Abstract
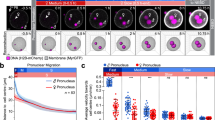
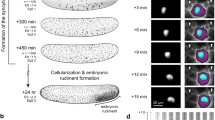
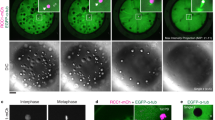
Studies of experimentally manipulated embryos1,2,3,4 have led to the long-held conclusion that the polarity of the mouse embryo remains undetermined until the blastocyst stage. However, recent studies5,6,7 reporting that the embryonic–abembryonic axis of the blastocyst arises perpendicular to the first cleavage plane, and hence to the animal–vegetal axis of the zygote, have led to the claim that the axis of the mouse embryo is already specified in the egg. Here we show that there is no specification of the axis in the egg. Time-lapse recordings show that the second polar body does not mark a stationary animal pole, but instead, in half of the embryos, moves towards a first cleavage plane. The first cleavage plane coincides with the plane defined by the two apposing pronuclei once they have moved to the centre of the egg. Pronuclear transfer experiments confirm that the first cleavage plane is not determined in early interphase but rather is specified by the newly formed topology of the two pronuclei. The microtubule networks that allow mixing of parental chromosomes before dividing into two may be involved in these processes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Tarkowski, A. K. Experiments on the development of isolated blastomeres of mouse eggs. Nature 184, 1286–1287 (1959)
Tarkowski, A. K. & Wroblewska, J. Development of blastomeres of mouse eggs isolated at the 4- and 8-cell stage. J. Embryol. Exp. Morphol. 18, 155–180 (1967)
Rossant, J. Postimplantation development of blastomeres isolated from 4- and 8-cell mouse eggs. J. Embryol. Exp. Morphol. 36, 283–290 (1976)
Tarkowski, A. K. Mouse chimaeras developed from fused eggs. Nature 190, 857–860 (1961)
Gardner, R. L. The early blastocyst is bilaterally symmetrical and its axis of symmetry is aligned with the animal–vegetal axis of the zygote in the mouse. Development 124, 289–301 (1997)
Gardner, R. L. Specification of embryonic axes begins before cleavage in normal mouse development. Development 128, 839–847 (2001)
Piotrowska, K., Wianny, F., Pedersen, R. A. & Zernicka-Goetz, M. Blastomeres arising from the first cleavage division have distinguishable fates in normal mouse development. Development 128, 3739–3748 (2001)
Piotrowska, K. & Zernicka-Goetz, M. Role for sperm in spatial patterning of the early mouse embryo. Nature 409, 517–521 (2001)
Plusa, B., Grabarek, J. B., Piotrowska, K., Glover, D. M. & Zernicka-Goetz, M. Site of the previous meiotic division defines cleavage orientation in the mouse embryo. Nature Cell Biol. 4, 811–815 (2002)
Schatten, G., Simerly, C. & Schatten, H. Microtubule configurations during fertilization, mitosis, and early development in the mouse and the requirement for egg microtubule-mediated motility during mammalian fertilization. Proc. Natl Acad. Sci. USA 82, 4152–4156 (1985)
Maro, B., Johnson, M. H., Webb, M. & Flach, G. Mechanism of polar body formation in the mouse oocyte: an interaction between the chromosomes, the cytoskeleton and the plasma membrane. J. Embryol. Exp. Morphol. 92, 11–32 (1986)
Maro, B., Howlett, S. K. & Webb, M. Non-spindle microtubule organizing centers in metaphase II-arrested mouse oocytes. J. Cell Biol. 101, 1665–1672 (1985)
Schatten, H., Schatten, G., Mazia, D., Balczon, R. & Simerly, C. Behavior of centrosomes during fertilization and cell division in mouse oocytes and in sea urchin eggs. Proc. Natl Acad. Sci. USA 83, 105–109 (1986)
Canman, J. C. et al. Determining the position of the cell division plane. Nature 424, 1074–1078 (2003)
Piotrowska, K. & Zernicka-Goetz, M. Early patterning of the mouse embryo–contributions of sperm and egg. Development 129, 5803–5813 (2002)
Johnson, M. H., Eager, D., Muggleton-Harris, A. & Grave, H. M. Mosaicism in organisation concanavalin A receptors on surface membrane of mouse egg. Nature 257, 321–322 (1975)
Mayer, W., Smith, A., Fundele, R. & Haaf, T. Spatial separation of parental genomes in preimplantation mouse embryos. J. Cell Biol. 148, 629–634 (2000)
