Abstract
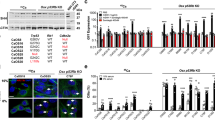
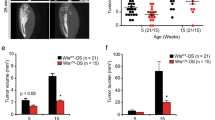
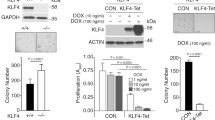
The secreted phosphoprotein osteopontin (OPN) is strongly associated with the process of neoplastic transformation, based both on its pattern of expression in vivo and in vitro and on functional analyses. We have used 3T3 cells derived from wildtype and OPN-deficient mice and transformed by transfection with oncogenic ras to assess the role of OPN in transformation in vitro and in tumorigenesis in vivo. There was no effect of an absence of OPN on the ability of the cells to undergo immortalization or to form morphologically transformed foci following ras transfection. Wildtype and OPN-deficient cell lines were established from such foci, and lines with similar ras mRNA levels selected for further analysis. Ras -transformed cell lines from both wildtype and OPN-deficient mice could form colonies in soft agar indicating that this process can occur in the absence of OPN. However, the ability of the OPN-deficient cell lines to form colonies was reduced as compared to wildtype cell lines. Tumorigenesis in syngeneic and nude mice was assessed for a subset of cell lines that formed colonies efficiently in soft agar. Cell lines unable to make OPN formed tumors in these mice much more slowly than wildtype cells, despite similar growth of the cells on plastic and in soft agar. Taken together, these results indicate that maximal transformation by ras requires OPN expression, and implicate increased OPN expression as an important effector of the transforming activity of the ras oncogene. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bayless KA, Meininger GA, Scholtz JM and Davis GE (1998) Osteopontin is a ligand for the α4β1integrin. J Cell Sci 111: 1165–1174
Behrend EI, Craig AM, Wilson SM, Denhardt DT and Chambers AF (1994) Reduced malignancy of ras -transformed NIH 3T3 cells expressing antisense osteopontin RNA. Cancer Res 54: 832–837
Bellahcène A and Castronovo V (1995) Increased expression of osteonectin and osteopontin, two bone matrix proteins, in human breast cancer. Am J Pathol 146: 95–100
Brooks PC, Clark RAF and Cheresh DA 1994 a) Requirement of vascular integrin αvβ3for angiogenesis. Science 264: 569–571
Brooks PC, Montgomery AM, Rosenfeld M, Reisfeld RA, Hu T, Klier G and Cheresh DA 1994 b) Integrin αvβ3antagonists promote 1 regression by inducing apoptosis of angiogenic blood vessels. Cell 79: 1157–1164
Brown LF, Papadopoulous-Sergiou A, Berse B, Manseau EJ, Tognazzi K, Perruzzi CA, Dvorak HF and Senger DR (1994) Osteopontin expression and distribution in human carcinomas. Am J Pathol 145: 610–623
Casson AG, Wilson SM, McCart JA, O’Malley FP, Ozcelik H, Tsao MS and Chambers AF (1997) ras mutation and expression of the ras -regulated genes osteopontin and cathepsin L in human esophageal cancer. Int J Cancer 72: 739–745
Chambers AF, Denhardt DT and Wilson SM (1990) RAS-transformed NIH3T3 cell lines, selected for metastatic ability in chick embryos, have increased proportions of p21-expressing cells and are metastatic in nude mice. Invasion Metastasis 10: 225–240
Chambers AF, Behrend EI, Wilson SM and Denhardt DT 1992 a) Induction of expression of osteopontin (OPN, secreted phosphoprotein) in metastatic, ras -transformed NIH-3T3 cells. Anticancer Res 12: 43–48
Chambers AF, Colella R, Denhardt DT and Wilson SM 1992 b) Increased expression of cathepsins L and B and decreased activity of their inhibitors in metastatic, ras -transformed NIH 3T3 cells. Mol Carcinog 5: 238–245
Chambers AF, Hota C and Prince CW (1993) Adhesion of metastatic, ras -transformed NIH 3T3 cells to osteopontin, fibronectin, and laminin. Cancer Res 53: 701–706
