Abstract
The tumour suppressor p53 transcriptionally regulates a range of target genes that control cell growth and survival. Mutations of p53 have been implicated in the development of ∼50% of human cancers, including those instigated by exposure to mutagens. Although numerically rare, cancers can arise as a consequence of inherited mutations, such as in the Li–Fraumeni syndrome, which is caused by mutation of one p53 allele. Gene-targeted mice deficient for p53 have been generated to study this familial cancer syndrome. On a C57BL/6 background, p53-deficient mice develop primarily thymic lymphoma and more rarely sarcoma. Evasion of apoptosis is considered to be essential for neoplastic transformation. As proteins of the Bcl-2 family are the critical regulators of apoptosis, we investigated the role of the pro-survival members Bcl-2, Bcl-xL and Bcl-w in cancer development in p53+/− and p53−/− mice by testing whether ABT-737, a pharmacological inhibitor of these proteins, could prevent or delay tumourigenesis. Our studies showed that ABT-737 prophylaxis only caused a minor delay and reduction in γ-radiation-induced thymic lymphoma development in p53−/− mice, but this was accompanied by a concomitant increase in sarcoma. These data show that, collectively, Bcl-2, Bcl-xL and Bcl-w have only minor roles in thymic lymphoma development elicited by defects in p53, and this may indicate that Mcl-1 and/or A1 may feature more prominently in this process.
Similar content being viewed by others
Main
Li–Fraumeni syndrome (LFS) is a rare inherited autosomal disorder characterised by development of a diverse range of cancer types, including sarcomas, leukaemias/lymphomas, colon, pancreatic or breast cancers and brain or adrenocorticoid tumours before the age of 45. Srivastava et al.1 and Malkin et al.2 have linked LFS to heterozygous germline mutations within the tumour-suppressor gene p53. Obligatory loss or mutation of the wild-type (wt) p53 allele plus additional lesions in oncogenes and/or tumour-suppressor genes drives the development of cancers in these individuals. To establish a mouse model of LFS, several groups have performed gene targeting in ES cells to generate mice that either lack p533, 4 or carry point mutations in p53 that are commonly found in human cancers.5 p53 mutant mice are highly prone to develop a similar range of cancers as those found in LFS patients,3, 4, 5 although lymphomas and sarcomas predominate with their relative frequencies affected by the genetic background.4
The p53 tumour-suppressor gene encodes a transcription factor that can be activated by a broad range of stress stimuli, including DNA damage, hypoxia or activation of certain oncogenes (e.g., c-myc).6 Upon activation, p53 transcriptionally regulates various target genes that control a broad range of effector pathways, including cell-cycle arrest, DNA repair, cellular senescence and apoptosis.6 Somatically acquired mutation or loss of p53 have been implicated in the development of ∼50% of sporadic human cancers. Importantly, loss of p53 function also increases resistance of tumour cells to a broad range of anticancer therapeutics, particularly those that elicit DNA damage.6 Therefore, development of new strategies that can kill tumour cells in a p53-independent manner is an important area of research.
Apoptosis is a form of programmed cell death that is essential for normal development and tissue homeostasis in multicellular organisms.7 Evasion of apoptosis is a hallmark of cancer cells that promotes neoplastic transformation in concert with other tumourigenic processes, such as self-sufficiency for cell growth and evasion of cellular senescence.8 Apoptosis is regulated by interactions between the pro-survival and pro-apoptotic members of the Bcl-2 family and, accordingly, many cancer types show abnormalities in the balance between these two factions.9 The pro-survival Bcl-2-family members share four distinct Bcl-2 Homology domains (BH1–BH4), and experiments using gene-targeted mice have shown that these proteins have tissue-specific as well as overlapping critical roles in cell survival.9 The pro-apoptotic proteins Bax and Bak also contain four BH domains and share substantial structural similarity to their Bcl-2 pro-survival relatives, but serve critical functions in the activation of the effector stages of apoptosis by mediating mitochondrial outer membrane permeabilisation (MOMP) and consequent activation of the caspase cascade.9, 10 The BH3-only members among the Bcl-2-family members (Bim, Puma, Noxa, Bid, Bad, Bmf, Bik and Hrk) share with each other and the wider family only the 16- to 24-amino-acid BH3 domain. Studies using gene-targeted mice have shown that BH3-only proteins are essential for initiation of apoptosis signalling, functioning in a cell death stimulus-specific as well as a cell type-specific manner.9, 11
New anticancer therapeutics are being developed to specifically target the pro-survival members of the Bcl-2 family using small-molecule mimetics of BH3-only proteins.12 ABT-737 and (the orally available) ABT-263, two BH3-mimetics that bind with high affinity to Bcl-2, Bcl-xL and Bcl-w, but not to Mcl-1 or A1, have proven to be highly effective as single agents in inhibiting the growth of small-cell lung carcinoma cells in xenograft mouse models.13, 14 In addition, ABT-737 synergised potently with many conventional chemotherapeutic drugs (e.g., cyclophosphamide and taxol) and inhibitors of oncogenic kinases (e.g., Gleevec for blockade of BCR-ABL in CML) in the killing of a broad range of cancers.15, 16
Given that evasion of apoptosis, often due to deregulated expression of pro-survival Bcl-2-family members, is thought to be critical for tumour development, it is theoretically possible that BH3 mimetics may find utility as a prophylactic strategy to prevent or delay development of malignant disease in genetically predisposed individuals, such as Li–Fraumeni patients. To test this hypothesis experimentally, we explored whether blockade of Bcl-2, Bcl-xL and Bcl-w by the BH3-mimetic ABT-737 could inhibit or delay tumourigenesis in p53-deficient mice. ABT-737 prophylactic treatment had only a minor impact, although it did cause a minor delay in onset and reduction in the incidence of γ-radiation-induced thymic lymphoma in p53-deficient mice, which was accompanied by a compensatory increase in sarcoma. These results indicate that Bcl-2, Bcl-xL and Bcl-w must only have a minor role in sustaining the survival of cells undergoing neoplastic transformation owing to defects in p53.
