Abstract
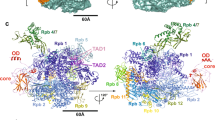
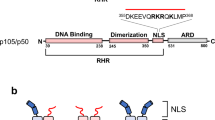
The structure of a large fragment of the p50 subunit of the human transcription factor NF-κB, bound as a homodimer to DNA, reveals that the Rel-homology region has two β-barrel domains that grip DNA in the major groove. Both domains contact the DNA backbone. The amino-terminal specificity domain contains a recognition loop that interacts with DNA bases; the car boxy-terminal dimerization domain bears the site of I-κB interaction. The folds of these domains are related to immunoglobulin-like modules. The amino-terminal domain also resembles the core domain of p53.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sen, R. & Baltimore, D. Cell 46, 705–716 (1986).
Grilli, M., Chiu, J. J.-S. & Lenardo, J. J. Int. Rev. exp. Cytol. 143, 1–62 (1993).
Baeuerle, P. A. Biochim. biophys. Acta 1072, 63–80 (1991).
Ghosh, S. et al. Cell 62, 1019–1029 (1990).
Kieran, J. et al. Cell 62, 1007–1018 (1990).
Ruben, S. M. et al. Science 251, 1490–1493 (1991).
Nolan, G. P., Ghosh, S., Liou, H. C., Tempst, P. & Baltimore, D. Cell 64, 961–969 (1991).
Steward, R. Science 238, 692–694 (1987).
Urban, M. B., Schreck, R. & Baeuerle, P. A. EMBO J. 10, 1817–1825 (1991).
Perkins, N. D. et al. Proc. natn. Acad. Sci. U.S.A. 89, 1529–1533 (1992).
Burke, P. A. et al. J. Exp. Med. 169, 1309–1321 (1989).
Israel, A. et al. EMBO J. 8, 3793–3800 (1989).
Baeuerle, P. A. & Baltimore, D. Science 242, 540–546 (1988).
Blank, V., Kourilsky, P. & Israel, A. EMBO J. 10, 4159–4167 (1991).
Beg, A. A. et al. Genes Dev. 6, 1899–1913 (1992).
Kidd, S. Cell 1, 623–635 (1992).
Fan, C.-M. & Maniatis, T. Nature 354, 395–398 (1991).
Kunsch, C., Ruben, S. M. & Rosen, C. A. Molec. cell. Biol. 12, 4412–4421 (1992).
Sodeoka, M., Larson, C. J., Chen, L., Lane, W. S. & Verdine, G. L. Biomed. Chem. Lett. 3, 1095–1100 (1993).
Baldwin, A. S. & Sharp, P. A. Molec. cell. Biol. 7, 305–313 (1987).
Ghosh, G., Van Duyne, G., Ghosh, S. & Sigler, P. B. Nature, 373, 303–310 (1995).
Cho, Y., Gorina, S., Jeffrey, P. D. & Pavletich, N. P. Science 265, 346–355 (1994).
Bork, P., Holm, L. & Sander, C. J. molec. Biol. 242, 309–320 (1994).
Juy, M. et al. Nature 357, 89–91 (1992).
Nekludova, L. & Pabo, C. O. Proc. natn. Acad. Sci. U.S.A. 91, 6948–6952 (1994).
Kumar, S., Rabson, A. B. & Gélinas, C. Molec. cell. Biol. 12, 3094–3106 (1992).
Seeman, N. C., Rosenberg, J. M. & Rich, A. Proc. natn. Acad. Sci. U.S.A. 73, 804–808 (1976).
Pavletich, N. P. & Pabo, C. O. Science 252, 809–817 (1991).
Ellenberger, T. E., Brandl, C. J., Struhl, K. & Harrison, S. C. Cell 71, 1223–1237 (1992).
Liu, J., Sodeoka, M., Lane, W. S. & Verdine, G. L. Proc. natn. Acad. Sci. U.S.A 91, 908–912 (1994).
Glover, J. N. M. & Harrison, S. C. Nature 373, 257–261 (1995).
Liu, J., Fan, Q. R., Sodeoka, M., Lane, W. S. & Verdine, G. L. Chem. Biol. 1, 47–55 (1994).
Thanos, D. & Maniatis, T. Cell 71, 777–789 (1992).
Logeat, F. et al. EMBO J. 10, 1827–1832 (1991).
Lehming, N., McGuire, S., Brickman, J. M. & Ptashne, M. EMBO J. (submitted).
Carson, M. J. appl. Crystallogr. 24, 958–961 (1991).
Epp, O., Lattman, E. E., Schiffer, M., Huber, R. & Palm, W. Biochemistry 14, 4943–4952 (1975).
Leahy, D. M., Hendrickson, W. A., Aukhel, I. & Erickson, H. P. Science 258, 987–991 (1992).
Lavery, R. & Sklenar, H. J. Biomolec. Struct. Dynam. 6, 63–91 (1988).
Kraulis, P. J. J. appl. Crystallogr. 24, 946–950 (1991).
Hendrickson, W. A., Horton, J. R. & LeMaster, D. M. EMBO J. 9, 1665–1672 (1990).
Kabsch, W. J. appl. Crystallogr. 21, 916–924 (1988).
Hendrickson, W. A., Smith, J. L., Phizackerley, R. P. & Merritt, E. A. Proteins: Structure, Function and Genetics 4, 77–88 (1988).
Ramakrishnan, V., Finch, J. T., Graziano, V., Lee, P. L. & Sweet, R. M. Nature 362, 219–223 (1993).
Bricogne, G. Acta crystallogr. A32, 832–847 (1976).
Jones, T. A., Zhou, J.-Y., Cowan, S. W. & Kjeldgaard, M. Acta crystallogr. A47, 110–119 (1991).
Brünger, A. T. X-PLOR Version 3.1.A System for X-ray Crystallography and NMR (Yale University Press, New Haven, CT, 1992).
Brünger, A. T. Nature 355, 472–475 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Müller, C., Rey, F., Sodeoka, M. et al. Structure of the NF-κB p50 homodimer bound to DNA. Nature 373, 311–317 (1995). https://doi.org/10.1038/373311a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/373311a0
This article is cited by
-
Neuroprotective effects of rutin against cuprizone-induced multiple sclerosis in mice
Inflammopharmacology (2024)
-
NF-κB signaling in neoplastic transition from epithelial to mesenchymal phenotype
Cell Communication and Signaling (2023)
-
Andrographolide-based potential anti-inflammatory transcription inhibitors against nuclear factor NF-kappa-B p50 subunit (NF-κB p50): an integrated molecular and quantum mechanical approach
3 Biotech (2023)
-
The amino acid region from 448-517 of CAMTA3 transcription factor containing a part of the TIG domain represses the N-terminal repression module function
Physiology and Molecular Biology of Plants (2023)
-
Evaluation of wound healing effect of Mallotus philippensis (Lam.) Mull. Arg. by in silico multitargets directed for multiligand approach
In Silico Pharmacology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.