Abstract
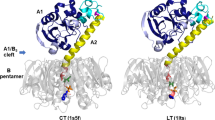
Examination of the structure of Escherichia coli heat-labile enterotoxin in the AB5 complex at a resolution of 2.3 Å reveals that the doughnut-shaped B pentamer binds the enzymatic A subunit using a hairpin of the A2 fragment, through a highly charged central pore. Putative ganglioside GM1-binding sites on the B subunits are more than 20 Å removed from the membrane-crossing Al subunit. This ADP-ribosylating (Al) fragment of the toxin has structural homology with the catalytic region of exotoxin A and hence also to diphtheria toxin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Foster, J. W. & Kinney, D. M. CRC crit. Rev. Microbiol. 11, 273–298 (1985).
Moss, J. & Vaughan, M. Adv. Enzym. 61, 303–379 (1988).
van Heyningen, S. in Current Topics in Membranes and Transport Vol. 18, 445–471 (Academic, New York, 1983).
Finkelstein, R. A. in Immunochemical and Molecular Genetic Analysis of Bacterial Pathogens (eds Owen, P. & Foster, T. J.) 85–102 (Elsevier New York, 1988).
Geary, S. J., Marchlewicz, B. A. & Finkelstein, R. A. Infect. Immunity 36, 215–220 (1982).
Dallas, W. S. & Falkow, S. Nature 288, 499–501 (1980).
Yamamoto, T., Gojobori, T. & Yokota, T. J. Bact. 169, 1352–1357 (1987).
Hirst, T. R., Sanchez, J., Kaper, J. B., Hardy, S. J. S. & Holmgren, J. Proc. natn. Acad. Sci. U.S.A. 81, 7752–7756 (1984).
Hofstra, H. & Witholt, B. J. biol. Chem. 259, 15182–15187 (1984).
Spicer, E. K. & Noble, J. A. J. biol. Chem. 257, 5716–5721 (1982).
Mekalanos, J. J., Collier, R. J. & Romig, W. R. J. biol. Chem. 254, 5855–5861 (1979).
Moss, J., Osborne, J. C. Jr. Fishman, P. H., Nakaya, S. & Robertson, D. C. J. biol. Chem. 256, 12861–12865 (1981).
Gill, D. M. Biochemistry 15, 1242–1248 (1976).
Gill, D. M. & Richardson, S. H. J. infect. Dis. 141, 64–70 (1980).
Cassel, D. & Pfeuffer, T. Proc. natn. Acad. Sci. U.S.A. 75, 2669–2673 (1978).
Moss, J. & Vaughan, M. J. biol. Chem. 252, 2455–2457 (1977).
Kahn, R. A. & Gilman, A. G. J. biol. Chem. 261, 7906–7911 (1986).
Gill, D. M. & Coburn, J. Biochemistry 26, 6364–6371 (1987).
Peterson, J. W. & Ochoa, L. G. Science 245, 857–859 (1989).
Mekalanos, J. J. et al. Nature 306, 551–557 (1983).
Clemens, J. D. et al. Lancet II, 124–127 (1986).
Schödel, F. & Will, H. Infect. Immunity 57, 1347–1350 (1989).
Sanchez, J., Svennerholm, A.-M. & Holmgren, J. FEBS Lett. 241, 110–114 (1988).
Dertzbaugh, M. T., Peterson, D. L. & Macrina, F. L. Infect. Immunity 58, 70–79 (1990).
Czerknsky, C., Russell, M. W., Lycke, N., Lindblad, M. & Holmgren, J. Infect. Immunity 57, 1072–1077 (1989).
Fujinaga, M., Gros, P. & van Gunsteren, W. F. J. appl. Crystallogr. 22, 1–8 (1989).
Read, R. J. Acta crystallogr. A42, 140–149 (1986).
Ladenstein, R. et al. J. molec. Biol. 203, 1045–1070 (1988).
Jacob, C. O., Leitner, M., Zamir, A., Salomon, D. & Arnon, R. EMBO J. 4, 3339–3343 (1985).
lida, T. et al. J. biol. Chem. 264, 14065–14070 (1989).
Kabsch, W. & Sander, C. Biopolymers 22, 2577–2637 (1983).
Janin, J., Miller, S. & Chothia, C. J. molec. Biol. 204, 155–164 (1988).
Goins, B. & Freire, E. Biochemistry 27, 2046–2052 (1988).
De Wolf, M. J. S., Fridkin, M. & Kohn, L. D. J. biol. Chem. 256, 5489–5496 (1981).
Ludwig, D. S., Holmes, R. K. & Schoolnik, G. K. J. biol. Chem. 260, 12528–12534 (1985).