Results
Prophylactic treatment with the BH3 mimetic ABT-737 does not prevent tumour development in p53+/− and p53−/− mice
Expression of endogenous pro-survival Bcl-2-family members is thought to be critical for the development of many cancers.17 We wanted to determine which pro-survival Bcl-2-family members are critical to sustain survival of cells undergoing neoplastic transformation and hence are essential for tumourigenesis in cancer-prone p53-deficient mice. To block all of Bcl-2, Bcl-xL and Bcl-w, we prophylactically treated p53+/− and p53−/− mice from the age of 4 weeks with ABT-737 (twice per week 50 mg/kg body weight, i.p.) or vehicle for 20 consecutive weeks and monitored the mice for tumour development. This dose of ABT-737 caused a significant decrease in platelets (P**=0.0036) as well as leukocytes (P***=0.0009) in the blood, and also resulted in a decrease in thymocytes (P**=0.0024) and splenocytes (P*=0.0321) of C57BL/6 (wt) mice (Supplementary Figure 1), indicating that this treatment regime was efficacious. Surprisingly, ABT-737 had no significant impact on the incidence or rate of tumour development in the p53+/− (P=0.0623) or p53−/− mice (P=0.5477; Figure 1a). Although vehicle-treated p53+/− mice showed a trend towards increased survival compared with ABT-737-treated p53+/− animals (median survival: vehicle=469 days versus ABT-737=312 days), this difference did not reach statistical significance (P=0.0623).
Prophylactic treatment of pre-malignant p53+/− and p53−/− mice with the BH3 mimetic ABT-737 does not prevent or even delay tumourigenesis. (a) Kaplan–Meier analysis of tumour-free survival comparing two cohorts of p53+/− mice as well as two cohorts of p53−/− mice, one prophylactically treated with the BH3 mimetic ABT-737 the other with vehicle between 4 and 24 weeks after birth (treatment period is indicated with a line). Median tumour onset of p53+/− mice prophylactically treated with ABT-737 was 312 days versus 469 days for p53+/− mice that had been treated with vehicle. This difference is not statistically significant (nABT−737=17, nVehicle=17; Mantel–Cox log–rank test: P=0.0623). Median tumour onset for p53−/− mice prophylactically treated with ABT-737 was 166 days versus 137 days for mice that had been treated with vehicle. This difference was also not statistically significant (nABT−737=9, nVehicle=16; Mantel–Cox log-rank test: P=0.5477). (b) Percentages of p53+/− mice within each cohort (ABT-737 prophylaxis or vehicle treatment) that developed the different types of malignancies (nABT−737=17, nVehicle=17). (c) Percentages of p53−/− mice within each cohort (ABT-737 prophylaxis or vehicle treatment) that developed the different types of malignancies (nABT−737=6, nVehicle=6)
Sick ABT-737 or vehicle-treated p53+/− mice succumbed mostly to either sarcoma or thymic lymphoma, and a small number developed carcinoma, such as prostate cancer (Figure 1b). There was no significant difference in lymphoma or sarcoma incidence between both treatment groups. The ABT-737-treated p53+/− mice presented with lower spleen and lymph node weights, and lower WBCs, compared with the vehicle-treated animals, but none of these differences were statistically significant (Figures 2a–c). Thymus weights were similar in both cohorts (Figure 2a). PCR genotyping of thymus samples collected from p53+/− mice that had to be killed either because of thymic lymphoma or sarcoma revealed loss of the wt p53 allele only in samples from thymic lymphoma-burdened mice but not from those that had developed sarcoma (Supplementary Figure 2). This result is consistent with previous reports that loss of heterozygosity is essential for thymic lymphoma development in p53+/− mice.3, 4 Flow-cytometric analysis using surface marker-specific antibodies (CD3, CD4 and CD8 for T cells and B220 for B cells) revealed no significant difference in T- and B-lymphoid-cell subset composition in thymi and spleens between both treatment groups.
Prophylactic treatment with ABT-737 does not alter the severity and manifestation of thymic lymphoma in p53+/− mice. (a) Thymus (P=0.2012, ±S.E.M.), spleen (P=0.3195, ±S.E.M.) and lymph node (P=0.1577, ±S.E.M.) weights from p53+/− mice that had been prophylactically treated with ABT-737 as compared with their vehicle-treated counterparts. (b) WBCs in tumour-burdened sick p53+/− mice that had been prophylactically treated with ABT-737 or vehicle (P=0.2305, ±S.E.M.). (c) Histological analysis of representative thymus, sternum and lung tissue samples of p53+/− mice treated with ABT-737 or vehicle (nABT−737=2, nVehicle=2). Tissue sections were stained with haematoxylin and eosin
Sick p53−/− mice mostly succumbed to thymic lymphoma, although in some cases this was also associated with sarcoma development and/or liver/kidney failure (Figure 1c). Mice that developed thymic lymphoma also commonly presented with infiltration of lymphoma cells into the lungs (which frequently led to breathing difficulties in the affected animals), spleen and, to a lesser extent, the liver and kidney (Figure 3c). No consistent differences in thymic lymphoma incidence (Figure 1c) and tumour dissemination (Figure 3) were observed between the two treatment groups at killing. Our analysis revealed a trend towards a decrease in thymus, spleen and lymph node weights as well as blood leukocyte counts in sick, tumour-burdened p53−/− mice that had been prophylactically treated with ABT-737 in comparison with mice that had been treated with vehicle, but none of these differences reached statistical significance (Figures 3a and b). The majority of thymi from sick animals (regardless of treatment cohort: ABT-737 or vehicle analysed by flow cytometry) revealed the presence of lymphoma cells, characterised by abnormal increase in cell size and an unusual distribution of T-cell surface markers, such as CD4LowCD8High. Collectively, these data show that prophylactic treatment with ABT-737 is unable to delay spontaneous tumour development or reduce the severity of malignant disease in p53+/− and p53−/− mice.
