Abstract
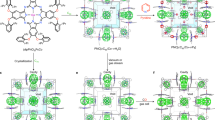
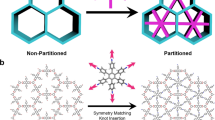
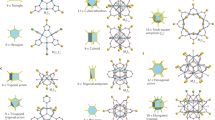
CRYSTAL engineering—the deliberate design and construction of crystal structures from molecular components—promises to provide solid-state materials with specific and useful chemical, mechanical, electronic or optical properties1. In most of the molecular crystals considered so far, van der Waals forces and hydrogen bonding govern the crystal packing2–7. Zeolites, pillared clays and related microporous materials, which have been studied extensively because their porous structures convey useful catalytic activity8,9, can now also be 'engineered' to some extent10,11. We are exploring ways12–14 to construct channelled solids with very different chemical architectures and potentially different catalytic activity from those of zeolites. Here we show that porphyrin building blocks can be used to construct three-dimensional networks with the topology of the PtS structure, containing large channels. In our materials the channels are filled with solvent molecules, and crystalline order is lost on solvent removal. Nevertheless, the results show that it is possible to use simple molecular building blocks to engineer specific frameworks which, if they can be made robust, may offer new catalytic potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bein, T. (ed.) Supramolecular Architecture 88–253 (ACS Symp. Ser. No. 499, Am. Chem. Soc., Washington DC, 1992).
Desiraju, G. R. Crystal Engineering: The Design of Organic Solids (Elsevier, Amsterdam, 1989).
Fagan, P. J. & Ward, M. D. Scient. Am. 267, 28–34 (1992).
Simard, M., Su, D. & Wuest, J. D. J. Am. chem. Soc. 113, 4696–4698 (1991).
Ermer, O. J. Am. chem. Soc. 110, 3747–3754 (1988).
MacNicol, D. D., McKendrick, J. J. & Wilson, D. R. Chem. Soc. Rev. 7, 65–87 (1978).
MacNicol, D. D. Inclusion Compounds Vol. 2, 1–45 (Academic, London, 1984).
Derouane, E. G., Lemos, F., Naccache, C. & Ribeiro, F. R. (eds) Zeolite Microporous Solids: Synthesis, Structure and Reactivity (NATO ASI Ser. C, Vol. 352, Kluwer Academic, Dordrecht, 1992).
Kerr, G. T. Scient. Am. 261, 82–87 (1989).
Estermann, M., McKusker, L. B., Baerlocher, C., Merrouche, A. & Kessler, H. Nature 352, 320–322 (1991).
Davis, M. E., Saldarriaga, C., Montes, C., Garces, J. & Crouder, C. Nature 331, 698–699 (1988).
Bein, T. (ed.) Supramolecular Architecture 256–273 (ACS Symp. Ser. No. 499, Am. Chem. Soc., Washington DC, 1992).
Hoskins, B. F. & Robson, R. J. Am. chem. Soc. 112, 1546–1554 (1990).
Gable, R. W., Hoskins, B. F. & Robson, R. J. chem. Soc., chem. Commun. 762–763 (1990).
Byrn, M. P. et al. J. Amer. chem. Soc. 115, 9480–9497 (1993).
Wohrle, D. in Phthalocyanins: Properties and Applications (eds Leznoff, C. C. & Lever, A. B. P.) 55–132 (VCH, New York, 1989).
Takemoto, K., Inaki, Y. & Ottenbrite, R. M. Functional Monomers and Polymers (Dekker, New York, 1987).
Collman, J. P. et al Proc. natn. Acad. Sci. U.S.A. 83, 4581–4585 (1986).
Gunter, M. J. et al. Inorg. Chem. 23, 283–300 (1984).
Fleischer, E. B. & Shachter, A. M. Inorg. Chem. 30, 3763–3769 (1991).
Abrahams, B. F., Hoskins, B. F. & Robson, R. J. Am. chem. Soc. 113, 3606–3607 (1991).
Sessler, J. L., Johnson, A. R., Lin, T.-Y. & Creager, S. E. J. Am. chem. Soc. 110, 3659–3661 (1988).
Walter, C. J., Anderson, H. L. & Sanders, J. K. M. J. chem. Soc. chem. Commun. 458–460 (1993).
Fujita, M., Kwon, Y. J., Washizu, S. & Ogura, K. J. Am. chem. Soc. 116, 1151–1152 (1994).
Sheldrick, G. M. in Crystallographic Computing Vol. 3 (eds Sheldrick, G. M., Kruger, C. & Goddard, R.) 175–189 (Oxford Univ. Press, 1985).
Sheldrick, G. M. SHELXS-76, a Program for Crystal Structure Determination (Univ. Cambridge, 1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abrahams, B., Hoskins, B., Michail, D. et al. Assembly of porphyrin building blocks into network structures with large channels. Nature 369, 727–729 (1994). https://doi.org/10.1038/369727a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/369727a0
This article is cited by
-
Research Progress on the Water Stability of a Metal-Organic Framework in Advanced Oxidation Processes
Water, Air, & Soil Pollution (2021)
-
Reticular chemistry in electrochemical carbon dioxide reduction
Science China Materials (2020)
-
Predominantly ligand guided non-covalently linked assemblies of inorganic complexes and guest inclusions
Journal of Chemical Sciences (2018)
-
Synthesis, molecular structure, photophysical properties and spectroscopic characterization of new 1D-magnesium(II) porphyrin-based coordination polymer
Research on Chemical Intermediates (2018)
-
NADPH:protochlorophyllide oxidoreductase B (PORB) action in Arabidopsis thaliana revisited through transgenic expression of engineered barley PORB mutant proteins
Plant Molecular Biology (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.