Abstract
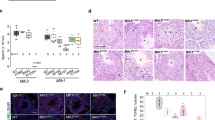
MSH5 (MutS homologue 5) is a member of a family of proteins known to be involved in DNA mismatch repair1,2. Germline mutations in MSH2, MLH1 and GTBP (also known as MSH6) cause hereditary non–polyposis colon cancer (HNPCC) or Lynch syndrome3,4,5,6,7,8. Inactivation of Msh2, Mlh1, Gtmbp (also known as Msh6) or Pms2 in mice leads to hereditary predisposition to intestinal and other cancers9,10,11,12,13,14. Early studies in yeast revealed a role for some of these proteins, including Msh5, in meiosis15,16,17. Gene targeting studies in mice confirmed roles for Mlh1 and Pms2 in mammalian meiosis12,13,14,18. To assess the role of Msh5 in mammals, we generated and characterized mice with a null mutation in Msh5. Msh5–/– mice are viable but sterile. Meiosis in these mice is affected due to the disruption of chromosome pairing in prophase I. We found that this meiotic failure leads to a diminution in testicular size and a complete loss of ovarian structures. Our results show that normal Msh5 function is essential for meiotic progression and, in females, gonadal maintenance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Modrich, P. & Lahue, R. Mismatch repair in replication fidelity, genetic recombination and cancer biology. Annu. Rev. Biochem. 65, 101–133 (1996).
Kolodner, R. Biochemistry and genetics of eukaryotic mismatch repair. Genes Dev. 10, 1433–1442 ( 1996).
Fishel, R.A. et al. The human mutator gene homolog MSH2 and its association with hereditary nonpolyposis colon cancer. Cell 75, 1027–1038 (1993).
Leach, F.S. et al. Mutations of a mutS homolog in hereditary nonpolyposis colorectal cancer. Cell 75, 1215–1225 (1993).
Bronner, C.E. et al. Mutation in the DNA mismatch repair gene homologue hMLH1 is associated with hereditary nonpolyposis colon cancer. Nature 368, 258–261 ( 1994).
Papadopoulos, N. et al. Mutation of a mutL homolog in hereditary colon cancer. Science 263, 1625–1629 (1994).
Akiyama, Y. et al. Germ–line mutation of the hMSH6/GTBP gene in an atypical hereditary nonpolyposis colorectal cancer kindred. Cancer Res. 57, 3920–3923 ( 1997).
Miyaki, M. et al. Germline mutation of MSH6 as the cause of hereditary nonpolyposis colorectal cancer. Nature Genet. 17, 271 –272 (1997).
de Wind, N., Dekker, M., Berns, A., Radman, M. & te Riele, H. Inactivation of the mouse Msh2 gene results in mismatch repair deficiency, methylation tolerance, hyperrecombination, and predisposition to cancer. Cell 82, 321– 330 (1995).
Reitmair, A.H. et al. MSH2–deficient mice are viable and susceptible to lymphoid tumours. Nature Genet. 11, 64 –70 (1995).
Edelmann, W. et al. Mutation in the mismatch repair gene Msh6 causes cancer susceptibility. Cell 91, 467– 477 (1997).
Baker, S.M. et al. Male mice defective in the DNA mismatch repair gene Pms2 exhibit abnormal chromosome synapsis in meiosis. Cell 82, 309–320 (1995).
Edelmann, W. et al. Meiotic pachytene arrest in MLH1–deficient mice. Cell 85, 1125–1134 ( 1996).
Baker, S.M. et al. Involvement of mouse Mlh1 in DNA mismatch repair and meiotic crossing over. Nature Genet. 13, 336–342 (1996).
Hollingsworth, N.M., Ponte, L. & Halsey, C. Msh5, a novel mutS homolog, facilitates meiotic reciprocal recombination between homologs in Saccharomyces cerevisiae but not mismatch repair. Genes Dev. 9, 1728– 1739 (1995).
Ross–Macdonald, P. & Roeder, G.S. Mutation of a meiosis–specific MutS homolog decreases crossing over but not mismatch correction. Cell 79, 1069– 1080 (1994).