Prophylactic treatment with ABT-737 does not alter the severity and manifestation of thymic lymphoma in p53−/− mice. (a) Thymus (P=0.5909, ±S.E.M.), spleen (P=0.1835, ±S.E.M.) and lymph node (P=0.2286, ±S.E.M.) weights from p53−/− mice that had been treated with either ABT-737 or vehicle. (b) WBCs of sick p53−/− mice that had been prophylactically treated with ABT-737 or vehicle (P=0.5764, ±S.E.M.). (c) Histological analysis of representative thymus, sternum and lung tissue samples of p53−/− mice treated with ABT-737 or vehicle (nABT−737=2, nVehicle=2). Tissue sections were stained with haematoxylin and eosin
Prophylactic treatment with the BH3 mimetic ABT-737 has a significant, albeit minor, impact on low-dose γ-irradiation-induced thymic lymphoma development in p53−/− mice
In order to mimic the effects of environmental mutagenic factors on tumourigenesis, we examined the impact of prophylactic treatment with ABT-737 on low-dose γ-radiation-induced thymic lymphoma development in p53+/− and p53−/− mice. Repeated exposure to low-dose γ-radiation elicits thymic lymphoma development in certain strains of mice, including C57BL/6, owing to activation of the endogenous radiation-induced leukemia virus (RadLV) and generation of oncogenic mutations resulting from DNA-strand breaks.18 Loss of one or both alleles of p53 significantly accelerates γ-radiation thymic lymphoma development,19 most likely because of loss of p53's ability to coordinate cell-cycle arrest and DNA-repair responses. As expected, when compared with non-irradiated p53+/− mice, both treatment cohorts (ABT-737 prophylactic treatment or vehicle) of γ-irradiated p53+/− mice showed a considerably accelerated tumour onset (compare Figures 1a and 4a). The median survival and tumour incidence of γ-irradiated p53+/− mice did not significantly differ between animals that had been treated prophylactically with ABT-737 and vehicle-treated mice (172 versus 190 days, respectively, P=0.8992; Figures 4a and b). The majority of mice in both treatment groups presented with thymic lymphoma and these tumours routinely showed, as expected,19 loss of the wt p53 allele (Supplementary Figure 2). Analysis of thymus, spleen and lymph node weights (Figure 5a), as well as blood leukocyte counts (Figure 5b), of sick, tumour-burdened, γ-irradiated p53+/− mice revealed no significant differences between the treatment groups despite a trend towards a decrease in lymph node weights (P=0.0691; Figure 5a). Flow-cytometric analysis revealed that most lymphomas were CD8+CD4− or CD4LowCD8High in both the ABT-737 and vehicle-treated mice. Histological analysis revealed some dissemination of lymphoma cells into the lungs and occasionally the liver and kidney (Figure 5c), but the extent of these pathological features was not different between the ABT-737 and vehicle-treated animals.
Prophylactic treatment with ABT-737 slows the development of thymic lymphoma in γ-irradiated p53−/− but not p53+/− mice. (a) Kaplan–Meier analysis of tumour-free survival comparing two cohorts of γ-irradiated p53+/− as well as two cohorts of γ-irradiated p53−/− mice, one prophylactically treated between 4 and 14 weeks after birth with the BH3 mimetic ABT-737 and the other with vehicle. The arrowheads indicate the days when mice had been γ-irradiated. The time of treatment with ABT-737 or vehicle is indicated by a line. Median tumour onset for γ-irradiated p53+/− mice prophylactically treated with ABT-737 was 172 days versus 190 days for γ-irradiated p53+/− mice that had been treated with vehicle (nABT−737=17, nVehicle=21). This difference is not statistically significant (Mantel–Cox log-rank test: P=0.8992). Median tumour onset for γ-irradiated p53−/− mice prophylactically treated with ABT-737 was 134 days versus 121 days for γ-irradiated p53−/− mice treated with vehicle. This difference is statistically significant (nABT−737=12, nVehicle=11; Mantel–Cox log-rank test: P**=0.0070). (b) Percentages of γ-irradiated p53+/− mice within each cohort (ABT-737 prophylaxis or vehicle treatment) with the indicated type of malignancy (nABT−737=17, nVehicle=21). (c) Percentages of γ-irradiated p53−/− mice within each cohort (ABT-737 prophylaxis or vehicle treatment) with the indicated type of malignancy (nABT−737=12, nVehicle=11)
Prophylactic treatment with ABT-737 does not alter the severity and manifestation of thymic lymphoma in γ-irradiated p53+/− mice. (a) No significant changes were observed in thymus (P=0.7315, ±S.E.M.), spleen (P=0.9649, ±S.E.M.) and lymph node (P=0.0691, ±S.E.M.) weights in tumour-burdened sick p53+/− mice that had been γ-irradiated and prophylactically treated with ABT-737 as compared with vehicle-treated controls. (b) WBCs in sick p53+/− mice that had been γ-irradiated and prophylactically treated with ABT-737 or vehicle (P=0.7140, ±S.E.M.). (c) Histological analysis of representative thymus, sternum and lung tissue samples from γ-irradiated p53+/− (nABT−737=2, nVehicle=2). Tissue sections were stained with haematoxylin and eosin
Remarkably, prophylactic treatment with ABT-737 significantly, albeit to a relatively minor extent, prolonged the tumour-free survival of γ-irradiated p53−/− mice (median survival: 134 days versus 121 days, respectively, P**=0.0070; Figure 4a). Autopsy of sick animals from both treatment groups showed that the majority developed thymic lymphoma that was routinely associated with lymphoma cell infiltration into the lungs, and in some cases also sarcoma or splenomegaly and lymphadenopathy (Figure 6c). In addition to delaying overall tumour development, ABT-737 prophylactic treatment also caused a pronounced reduction in the incidence of thymic lymphoma and a significant reduction in thymus weight and a marked but not significant decrease in leukocytes in the peripheral blood (Figures 4c, and 6a and b). This was, however, accompanied by a compensatory increase in sarcoma in γ-irradiated p53−/− mice that had been exposed to ABT-737 (Figure 4c). No marked differences in lymphoma burden or lymphoma cell dissemination into lungs and other tissues were detected between the two cohorts (Figures 6a–c). Most of these tumours were CD8 SP or CD4LowCD8High (Supplementary Figure 3), a phenotype frequently observed in radiation-induced thymic lymphomas.20, 21 These results demonstrate that prophylactic treatment with ABT-737 can inhibit γ-radiation-induced thymic lymphoma development in p53−/− mice, albeit only to a minor extent.
