Abstract
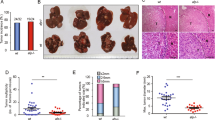
Fas-L (CD95L, APO-1L) expresses in a variety of tumours and has been proposed to play a role in tumour formation and metastasis. The contribution of Fas-L to tumour growth, however, is not conclusive especially in systems using cells with over-expressed Fas-L. In this study we down-regulated the expression o Fas-L in human glioma cells by a hammerhead ribozyme (Fas-Lribozyme) targeting against Fas-L mRNA. Fas-Lribozyme-carrying cells exhibited slightly enhanced growth rate and less degree of spontaneous apoptosis in vitro as compared with vector controls. In nude mice, Fas-Lribozyme-carrying cells grew faster with lesser apoptosis, formed bigger tumour with significantly fewer infiltrating cells in the tumour area, and triggered relatively milder tumour-associated liver damage than vector controls did. Thus, down-regulation of Fas-L not only improved viability of glioma cells but also reduces local immune responses that may consequently affect tumour formation. Taken together, our findings imply that endogenous expression of Fas-L in malignant cells is not always growth promoting. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Alderson MR, Armitage RJ, Maraskovsky EW, Tough T, Roux E, Schooley K, Ramsdell F and Lynch DH (1993) Fas transduces activation signals in normal human T lymphocytes. J Exp Med 178: 2231–2235
Alderson MR, Tough TW, Davis-Smith T, Braddy S, Falk B, Schooley KA, Goodwin RG, Smith CA, Ramsdell F and Lynch DH (1995) Fas ligand mediates activation-induced cell death in human T lymphocytes. J Exp Med 181: 71–77
Arai H, Gordon D, Nabel EG and Nabel GJ (1997) Gene transfer of Fas ligand induces tumor regression in vivo. Proc Natl Acad Sci USA 94: 13862–13867
Birikh KR, Paul A and Eckstein HF (1997) The structure, function and application of the hammerhead ribozyme. Eur J Biochem 245: 1–16
Chang MY, Won SJ and Liu HS (1997) A ribozyme specifically suppresses transformation and tumorigenicity of Ha-ras oncogene transformed NIH-3T3 cell lines. J Cancer Res Clin Onc 123: 91–99
Chattergoon MA, Kim JJ, Yang JS, Robinson TM, Lee DJ, Dentchev T, Wilson DM, Ayyavoo V and Weiner DB (2000) Targeted antigen delivery to antigen-presenting cells including dendritic cells by engineered Fas-mediated apoptosis. Nature Biotech 18: 974–979
Chen JJ, Sun Y and Nabel GJ (1998) Regulation of the proinflammatory effects of Fas ligand (CD95L). Science 282: 1714–1717
Chervonsky AV, Wang Y, Wong FS, Visintin I, Flavell RA, Janeway CA and Matis LA (1997) The role of Fas in autoimmune diabetes. Cell 89: 17–24
Cleary ML, Smith SD and Sklar J (1986) Cloning and structural analysis of cDNAs for bcl-2 and a hybrid bcl-2/immunoglobulin transcript resulting from the t(14;18) translocation. Cell 47: 19–28
Du Z, Ricordi C, Podack E and Pastori RL (1996) A hammerhead ribozyme that cleaves perforin and fas-ligand RNAs in vitro. Biochem Biophy Res Comm 226: 595–600
Gratas C, Tohma Y, Van Meir EG, Klein M, Tenan M, Ishii N, Tachibana O, Kleihues P and Ohgaki H (1997) Fas ligand expression in glioblastoma cell lines and primary astrocytic brain tumors. Brain Pathol 7: 863–869
Hahne M, Rimoldi D, Schroter M, Romero P, Schreier M, French LE, Schneider P, Bornand T, Fontana A, Lienard D, Cerottini J and Tschopp J (1996) Melanoma cell expression of Fas (Apo-I/CD95) ligand: implications for tumor immune escape. Science 274: 1363–1366
Horino K, Nishiura H, Ohsako T, Shibuya Y, Hiraoka T, Kitamura N and Yamamoto T (1998) A monocyte chemotactic factor, S19 ribosomal protein dimer, in phagocytic clearance of apoptotic cells. Lab Invest 78: 603–617
Itoh N, Yonehara S, Ishii A, Yonehara M, Mizushima S, Sameshima M, Hase A, Seto Y and Nagata S (1991) The polypeptide encoded by the cDNA for human cell surface antigen Fas can mediate apoptosis. Cell 66: 233–243
Nagata S (1997) Apoptosis by death factor. Cell 88: 355–365
Ogasawara J, Watanabe-Fukunaga R, Adachi M, Matsuzawa A, Kasugai T, Kitamura Y, Itoh N, Suda T and Nagata S (1993) Lethal effect of the anti-Fas antibody in mice. Nature 364: 806–809
Owen-Schaub LB, Radinsky R, Kruzel E, Berry K and Yonehara S (1994) Anti-Fas on nonhematopoietic tumors: levels of Fas/APO-1 and bcl-2 are not protective of biological responsiveness. Cancer Res 54: 1580–1586
Poeschla E and Wong-Staal F (1997) Antiviral and anticancer ribozymes. Cur Opin Oncol 6: 601–606
Reid S, Cross R and Snow ED (1996) Combined hoechst 33342 and merocyanine 540 staining to examine murine B cell cycle sage, viability and apoptosis. J Immunol Meth 192: 43–54
