Abstract
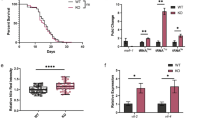
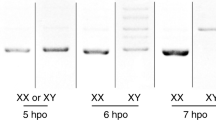
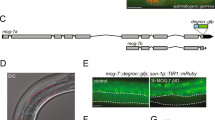
IN the Caenorhabditis elegans hermaphrodite germ line, sperm and then oocytes are made from a common pool of germ-cell precursors. The decision to differentiate as a sperm or an oocyte is regulated by the sex-determining gene,fem-3. Expression of fem-3 in the hermaphrodite germ line directs spermatogenesis and must be negatively regulated to allow the switch to oogenesis1,2. In adult hermaphrodites (which are producing oocytes), mostfem-3 RNA is found in the germ line3, consistent with both the requirement for fem-3 in hermaphrodite spermatogenesis and the maternal effects of fem-3 on embryonic sex determination1,2Whereas loss-of-function mutants in fem-3 produce only oocytes, hermaphrodites carrying any of nine fem-3 gain-of-function (gf) mutations make none; instead sperm are produced continuously and in vast excess over wild-type amounts1Genetic analyses suggest that fem-3(gf) mutations have escaped a negative control required for the switch to oogenesis1. Here we report that all nine fem-3(gf) mutants carry sequence alterations in the fem-3 3′ untranslated region (3′ UTR). There is no increase in the steady-state level of fem-3(gf) RNA over wild-type, but there is an increase in the polyadenylation of fem-3(gf) RNA that is coincident with the unregulated fem-3 sactivity. Results of a titration experiment support the hypothesis that a regulatory factor may bind the fem-3 3′ UTR. We speculate that fem-3 RNA is regulated through its 3′ UTR by binding a factor that inhibits translation, and discuss the idea that this control may be part of a more general regulation of maternal RNAs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Barton, M. K., Schedl, T. & Kimble, J. Genetics 115, 107–119 (1987).
Hodgkin, J. Genetics 114, 15–52 (1986).
Rosenquist, T. A. & Kimble, J. Genes Dev. 2, 606–616 (1988).
Wickens, M. Trends biochem. Sci. 15, 320–324 (1990).
Jackson, R. J. & Standart, N. Cell 62, 15–24 (1990).
McGrew, L. L., Dworkin-Rastl, E., Dworkin, M. B. & Richter, J. D. Genes Dev. 3, 803–815 (1989).
Vassalli, J.-D. et al. Genes Dev. 3, 2163–2171 (1989).
Braun, R. E., Peschon, J. J., Behringer, R. R., Brinster, R. L. & Palmiter, R. D. Genes Dev. 3, 793–802 (1989).
Kruys, V. et al. Proc. natn. Acad. Sci. U.S.A. 84, 6030–6034 (1987).
Kruys, V., Marinx, O., Shaw, G., Deschamps, J. & Huez, G. Science 245, 852–854 (1989).
Ch'ng, J. L. C., Shoemaked, D. L., Schimmel, P. & Holmes, E. W. Science 248, 1003–1006 (1990).
Doniach, T. Genetics 114, 53–76 (1986).
Schedl, T. & Kimble, J. Genetics 119, 43–61 (1988).
Vournakis, J. N., Efstratiadis, A. & Kafatos, F. C. Proc. natn. Acad. Sci. U.S.A. 72, 2959–2963 (1977).
Fire, A., Harrison, S. M. W. & Dixon, D. Gene 93, 189 (1990).
Fire, A. EMBO J. 5, 2673–2680 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ahringer, J., Kimble, J. Control of the sperm–oocyte switch in Caenorhabditis elegans hermaphrodites by the fem-3 3′ untranslated region. Nature 349, 346–348 (1991). https://doi.org/10.1038/349346a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/349346a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.