Prophylactic treatment with ABT-737 has a minor impact on the severity and manifestation of thymic lymphoma in γ-irradiated p53−/− mice. (a) Significantly decreased thymus (P*=0.0378, ±S.E.M.), but not spleen (P=0.1273, ±S.E.M.) and lymph node (P=0.2104, ±S.E.M.), weights in tumour-burdened sick p53−/− mice that had been γ-irradiated and prophylactically treated with ABT-737 as compared with vehicle-treated controls. (b) WBCs of sick p53−/− mice that had been γ-irradiated and prophylactically treated with ABT-737 or vehicle (P=0.3365, ±S.E.M.). (c) Histological analysis of representative thymus, sternum and lung tissue samples of sick γ-irradiated p53−/− mice (nABT−737=2, nVehicle=2). Tissue sections were stained with haematoxylin and eosin
Thymic lymphoma cells from p53-deficient mice, regardless of ABT-737 prophylaxis or vehicle treatment, are relatively resistant to in vitro treatment with ABT-737
To explore whether thymic lymphoma cells that develop under the pressure of ABT-737 prophylactic treatment might be selected for resistance to apoptotic stimuli, we generated cell lines (CLs) from three different primary thymic lymphomas of untreated p53−/− mice as well as from γ-irradiated p53−/− that had either been treated prophylactically with ABT-737 or vehicle. These CLs and as control, the ABT-737-sensitive pre-B lymphoma-derived CL Eμ-myc/rv-bcl-2,15 were exposed to 5 μM ABT-737, 30 nM dexamethasone or vehicle (dimethyl sulphoxide (DMSO)) in culture, and their viability was monitored over 24 h (Figure 7). We observed no significant differences in the response to ABT-737 or dexamethasone between the thymic lymphoma-derived CLs from ABT-737 prophylactically treated mice and those from vehicle control-exposed animals. In comparison with the Eμ-myc/rv-bcl-2 lymphoma cells, all thymic lymphoma cells from p53-deficient mice were relatively resistant to ABT-737 (Figure 7). In agreement with a previous study,22 dexamethasone efficiently induced apoptosis of p53-deficient thymic lymphoma cells, whereas, as expected,23 the Bcl-2-overexpressing Eμ-myc/rv-bcl-2 lymphoma cells were highly resistant to this glucocorticoid. Taken together, these data show that ABT-737 prophylaxis does not alter the response of thymic lymphomas originating in p53-deficient mice to ABT-737 or dexamethasone.
Response of thymic lymphoma derived CLs from p53−/− mice that had been prophylactically treated with ABT-737 or vehicle to apoptotic stimuli in vitro. Thymic lymphoma-derived CLs from γ-irradiated p53−/− mice that had either been left untreated (n=2) or had been γ-irradiated and prophylactically treated with ABT-737 (n=3) or vehicle (n=3) and an ABT-737-sensitive control CL, Eμ-myc/rv-bcl-2 (triplicate), were treated in culture with either 5 μM ABT-737, 30 nM dexamethasone or DMSO (vehicle control) for 0, 8 or 24 h and cell viability was monitored by fluorescence-activated cell sorting (FACS) analysis
Thymic lymphomas arising in γ-irradiated p53−/− mice prophylactically treated with ABT-737, display no differences in RNA and protein expression of Bcl-2 family members when compared to those from vehicle treated control mice
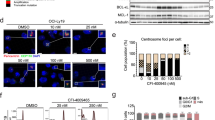
Quantitative RT-PCR and western blot analysis revealed that the levels of Bcl-2 (P=5379), Bcl-xL (P=0.3121), Mcl-1 (P=0.6667), Bcl-w (P=0.5119) and A1 (P=0.7927) were not markedly different between thymic lymphomas from the two treatment groups (Figures 8a and b). In certain tumours, however, we occasionally observed a minor increase in the protein levels of Mcl-1 and/or Bcl-xL of ABT-737- or vehicle-treated animals as compared with extracts from C57BL/6 thymi, which were used as control. In addition, there was no consistent difference between the various thymic lymphomas in the levels of the pro-apoptotic BH3-only proteins Bim, Bid or Puma. We did, however, observe a downregulation of Bim and Bid protein levels when comparing the primary tumour cells and their derivative CLs, which might explain the decreased sensitivity to ABT-737 treatment in our in vitro experiment (Figure 7). Collectively, our results show that prophylactic treatment with ABT-737 during thymic lymphoma development does not select for aberrations in the expression of the pro-survival or pro-apoptotic Bcl-2-family members or other general defects in the apoptotic machinery.