Rensing-Ehl A, Frei K, Flury R, Matiba B, Mariani SM, Weller M, Aebischer P, Krammer PH and Fontana A (1995) Local Fas/APO-1 (CD95) ligand-mediated tumor cell killing in vivo. Eur J Immunol 25: 2253–2258
Saas P, Walker PR, Hahne M, Quiquerez AL, Schnuriger V, Perrin G, French L, Van Meir EG, de Tribolet N, Tschopp J and Dietrich PY (1997) Fas ligand expression by astrocytoma in vivo: maintaining immune privilege in the brain?. J Clin Invest 99: 1173–1178
Sato T, Irie S, Kitada S and Reed J (1995) FAP-1 a protein tyrosine phosphatase that associates with Fas. Science 268: 411–415
Scanlon KJ, Jiao L, Funato T, Wang W, Tone T, Rossi JJ and Kashani-sabet M (1991) Ribozyme-mediated cleavage of c-fos mRNA reduces gene expression of DNA synthesis enzymes and metallothionein. Proc Natl Acad Sci USA 88: 10591–10595
Seino KI, Kayagaki N, Tsukada N, Fukao K, Yagita H and Okumura K (1997) Transplantation of CD95 ligand-expressing grafts. Influence of transplantation site and difficulty in protecting allo- and xenografts. Transplantation 64: 1050–1054
Shinohara H, Yagita H, Ikawa Y and Oyaizu N (2000) Fas drives cell cycle progression in glioma cells via extracellular signal-regulated kinase activation. Cancer Res 60: 1766–1772
Shinoura N, Yoshida Y, Sadata A, Hanada KI, Yamamoto S, Kirino T, Asai A and Hamada H (1998) Apoptosis by retrovirus- and adenovirus-mediated gene transfer of Fas ligand to glioma cells: implications for gene therapy. Hum Gene Ther 9: 1983–1993
Shiraki K, Tsuji N, Shiod T, Isselbacher KJ and Takahashi H (1997) Expression of Fas ligand in liver metastases of human colonic adenocarcinomas. Proc Natl Acad Sci USA 94: 6420–6425
Strand S, Hofmann WJ, Hug H, Muller M, Otto G, Strand D, Mariani SM, Stremmel W, Krammer PH and Galle PR (1996) Lymphocyte apoptosis induced by CD95 (APO-1/Fas) ligand-expressing tumor cells – A mechanism of immune evasion?. Nature Med 2: 1361–1366
Suzuki I and Fink PJ (1998) Maximal proliferation of cytotoxic T lymphocytes requires reverse signaling through Fas ligand. J Exp Med 187: 123–128
Takahashi T, Tanaka M, Inazawa J, Abe T, Suda T and Nagata S (1994) Human Fas ligand: gene structure, chromosomal location and species specificity. Int Immunol 6: 1567–1574
Van Parijs L and Abbas AK (1996) Homeostasis and self-tolerance in the immune system: turning lymphocytes off. Science 280: 243–248
Walker PR, Saas P and Dietrich PY (1998) Tumor expression of Fas ligand (CD95L) and the consequences. Curr Opin Immunol 10: 564–572
Weller M, Frei K, Groscurth P, Krammer PH, Yonekawa Y and Fontana A (1994) Anti-Fas/APO-1 antibody-mediated apoptosis of cultured human glioma cells. Induction and modulation of sensitivity by cytokines. J Clin Invest 94: 954–964
Yang BC and Yang TL (1998) Differential expression of cytokine genes and apoptosis in glioma cell lines upon exposure to bacteria and lipopolysaccharides. J Microbiol Immunol Infect 31: 95–100
Yang BC, Wang YS, Wang CH, Lin HH, Tang MJ and Yang TL (1999) Insulin-elicited transient apoptosis in serum-starved glioma cells involved Fas/Fas-L and Bcl-2. Cell Biol Intern 23: 533–540
Yang BC, Wang YS, HS Liu, and Lin SJ (2000) Ras signaling is involved in the expression of Fas-L in glioma. Lab Invest 80: 529–537
Zeytun A, Nagarkatti M and Nagarkatti PS (2000) Growth of FasL-bearing tumor cells in syngeneic murine host induces apoptosis and toxicity in Fas+ organs. Blood 95: 2111–2117
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Chio, CC., Wang, YS., Chen, YL. et al. Down-regulation of Fas-L in glioma cells by ribozyme reduces cell apoptosis, tumour-infiltrating cells, and liver damage but accelerates tumour formation in nude mice. Br J Cancer 85, 1185–1192 (2001). https://doi.org/10.1054/bjoc.2001.2055
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.2055
Keywords
This article is cited by
-
Molecular and clinical characterization of PTRF in glioma via 1,022 samples
BMC Cancer (2023)
-
Comprehensive analysis of epigenetics regulation, prognostic and the correlation with immune infiltrates of GPX7 in adult gliomas
Scientific Reports (2022)
-
The multifaceted mechanisms of malignant glioblastoma progression and clinical implications
Cancer and Metastasis Reviews (2022)
-
Phenotypic plasticity of myeloid cells in glioblastoma development, progression, and therapeutics
Oncogene (2021)
-
Enhancing antimelanoma immune responses through apoptosis
Cancer Gene Therapy (2003)