213 (2004)
Gonczy, P. & DiNardo, S. The germ line regulates somatic cyst cell proliferation and fate during Drosophila spermatogenesis. Development 122, 2437–2447 (1996)
Kiger, A. A., Jones, D. L., Schulz, C., Rogers, M. B. & Fuller, M. T. Stem cell self-renewal specified by JAK-STAT activation in response to a support cell cue. Science 294, 2542–2545 (2001)
de Nooij, J. C., Letendre, M. A. & Hariharan, I. K. A cyclin-dependent kinase inhibitor, Dacapo, is necessary for timely exit from the cell cycle during Drosophila embryogenesis. Cell 87, 1237–1247 (1996)
Pavletich, N. P. Mechanisms of cyclin-dependent kinase regulation: structures of Cdks, their cyclin activators, and Cip and INK4 inhibitors. J. Mol. Biol. 287, 821–828 (1999)
Meyer, C. A. et al. Drosophila p27Dacapo expression during embryogenesis is controlled by a complex regulatory region independent of cell cycle progression. Development 129, 319–328 (2002)
Datto, M. B. et al. Transforming growth factor beta induces the cyclin-dependent kinase inhibitor p21 through a p53-independent mechanism. Proc. Natl Acad. Sci. USA 92, 5545–5549 (1995)
Lane, M. E. et al. Dacapo, a cyclin-dependent kinase inhibitor, stops cell proliferation during Drosophila development. Cell 87, 1225–1235 (1996)
Enright, A. J. et al. MicroRNA targets in Drosophila . Genome Biol. 5, R1 (2003)
Stark, A., Brennecke, J., Russell, R. B. & Cohen, S. M. Identification of Drosophila MicroRNA targets. PLoS Biol. 1, E60 (2003)
Lewis, B. P., Shih, I. H., Jones-Rhoades, M. W., Bartel, D. P. & Burge, C. B. Prediction of mammalian microRNA targets. Cell 115, 787–798 (2003)
He, L. et al. A microRNA polycistron as a potential human oncogene. Nature doi:10.1038/nature03552 (in the press)
Margolis, J. & Spradling, A. Identification and behavior of epithelial stem cells in the Drosophila ovary. Development 121, 3797–3807 (1995)
Acknowledgements
We thank B. Calvi, B. Wakimoto and E. Ward for comments on the manuscript, and the members of the H.R.-B. laboratory for suggestions throughout the course of this study. We also thank E. Kerr for making dacapo constructs; V. Shcherbatyy for creating transgenic lines; B. Akiyashi for UAS-Dap experiments; K. Kim for the dcr-1d102 strain; and M. Lilly, C. Lehner, I. Hariharan, T. Orr-Weaver, D. McKearin, P. ten Dijke, A. Spradling and C.-H. Heldin for flies, antibodies and advice. This work was supported by the Schultz Fellowship for S.D.H., grants from the National Institutes of Health to R.W.C and H. R.-B., and the American Heart Association and the American Cancer Society to H.R.-B.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods, additional references, Supplementary Table S1 and Supplementary Figure Legends. (DOC 50 kb)
Supplementary Figure S1
The dcr-1d102 allele yields a phenotype identical to, albeit slightly milder than the dcr-1Q1147X allele. (JPG 309 kb)
Supplementary Figure S2
The dcr1-related reduction in cell division rate and cell cycle delay are germline stem cell-specific. (a-a'') (JPG 455 kb)
Supplementary Figure S3
Analysis of Dacapo expression in GSCs. (JPG 459 kb)
Supplementary Figure S4
Computational prediction of miRNA binding sites in the dap 3' UTR. (DOC 29 kb)
Rights and permissions
About this article
Cite this article
Hatfield, S., Shcherbata, H., Fischer, K. et al. Stem cell division is regulated by the microRNA pathway. Nature 435, 974–978 (2005). https://doi.org/10.1038/nature03816
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature03816
This article is cited by
-
Transcriptome Analysis Reveals the Regulation Role of miR-144-5p in Intestinal Immunity of Japanese Flounder (Paralichthys olivaceus)
Journal of Ocean University of China (2023)
-
Assessment of Association between miR-146a Polymorphisms and Expression of miR-146a, TRAF-6, and IRAK-1 Genes in Patients with Brucellosis
Molecular Biology Reports (2022)
-
ANMDA: anti-noise based computational model for predicting potential miRNA-disease associations
BMC Bioinformatics (2021)
-
RNA interference of an orthologue of Dicer of Meloidogyne incognita alludes to the gene’s importance in nematode development
Scientific Reports (2021)
-
OCT4 maintains self-renewal and reverses senescence in human hair follicle mesenchymal stem cells through the downregulation of p21 by DNA methyltransferases
Stem Cell Research & Therapy (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.