Prophylactic treatment with ABT-737 does not alter the expression of mRNA or the protein levels of Bcl-2-family members in thymic lymphomas arising in γ-irradiated p53−/− mice. (a) Analysis of the mRNA levels of the genes indicated using quantitative RT-PCR on cDNAs generated from total RNA from thymic lymphomas arising in γ-irradiated p53−/− mice that had been prophylactically treated with either ABT-737 (n=5) or vehicle (n=5). Levels of mRNAs were normalised to the mRNA levels of these genes found in thymi of healthy, untreated (i.e., non-irradiated, no ABT-737 prophylaxis) C57BL/6 (wt) mice. (b) Western blot analysis using antibodies against the indicated Bcl-2-family members and Hsp-70 (loading control) of primary thymic lymphoma cells (T) and CLs derived from these tumours that arose in γ-irradiated p53−/− mice that had been prophylactically treated with ABT-737 (n=2) or vehicle (n=2). As control we used a protein extract from thymocytes of a healthy, untreated (i.e., non-irradiated, no ABT-737 prophylactic treatment) C57BL/6 (wt, control (Ctrl)) mouse
Discussion
Small molecules that can directly activate the programmed cell death machinery are of great clinical interest for anticancer therapy.12 Much effort has been invested to develop small-molecule mimetics of the pro-apoptotic BH3-only proteins that can bind pro-survival Bcl-2-family members to elicit apoptotic death in cancer cells.13, 14 Because of their ability to directly induce apoptosis, Bcl-2-family inhibitors offer great potential for treatment of cancer, especially in treating malignancies with a dependency on Bcl-2 or Bcl-xL for their survival.12 Here, we investigated the role of Bcl-2, Bcl-xL and Bcl-w inhibition not as a modality for treatment of established malignant cancers, but as a potential prophylactic strategy to prevent tumour development in the first place.
To address this question, we used gene-targeted mice deficient for the tumour suppressor p53, a suitable model of the human LFS, although these animals are more predisposed to thymic lymphoma rather than sarcoma as seen in the human condition.3, 4 We examined whether pharmacological blockade of Bcl-2, Bcl-xL and Bcl-w using the BH3 mimetic ABT-737 in pre-malignant p53+/− and p53−/− mice could prevent or delay tumour development. In particular, given that Bcl-xL24 and Bcl-225 are both critical for cell survival during normal T lymphopoiesis, it appeared promising that their inhibition could interfere with thymic lymphoma development. Moreover, although Bcl-w is not readily detectable in thymic lymphomas elicited by p53 deficiency (Figure 8b) and is not essential for T-cell development,26 it is expressed in T lymphoid cells (including progenitors)27 and hence might contribute to the survival of nascent thymic lymphoma cells. Our results showed, however, that prophylactic treatment with ABT-737 had no significant impact on spontaneous tumour development in p53-deficient mice (Figure 1a). This indicates that endogenous expression of pro-survival Bcl-2-family members other than or in addition to Bcl-2, Bcl-xL or Bcl-w may be critical for survival of cells undergoing neoplastic transformation in the development of both thymic lymphoma and sarcoma. Mcl-1 appears to be a good candidate for this function as it is critical for survival of T-cell progenitors28 as well as stem/progenitor cells in many other lineages.29 Moreover, it is also possible that repression of pro-apoptotic Bcl-2-family members, such as Bim30 or Puma,31 maintains survival of p53-deficient cells undergoing neoplastic transformation. Finally, it remains of course possible that prophylaxis with higher doses of ABT-737, such as the (∼4–7 times higher) conventional dose of 75–100 mg/kg body weight/day commonly used for short-term studies (e.g., see Cragg et al.32), could significantly delay tumour development in p53-deficient mice. The treatment regime applied in this study was chosen because of the intent to administer ABT-737 and vehicle for a prolonged rather than short period of time; notably this dose caused a significant reduction in platelets and certain lymphocyte populations (Supplementary Figure 1), demonstrating that it was efficacious.
We also investigated whether prophylactic treatment with ABT-737 could delay tumourigenesis in p53+/− and p53−/− mice that had been exposed to repeated low-dose γ-irradiation to mimic environmental mutagenic factors that can promote neoplastic transformation, as a model for tumourigenesis provoked by environmental insults that cause DNA damage. Consistent with previous studies,19 the γ-irradiated p53−/− and p53+/− mice succumbed to tumours, almost exclusively thymic lymphomas, considerably faster than their non-irradiated counterparts. Interestingly, the γ-irradiated p53−/− mice prophylactically treated with ABT-737 survived significantly, albeit only marginally, longer (P**=0.0070) than the corresponding control mice injected with vehicle (Figure 4a). Although ABT-737 prophylaxis caused a 20% reduction in lymphoma incidence, this was accompanied by a compensatory increase in sarcoma (Figure 4c). These observations indicate that Bcl-2, Bcl-xL and Bcl-w may have a minor role in maintaining the survival of ‘cancer-initiating cells’ that give rise to γ-radiation-induced thymic lymphoma, but appear dispensable for the survival of ‘cancer-initiating cells’ giving rise to sarcomas.