Prolla, T.A., Christie, D.–M. & Liskay, R.M. Dual requirement in yeast DNA mismatch repair for MLH1 and PMS1, two homologs of the bacterial MutL gene. Mol. Cell. Biol. 14, 407– 415 (1994).
Plug, A.W. et al. Changes in protein composition of meiotic nodules during mammalian meiosis. J. Cell Science 111, 413– 423 (1998).
Winand, N.J., Panzer, J.A. & Kolodner, R.D. Cloning and characterization of the human and C.elegans homologs of the Saccharomyces cerevisiae Msh5 gene. Genomics 53, 69–80 ( 1998).
Enders, G.C. & May, J.J. Developmentally regulated expression of a mouse germ cell nuclear antigen examined from embryonic day 11 to adult in male and female mice. Dev. Biol. 163, 331–340 (1994).
Moens, P.B., Pearlman, R.E., Traut, W. & Heng, H.H. Chromosome cores and chromatin at meiotic prophase. Curr. Top. Dev. Biol. 37, 241–263 ( 1998).
Plug, A.W., Xu, J., Reddy, G., Golub, E.I. & Ashley, T. Presynaptic association of Rad51 protein with selected sites in meiotic chromatin. Proc. Natl Acad. Sci. USA 93, 5920–5924 (1996).
Wassarman, P.M. Zona pellucida glycoproteins. Annu. Rev. Biochem. 57 , 415–442 (1988).
Yoshida, K. et al. The mouse RecA–like gene Dmc1 is required for homologous chromosome synapsis during meiosis. Mol. Cell 1, 707–718 (1998).
Pittman, D.L. et al. Meiotic prophase arrest with failure of chromosome synapsis in mice deficient for Dmc1, a germline–specific RecA homolog. Mol. Cell 1, 697–705 (1998).
Singh, R.P. & Carr, D.H. The anatomy and histology of XO human embryos and fetuses. Anat. Rec. 155, 369 (1966).
McDonough, P.G. Gonadal dysgenesis and its variants. Pediatr. Clin. North Am. 19, 631–652 (1972).
Ioffe, E. et al. WW6: An embryonic stem cell line with an inert genetic marker that can be traced in chimeras. Proc. Natl Acad. Sci. USA 92, 7357–7361 (1995).
Counce, S.J. & Meyer, G.F. Differentiation of the synaptonemal complex and the kinetochore in Locust spermatocytes studied by whole mount electron microscopy. Chromosoma 44, 231– 253 (1973).
Spyropoulos, B. & Moens, P.B. In situ hybridization of meiotic prophase chromosomes. Methods Mol. Biol. 33, 131–139 ( 1994).
Acknowledgements
We thank the following colleagues for generously providing antibodies and advice: P. Moens, B. Spyropoulos and G. Enders. The expert technical assistance of H. Hou Jr, L. Zhu and members of the AECOM Analytical Imaging Facility (F. Maculuso, L. Gunther and C. Marks) is appreciated. We also thank S. Shenoy for training and help with the CCD imaging system. This work was supported by NIH grants (CA 76329 to W.E.; CA 67944 and NO1–CN–65031 to R.K.; CA 44704 to R.D.K.), the American Cancer Society (R.K.) and a Cancer Center grant to AECOM (CA13330).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Edelmann, W., Cohen, P., Kneitz, B. et al. Mammalian MutS homologue 5 is required for chromosome pairing in meiosis . Nat Genet 21, 123–127 (1999). https://doi.org/10.1038/5075
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/5075
This article is cited by
-
High amount of fertility reducing tumors and procedures, but no evidence for premature ovarian failure in female Lynch syndrome patients
Familial Cancer (2024)
-
SRSF1 regulates primordial follicle formation and number determination during meiotic prophase I
BMC Biology (2023)
-
Novel bi-allelic MSH4 variants causes meiotic arrest and non-obstructive azoospermia
Reproductive Biology and Endocrinology (2022)
-
Mutations of MSH5 in nonobstructive azoospermia (NOA) and rescued via in vivo gene editing
Signal Transduction and Targeted Therapy (2022)
-
PRC1-mediated epigenetic programming is required to generate the ovarian reserve
Nature Communications (2022)