Paweletz, N., Mazia, D. & Finze, E. M. Fine structural studies of the bipolarization of the mitotic apparatus in the fertilized sea urchin egg. II. Bipolarization before the first mitosis. Eur. J. Cell Biol. 44, 205–213 (1987)
Schatten, G. Sperm incorporation, the pronuclear migrations, and their relation to the establishment of the first embryonic axis: time-lapse video microscopy of the movements during fertilization of the sea urchin Lytechinus variegatus. Dev. Biol. 86, 426–437 (1981)
Zamboni, L., Chakraborty, J. & Smith, D. M. First cleavage division of the mouse zygote. An ultrastructural study. Biol. Reprod. 7, 170–193 (1972)
Heald, R. et al. Self-organization of microtubules into bipolar spindles around artificial chromosomes in Xenopus egg extracts. Nature 382, 420–425 (1996)
Carazo-Salas, R. E. & Karsenti, E. Long-range communication between chromatin and microtubules in Xenopus egg extracts. Curr. Biol. 13, 1728–1733 (2003)
Alarcon, V. B. & Marikawa, Y. Deviation of the blastocyst axis from the first cleavage plane does not affect the quality of mouse postimplantation development. Biol. Reprod. 69, 1208–1212 (2003)
Ho, Y., Wigglesworth, K., Eppig, J. J. & Schultz, R. M. Preimplantation development of mouse embryos in KSOM: augmentation by amino acids and analysis of gene expression. Mol. Reprod. Dev. 41, 232–238 (1995)
McGrath, J. & Solter, D. Nuclear transplantation in the mouse embryo by microsurgery and cell fusion. Science 220, 1300–1302 (1983)
Acknowledgements
We thank Y. Kaneda for HVJ; Z. Polanski, N. Bobola, A. Tomilin, P. Nielsen, R. Cassada and M. Hoffman for discussions and reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Movie 1
This movie shows a time-lapse recording of the embryos developing from zygote to 2-cell. The time-lapse corresponds to the sequential pictures of Fig.1b, covering a period of 7.5 hours. (MOV 1582 kb)
Supplementary Movie 2
This movie shows a time-lapse recording of the embryos developing from zygote to 2-cell. The time-lapse corresponds to the sequential pictures of Fig.1c, covering a period of 7.5 hours. (MOV 1155 kb)
Supplementary Movie 3
This movie shows a time-lapse recording of the embryo developing from zygote to 2-cell. The time-lapse corresponds to the sequential pictures of Fig.2a, covering a period of 17 hours. (MOV 2650 kb)
Supplementary Movie 4
This movie shows a time-lapse recording of the embryos developing from zygote to 2-cell. The time-lapse corresponds to the sequential pictures of Fig.2b, covering a period of 18 hours. (MOV 3305 kb)
Supplementary Movie 5
This movie shows a time-lapse recording of the embryos developing from zygote to 2-cell. The time-lapse corresponds to the sequential pictures of Fig.2c, covering a period of 19 hours. (MOV 4512 kb)
Supplementary Movie 6
This movie shows a time-lapse recording of the embryos developing from zygote to 2-cell following transfer of female pronucleus into fertilized egg from which male pronucleus was removed. Female pronucleus was placed in the pole opposite from 2pb. The time-lapse corresponds to the sequential pictures of Fig.4e, covering a period of 10 hours. (MOV 1967 kb)
Supplementary Methods
Describes the methods for pronuclear transfer and immunofluorescence staining. (DOC 20 kb)
Rights and permissions
About this article
Cite this article
Hiiragi, T., Solter, D. First cleavage plane of the mouse egg is not predetermined but defined by the topology of the two apposing pronuclei. Nature 430, 360–364 (2004). https://doi.org/10.1038/nature02595
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02595
This article is cited by
-
The neglected part of early embryonic development: maternal protein degradation
Cellular and Molecular Life Sciences (2020)
-
Fertilization and Cleavage Axes Differ In Primates Conceived By Conventional (IVF) Versus Intracytoplasmic Sperm Injection (ICSI)
Scientific Reports (2019)
-
Single-cell transcriptional profiling: a window into embryonic cell-type specification
Nature Reviews Molecular Cell Biology (2018)
-
Time-lapse imaging provides further evidence that planar arrangement of blastomeres is highly abnormal
Archives of Gynecology and Obstetrics (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.