Thymic lymphoma burden was frequently observed to be somewhat lower in ABT-737 prophylactically treated mice as compared to vehicle-treated animals (Figures 3, 5 and 6). This reduction may have been caused by a minor cytotoxic effect on lymphoma cells by the BH3 mimetic, particularly in mice where the final injection of ABT-737 and killing coincided because of the advanced progress of the malignant disease. Alternatively, ABT-737 might affect lymphoma growth indirectly, for example, by impairing the survival of stromal cells that are critical for lymphoma dissemination and/or survival. This latter explanation may account for the reduction in overall lymphoma burden seen in moribund mice that had received their final injection of ABT-737 a longer period of time before they were killed.
In conclusion, our studies showed that Bcl-2, Bcl-xL and Bcl-w have only a minor role in the development of thymic lymphoma and sarcoma in p53-deficient mice. We therefore reason that Mcl-1 and/or A1, the other two anti-apoptotic Bcl-2-family members, may have a more critical role in sustaining the survival of cells undergoing neoplastic transformation in this hematopoietic malignancy. This would be consistent with the finding that both Mcl-128 and A133 are critical in sustaining the survival of non-transformed T lymphoid cells at several stages of differentiation. Our observations may indicate that blockade of Mcl-1 and/or A1, rather than inhibition of Bcl-2, Bcl-xL and/or Bcl-w, is required to impede tumourigenesis in patients with germline mutations in p53. Although blockade of Bcl-2, Bcl-xL and Bcl-w by ABT-737 had only a minor impact on tumour development in p53-deficient mice, such treatment may still be efficacious in inhibiting tumourigenesis in other inherited cancer syndromes, most likely those that are associated with high levels of Bcl-2 and/or Bcl-xL expression.
Materials and Methods
Experimental animals
All experiments were conducted according to the guidelines of the Walter and Eliza Hall Institute of Medical Research Animal Ethics Committee. p53-deficient mice on a mixed C57BL/6/129SV genetic background4 were provided by Professor Tyler Jacks from the Massachusetts Institute of Technology (Cambridge, MA, USA) and were backcrossed onto a C57BL/6 background for >20 generations before commencement of these studies.
Treatment of mice with γ-radiation and/or ABT-737
Mice (p53+/− and p53−/−) at the age of 4 weeks were either left untreated or exposed to 1.5 Gy of γ-radiation once a week for four consecutive weeks using a 60Co source to elicit thymic lymphoma development.18 Between the ages of 4 and 24 weeks, nine mice of the non-irradiated cohort were injected intra-peritoneally twice a week with ABT-737 (50 mg/kg body weight) and 16 mice with vehicle. The γ-irradiated cohort was treated for the first time with ABT-737 or vehicle on the day when the first dose of γ-radiation commenced. Subsequently, ABT-737 or vehicle was applied twice a week for 10 consecutive weeks. Mice were examined daily and killed when declared unwell by the animal technicians. ABT-737 was formulated in 30% propylene glycol, 5% Tween-80 and 65% D5W (5% dextrose in water, pH 4.2).
Blood analysis
Blood was taken at the time of killing by cardiac puncture and analysed using an Advia blood analyser (Siemens, Deerfield, IL, USA). Blood parameters were plotted by using GraphPad Prism (GraphPad Software Inc., La Jolla, CA, USA).
Flow-cytometric analysis
Lymphoid organs were harvested and single-cell suspensions were prepared. Red cells were depleted using red cell lysis buffer. Cells (5 × 104) were stained for surface markers using fluorochrome-conjugated monoclonal antibodies to mouse CD4, CD8, CD3 and B220 for 30 min in balanced salt solution (BSS) supplemented with 2% fetal calf serum (FCS) and 10% normal rat serum.
Histological analysis
Soft tissues, sternum and spine were harvested and fixed in 80% Histochoice/20% ethanol and embedded in paraffin, and stained with haematoxylin and eosin and analysed by a qualified pathologist (PW). Representative images of the thymus, sternum and lung were taken at a magnification of × 25.
Genotyping
DNA samples from tail biopsies of mice or thymic lymphoma cells were analysed by PCR using primer sets that can detect wt and deleted p53 alleles. Samples were compared to tail-derived DNA from 3-week-old p53+/− and p53−/− mice. Mcl-1 primers were used as control for quality of extracted DNA.
Cell culture and cell survival assays
Thymic lymphoma cells were cultured at 37 °C in a humidified 10% CO2 incubator in Dulbecco's modified Eagle's medium supplemented with 10% FCS (Bovogen Biologicals PTY, East Keilor, Victoria, Australia), 50 μM β-mercaptoethanol (Sigma Aldrich, Castle Hill, NSW, Australia) and 100 μM asparagine (Sigma Aldrich). Cells were grown for 2 weeks in culture before being subjected to cell survival assays. For cell survival assays, 5 × 104 cells/well in a 96-well flat bottom plate were treated with either 5 μM ABT-737, 30 nM dexamethasone or DMSO (Sigma Aldrich). Cell viability was determined by flow-cytometric analysis, considering cells that are not stained with either FITC-conjugated Annexin-V (1 μg/ml) or propidium iodide (2 μg/ml) as live cells.
qRT-PCR analysis
Quantitative PCR (qRT-PCR) analysis was performed by using TaqMan probes according to the manufacturer's instructions and analysed by ABI-7900 (Applied Biosystems, Mulgrave, Victoria, Australia). Details of TaqMan gene expression assays will be provided on request.
