Abstract
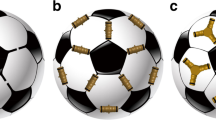
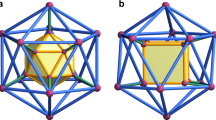
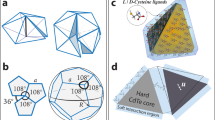
Spontaneous self-assembly processes that lead to discretespherical molecular structures are common in nature. Sphericalviruses1 (such ashepatitis B) and fullerenes2 are well-known examples inwhich non-covalent and covalent forces,respectively, direct the assembly of smaller subunits intolarger superstructures. A common feature of theseshell-like architectures is their ability to encapsulateneutral and/or charged guests whose size, shape and chemicalexteriors complement those of the host's innersurface3,4. Their interiors can often beregarded as a new phase of matter5, capable of controlling the flowof reactants, transients and products, and of catalysingreactions of both chemical and biological relevance. Suchproperties have inspired the recent emergence ofmonomolecular5,6,7 and supramolecular dimeric molecularcapsules8,9, many of which have been basedon the head-to-head alignment of bowl-shapedpolyaromatic macrocycles such as calix[4]arenes5,7,9. But true structural mimicry offrameworks akin to viruses and fullerenes, which are based onthe self-assembly of n > 3 subunits,and where surface curvature is supplied by edge sharing of regularpolygons, has remained elusive. Here we present anexample of such a system: a chiral spherical molecular assemblyheld together by 60 hydrogen bonds (1) (Fig. 1). We demonstrate the ability of 1, which consists of six calix[4]resorcinarenes 2 and eight water molecules, to self-assemble and maintain its structure in apolar media and to encapsulate guest species within a well-defined cavity that possesses an internal volume of about 1,375 Å3. Single crystal X-ray analysis shows that its topology resembles that of a spherical virus1 and conforms to the structure of a snub cube, one of the 13 Archimedean solids10.

The spheroid is held together by 60 hydrogen bonds (hydrogen bond donor/hydrogen bond acceptor/hydrogen bond type/number of hydrogen bonds): calixarene/calixarene/intramolecular/24; calixarene/calixarene/intermolecular/12; calixarene/water/intermolecular/12; water/calixarene/intermolecular/12 (24 + 12 + 12 + 12 = 60), and is commercially available (Aldrich Chemical Company).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Caspar, D. & Klug, A. Physical principles in the construction of regular viruses. Cold Spring Harb. Symp. Quant. Biol. 27, 1–24 (1962).
Kroto, H. W., Heath, J. R., O'Brien, S. C., Curl, R. F. & Smalley, R. E. C60: Buckminsterfullerene. Nature 318, 162–163 (1985).
Casjens, S. in Virus Structure and Assembly (ed. Casjens, S.) 75–147 (Jones and Bartlett, Boston, (1995)).
Schwarz, H., Weiske, T., Böhme, D. K. & Hrus˘ák, J. in Buckminsterfullerenes (eds Billups, W. E. & Ciufolini, M. A.) 257–283 (VCH, New York, (1993)).
Sherman, J. C. & Cram, D. J. Carcerand interiors provide a new phase of matter. J. Am. Chem. Soc. 111, 4527–4528 (1989).
Garel, L., Dutata, J.-P. & Collet, A. Complexation of methane and chlorofluorocarbons by cryptophane-A in organic solution. Angew. Chem. Int. Edn Engl. 32, 1169–1171 (1993).
Timmerman, P., Verboom, W., van Veggel, F. C., van Duynhoven, J. P. & Reinhoudt, D. N. Anovel type of stereoisomerism in calix[4]arene-based carceplexes. Angew. Chem. Int. Edn. Engl. 33, 2345–2348 (1994).
Kang, J. & Rebek, J. Jr. Acceleration of a Diels–Alder reaction by a self-assembled molecular capsule. Nature 385, 50–52 (1997).
Shimizu, K. D. & Rebek, J. Jr. Synthesis and assembly of self-complementary calix[4]arenes. Proc. Natl. Acad. Sci. USA 92, 12403–12407 (1995).
Wenninger, M. J. Polyhedron Models (Cambridge Univ. Press, New York, (1971)).
MacGillivray, L. R. & Atwood, J. L. Rational design of multi-component calix[4]arenes and control of their alignment in the solid state. J. Am. Chem. Soc. 119, 6931–6932 (1997).
Barbour, L. J. CAVITY, program for the determination of the volume of a molecular cavity.University of Missouri-Columbia, Missouri, USA((1997)).
Wales, D. J. & Ohmine, I. Rearrangements of model (H2O)8and (H2O)20clusters. J. Chem. Phys. 98, 7257–7268 (1993).
Aoyama, Y., Tanaka, Y. & Sugahara, S. Molecular recognition. 5. Molecular recognition of sugars via hydrogen-bonding interactions with a synthetic polyhydroxy macrocycle. J. Am. Chem. Soc. 111, 5397–5404 (1989).
Mathiowitz, E. et al. Biologically erodable microspheres as potential oral drug delivery systems. Nature 386, 410–414 (1997).
Atwood, J. L., Koutsantonis, G. A. & Raston, C. L. Purification of C60and C70by selective complexation with calixarenes. Nature 368, 229–231 (1994).
Sheldrick, G. M. Phase annealing in SHELX-90: direct methods for larger structures. Acta Crystallogr. A 46, 467–473 (1990).
Sheldrick, G. M. SHELXL93, program for crystal structure refinement, University of Göttingen, Germany((1993)).
Barbour, L. J. RES2INS, program for graphic preparation of SHELXL instruction files.University of Missouri-Columbia, Missouri, USA((1997)).
Acknowledgements
We thank L. J. Barbour for writing the program CAVITY and K. T. Holman for synthesizing 2a. This work was supported by the National Science Foundation (NSF) and the Natural Sciences and Engineering Research Council of Canada (NSERC) (research fellowship L.R.M.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
Supplementary Information (PDF 367 kb)
Rights and permissions
About this article
Cite this article
MacGillivray, L., Atwood, J. A chiral spherical molecular assembly held together by 60 hydrogen bonds. Nature 389, 469–472 (1997). https://doi.org/10.1038/38985
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/38985
This article is cited by
-
Syntheses, structures, and biological activities of two supramolecules consisting of resorcinolcalix[4]arene and amino-pyridine moieties
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2024)
-
Symmetry-breaking host–guest assembly in a hydrogen-bonded supramolecular system
Nature Communications (2023)
-
Biomimetic tail-to-head terpene cyclizations using the resorcin[4]arene capsule catalyst
Nature Protocols (2023)
-
Post-synthetic metalation of organic cage for enhanced porosity and catalytic performance
Science China Chemistry (2023)
-
Hopping protons in supramolecular catalysis
Nature Chemistry (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.