Tsuji, T., Honda, T., Miwatani, T., Wakabayashi, S. & Matsubara, H. J. biol Chem. 260, 8552–8558 (1985).
Montfort, W. et al. J. biol. Chem. 262, 5398–5403 (1987).
Tomasi, M. & Montecucco, C. J. biol. Chem. 256, 11177–11181 (1981).
Wisnieski, B. J. & Bramhall, J. S. Nature 289, 319–321 (1981).
Surewicz, W. K., Leddy, J. J. & Mantsch, H. H., Biochemistry 29, 8106–8111 (1990).
Dwyer, J. D. & Bloomfield, V. A. Biochemistry 21, 3227–3231 (1982).
Ribi, H. O., Ludwig, D. S., Mercer, K. L., Schoolnik, G. K. & Kornberg, R. D. Science 239, 1272–1276 (1988).
McDaniel, R. V. & Mclntosh, T. J. Biophys. J. 49, 94–96 (1986).
Allured, V. S., Collier, R. J., Carroll, S. F. & McKay, D. B. Proc. natn. Acad. Sci. U.S.A. 83, 1320–1324 (1986).
Brandhuber, B. J., Allured, V. S., Falbel, T. G. & McKay, D. B. Proteins 3, 146–154 (1988).
Carroll, S. F. & Collier, R. J. J. biol. Chem. 262, 8707–8711 (1987).
Douglas, C. M. & Collier, R. J. J. Bact. 169, 4967–4971 (1987).
Tsuji, T. et al. J. biol. Chem. 265, 22520–22525 (1990).
Harford, S., Dykes, C. W., Hobden, A. N., Read, M. J. & Halliday, I. J. Eur. J. Biochem. 183, 311–316 (1989).
Lai, C.-Y., Xia, Q.-C. & Salotra, P. T. Biochem. biophys. Res. Commun. 116, 341–348 (1983).
Okamoto, K. et al. J. Bact. 170, 2208–2211 (1988).
Lukac, M. & Collier, R. J. Biochemistry 27, 7629–7632 (1988).
Galloway, T. S. & van Heyningen, S. Biochem. J. 244, 225–230 (1987).
Pickett, C. L., Twiddy, E. M., Coker, C. & Holmes, P. K. J. Bact. 171, 4945–4952 (1989).
Pronk, S. E. et al. J. biol. Chem. 260, 13580–13584 (1985).
Messerschmidt, A. & Pflugrath, J. W. J. appl. Crystallogr. 20, 306–315 (1987).
Kabsch, W. J. appl. Crystallogr. 21, 916–924 (1988).
Leslie, A. G. W. Acta crystallogr. A43, 134–136 (1987).
Jones, T. A. J. appl. Crystallogr. 11, 268–272 (1978).
Jones, T. A. & Thirup, S. EMBO J. 5, 819–822 (1986).
Tronrud, D. E., Ten Eyck, L. F. & Matthews, B. W. Acta crystallogr. A43, 489–501 (1987).
Sandkvist, M., Hirst, T. R. & Bagdasarian, M. J. Bact. 169, 4570–4576 (1987).
Gray, G. L. et al. Proc. natn. Acad. Sci. U.S.A. 81, 2645–2649 (1984).
Greenfield, L. et al. Proc. natn. Acad. Sci. U.s.a. 80, 6853–6857 (1983).
Ratti, G., Rappouloli, R. & Giannini, G. Nucleic Acids Res. 11, 6589–6595 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sixma, T., Pronk, S., Kalk, K. et al. Crystal structure of a cholera toxin-related heat-labile enterotoxin from E. coli. Nature 351, 371–377 (1991). https://doi.org/10.1038/351371a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/351371a0
This article is cited by
-
Genomic virulence features of Beauveria bassiana as a biocontrol agent for the mountain pine beetle population
BMC Genomics (2023)
-
Cytolethal distending toxin: from genotoxin to a potential biomarker and anti-tumor target
World Journal of Microbiology and Biotechnology (2021)
-
Improved expression of porcine epidemic diarrhea antigen by fusion with cholera toxin B subunit and chloroplast transformation in Nicotiana tabacum
Plant Cell, Tissue and Organ Culture (PCTOC) (2019)
-
Towards new cholera prophylactics and treatment: Crystal structures of bacterial enterotoxins in complex with GM1 mimics
Scientific Reports (2017)
-
1H, 13C, 15N backbone assignment of the human heat-labile enterotoxin B-pentamer and chemical shift mapping of neolactotetraose binding
Biomolecular NMR Assignments (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.