Western blot analysis
Western blot analysis was conducted following standard protocols using extracts from primary thymic lymphomas or CLs derived from these tumours. Western blots were probed using monoclonal hamster anti-mouse Bcl-2, monoclonal mouse anti-mouse Bcl-xL (BD Pharmingen, San Diego, CA, USA), monoclonal rat anti-mouse Mcl-1, monoclonal rat anti-mouse Bcl-w, monoclonal rat anti-mouse A1, monoclonal rat anti-mouse Bid34 (last four, all gifts from Professor David Huang, The Walter and Eliza Hall Institute), polyclonal rabbit anti-mouse Bim (Stressgen, Sapphire Bioscience PTY.LTD, Waterloo, NSW, Australia) and polyclonal rabbit anti-mouse Puma (Abcam, Sapphire Bioscience PTY.LTD; Ab-27669) antibodies. Probing using monoclonal anti-mouse Hsp-70 antibody (gift from Robin Anderson, Peter MacCallum Cancer Institute, Melbourne, Australia) was used as a loading control. Monoclonal goat anti-mouse, anti-rat and anti-rabbit IgG antibodies coupled to HRP were purchased from Southern Biotech (In Vitro Technologies PTY.LTD, Noble Park, Victoria, Australia).
Statistical analysis
Kaplan–Meier mouse survival curves were generated and analysed using GraphPad Prism (GraphPad Software Inc). Mouse cohorts were compared by log-rank Mantel–Cox test. P-values less than 0.05 were considered significant.
In vitro cell survival, blood cell counts, organ weights and RNA levels were plotted and analysed using GraphPad Prism, using two-tailed Student's t-test for comparing two groups.
Abbreviations
- qRT-PCR:
-
quantitative PCR
- FACS:
-
fluorescence-activated cell sorting
- FCS:
-
fetal calf serum
- DMSO:
-
dimethyl sulphoxide
- BSS:
-
balanced salt solution
- CL:
-
cell line
- wt:
-
wild type
- Ctrl:
-
control value
- WBC:
-
white blood cell count
- Eμ-myc/rv-bcl-2:
-
Eμ-myc B lymphoma cells that had been retrovirally transduced with a Bcl-2 expression construct
References
Srivastava S, Zou ZQ, Pirollo K, Plattner W, Chang EH . Germline transmission of a mutated p53 gene in a cancer-prone family with Li–Fraumeni syndrome. Nature 1990; 348: 747–749.
Malkin D, Li FP, Strong LC, Fraumeni JFJ, Nelson CE, Kim DH et al. Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science 1990; 250: 1233–1238.
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery CAJ, Butel JS et al. Mice deficient for p53 are developmentally normal but are susceptible to spontaneous tumours. Nature 1992; 356: 215–221.
Jacks T, Remington L, Williams BO, Schmitt EM, Halachmi S, Bronson RT et al. Tumor spectrum analysis in p53-mutant mice. Curr Biol 1994; 4: 1–7.
Olive KP, Tuveson DA, Ruhe ZC, Yin B, Willis NA, Bronson RT et al. Mutant p53 gain of function in two mouse models of Li–Fraumeni syndrome. Cell 2004; 119: 847–860.
Vousden KH, Lane DP . p53 in health and disease. Nat Rev Mol Cell Biol 2007; 8: 275–283.
Hotchkiss RS, Strasser A, McDunn JE, Swanson PE . Cell death. N Engl J Med 2009; 361: 1570–1583.
Hanahan D, Weinberg RA . The hallmarks of cancer. Cell 2000; 100: 57–70.
Youle RJ, Strasser A . The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol 2008; 9: 47–59.
Green DR, Kroemer G . The pathophysiology of mitochondrial cell death. Science 2004; 305: 626–629.
Huang DCS, Strasser A . BH3-only proteins – essential initiators of apoptotic cell death. Cell 2000; 103: 839–842.
Lessene G, Czabotar PE, Colman PM . BCL-2 family antagonists for cancer therapy. Nat Rev Drug Discov 2008; 7: 989–1000.
Oltersdorf T, Elmore SW, Shoemaker AR, Armstrong RC, Augeri DJ, Belli BA et al. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature 2005; 435: 677–681.
Park CM, Bruncko M, Adickes J, Bauch J, Ding H, Kunzer A et al. Discovery of an orally bioavailable small molecule inhibitor of prosurvival B-cell lymphoma 2 proteins. J Med Chem 2008; 51: 6902–6915.
Mason KD, Vandenberg CJ, Scott CL, Wei AH, Cory S, Huang DC et al. In vivo efficacy of the Bcl-2 antagonist ABT-737 against aggressive Myc-driven lymphomas. Proc Natl Acad Sci USA 2008; 105: 17961–17966.
Cragg MS, Harris C, Strasser A, Scott CL . Unleashing the power of inhibitors of oncogenic kinases through BH3 mimetics. Nat Rev Cancer 2009; 9: 321–326.
Cory S, Huang DC, Adams JM . The Bcl-2 family: roles in cell survival and oncogenesis. Oncogene 2003; 22: 8590–8607.
Kaplan HS, Brown MB . Further observations on inhibition of lymphoid tumor development by shielding and partial-body irradiation of mice. J Natl Cancer Inst 1951; 12: 427–436.
Kemp CJ, Wheldon T, Balmain A . p53-deficient mice are extremely susceptible to radiation-induced tumorigenesis. Nat Genet 1994; 8: 66–69.
Michalak EM, Vandenberg CJ, Delbridge ARD, Wu L, Scott CL, Adams JM et al. Apoptosis-promoted tumorigenesis: gamma-irradiation-induced thymic lymphomagenesis requires Puma-driven leukocyte death. Genes Dev 2010; 24: 1608–1613.
Labi V, Erlacher M, Krumschnabel G, Manzl C, Tzankov A, Pinon J et al. Apoptosis of leukocytes triggered by acute DNA damage promotes lymphoma formation. Genes Dev 2010; 24: 1602–1607.
Strasser A, Harris AW, Jacks T, Cory S . DNA damage can induce apoptosis in proliferating lymphoid cells via p53-independent mechanisms inhibitable by Bcl-2. Cell 1994; 79: 329–339.
Strasser A, Harris AW, Cory S . Bcl-2 transgene inhibits T cell death and perturbs thymic self-censorship. Cell 1991; 67: 889–899.
Motoyama N, Kimura T, Takahashi T, Watanabe T, Nakano T . bcl-x prevents apoptotic cell death of both primitive and definitive erythrocytes at the end of maturation. J Exp Med 1999; 189: 1691–1698.
Bouillet P, Cory S, Zhang L-C, Strasser A, Adams JM . Degenerative disorders caused by Bcl-2 deficiency are prevented by loss of its BH3-only antagonist Bim. Dev Cell 2001; 1: 645–653.
Print CG, Loveland KL, Gibson L, Meehan T, Stylianou A, Wreford N et al. Apoptosis regulator Bcl-w is essential for spermatogenesis but appears otherwise redundant. Proc Natl Acad Sci USA 1998; 95: 12424–12431.
O’Reilly LA, Print C, Hausmann G, Moriishi K, Cory S, Huang DCS et al. Tissue expression and subcellular localization of the pro-survival molecule Bcl-w. Cell Death Differ 2001; 8: 486–494.
Opferman JT, Letai A, Beard C, Sorcinelli MD, Ong CC, Korsmeyer SJ . Development and maintenance of B and T lymphocytes requires antiapoptotic MCL-1. Nature 2003; 426: 671–676.
Opferman J, Iwasaki H, Ong CC, Suh H, Mizuno S, Akashi K et al. Obligate role of anti-apoptotic MCL-1 in the survival of hematopoietic stem cells. Science 2005; 307: 1101–1104.
Bouillet P, Metcalf D, Huang DCS, Tarlinton DM, Kay TWH, Köntgen F et al. Proapoptotic Bcl-2 relative Bim required for certain apoptotic responses, leukocyte homeostasis, and to preclude autoimmunity. Science 1999; 286: 1735–1738.
Villunger A, Michalak EM, Coultas L, Müllauer F, Böck G, Ausserlechner MJ et al. p53- and drug-induced apoptotic responses mediated by BH3-only proteins Puma and Noxa. Science 2003; 302: 1036–1038.
Cragg MS, Jansen ES, Cook M, Strasser A, Scott CL . Treatment of B-RAF mutant human tumor cells with a MEK inhibitor requires Bim and is enhanced by a BH3 mimetic. J Clin Invest 2008; 118: 3651–3659.
Mandal M, Borowski C, Palomero T, Ferrando AA, Oberdoerffer P, Meng F et al. The BCL2A1 gene as a pre-T cell receptor-induced regulator of thymocyte survival. J Exp Med 2005; 201: 603–614.
Kaufmann T, Tai L, Ekert PG, Huang DC, Norris F, Lindemann RK et al. The BH3-only protein bid is dispensable for DNA damage- and replicative stress-induced apoptosis or cell-cycle arrest. Cell 2007; 129: 423–433.
Acknowledgements
We thank Abbott Laboratories and Genentech Inc. for providing us with ABT-737; Dr. T Jacks for p53-deficient mice; Drs. LA O’Reilly and DC Huang, and Mr. ARD Delbridge, for advice and reagents; K McKenzie, G Siciliano, N Iannarella, L Reid and K Vella for expert animal care; B Helbert and C Young for genotyping; J Corbin for automated blood analysis; E Tsui, V Babo, K Weston, Y Hoang and S Hasanein for histology, and D Quilici, T Nikolaou and G Thomas for γ-irradiation. This work was supported by grants and fellowships from the Cancer Council of Victoria (to PNK), the National Health and Medical Research Council (Program Grant no. 461221; NHMRC Australia Fellowship), the NIH (CA43540) and the Leukemia and Lymphoma Society (SCOR Grant no. 7413), University of Melbourne International Research Scholarship, University of Melbourne International Fee Remission Scholarship, Cancer Therapeutics CRC Top-up Scholarship (last three to SG) and operational infrastructure grants through the Australian Government IRISS and the Victorian State Government OIS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by S Kaufmann
Supplementary Information accompanies the paper on Cell Death and Differentiation website
Rights and permissions
About this article
Cite this article
Grabow, S., Waring, P., Happo, L. et al. Pharmacological blockade of Bcl-2, Bcl-xL and Bcl-w by the BH3 mimetic ABT-737 has only minor impact on tumour development in p53-deficient mice. Cell Death Differ 19, 623–632 (2012). https://doi.org/10.1038/cdd.2011.133
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cdd.2011.133
Keywords
This article is cited by
-
BCL-w: apoptotic and non-apoptotic role in health and disease
Cell Death & Disease (2020)
-
BCL2 and miR-15/16: from gene discovery to treatment
Cell Death & Differentiation (2018)
-
Survival rates of homozygotic Tp53 knockout rats as a tool for preclinical assessment of cancer prevention and treatment
Cellular & Molecular Biology Letters (2017)
-
Role of miR-15/16 in CLL
Cell Death & Differentiation (2015)
-
The oncogenic properties of EWS/WT1 of desmoplastic small round cell tumors are unmasked by loss of p53 in murine embryonic fibroblasts
BMC Cancer (2013)