Chen H, Ke Y, Oates AJ, Barraclough R and Rudland PS (1997) Isolation of and effector for metastasis-inducing DNAs from a human metastatic carcinoma cell line. Oncogene 14: 1581–1588
Clezardin P (1998) Recent insights into the role of integrins in cancer metastasis. Cell Mol Life Sci 54: 541–548
Craig AM, Bowden GT, Chambers AF, Spearman MA, Greenberg AH, Wright JA, McLeod M and Denhardt DT (1990) Secreted phosphoprotein mRNA is induced during multistage carcinogenesis in skin and correlates with the metastatic potential of murine fibroblasts. Int J Cancer 46: 133–137
Crawford HC, Matrisian LM and Liaw L (1998) Distinct roles of osteopontin in host defense activity and tumour survival during squamous cell carcinoma progression in vivo. Cancer Res 58: 5206–5215
Denda S, Reichardt LF and Müller U (1998) Identification of osteopontin as a novel ligand for the integrin α8β1and potential roles for this integrin-ligand interaction in kidney morphogenesis. Mol Biol Cell 9: 1425–1435
Denhardt DT and Chambers AF (1994) Overcoming obstacles to metastasis – a defense against host defenses: Osteopontin (OPN) as a shield against attack by cytotoxic host cells. J Cell Biochem 56: 48–51
Feng B, Rollo EE and Denhardt DT (1995) Osteopontin (OPN) may facilitate metastasis by protecting cells from macrophage NO-mediated cytotoxicity: evidence from cell lines down-regulated for OPN expression. Clin Exp Metastasis 13: 453–462
Freedman VH and Shin SI (1974) Cellular tumourigenicity in nude mice: correlation with cell growth in semi-solid medium. Cell 3: 355–359
Frisch SM and Ruoslahti E (1997) Integrins and anoikis. Curr Opin Cell Biol 9: 701–706
Gardner HAR, Berse B and Senger DR (1994) Specific reduction in osteopontin synthesis by antisense RNA inhibits the tumourigenicity of transformed Rat1 fibroblasts. Oncogene 9: 2321–2326
Giachelli CM, Lombardi D, Johnson RJ, Murry CE and Almeida M (1998) Evidence for a role of osteopontin in macrophage infiltration in response to pathological stimuli in vivo. Am J Pathol 152: 353–358
Goligorsky MS, Noiri E, Kessler H and Romanov V (1997) Therapeutic potential of RGD peptides in acute renal injury. Kidney Int 51: 1487–1492
Gossen M and Bujard H (1992) Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc Natl Acad Sci USA 89: 5547–5551
Guo X, Zhang YP, Mitchell DA, Denhardt DT and Chambers AF (1995) Identification of a ras -activated enhancer in the mouse osteopontin promoter and its interaction with a putative ETS-related transcription factor whose activity correlates with the metastatic potential of the cell. Mol Cell Biol 15: 476–487
Hirota S, Ito A, Nagoshi J, Takeda M, Kurata A, Takatsuka Y, Kohri K, Nomura S and Kitamura Y (1995) Expression of bone matrix messenger ribonucleic acids in human breast cancers. Possible involvement of osteopontin in development of calcifying foci. Lab Invest 72: 64–69
Hu DD, Hoyer JR and Smith JW 1995 a) Calcium suppresses cell adhesion to osteopontin by attenuating binding affinity for integrin αvβ3 . J Biol Chem 270: 9917–9925
Hu DD, Lin ECK, Kovach NL, Hoyer JR and Smith JW 1995 b) A biochemical characterization of the binding of osteopontin to integrins αvβ1and αvβ5 . J Biol Chem 270: 26232–26238
Hwang SM, Lopez CA, Heck DE, Gardner CR, Laskin DL, Laskin JD and Denhardt DT (1994) Osteopontin inhibits induction of nitric oxide synthase gene expression by inflammatory mediators in mouse kidney epithelial cells. J Biol Chem 269: 711–715
Hynes RO (1992) Integrins: versatility, modulation, and signaling in cell adhesion. Cell 69: 11–25
Juliano RL (1994) Signal transduction by integrins and its role in the regulation of tumour growth. Cancer Metastasis Rev 13: 25–30
Katagiri YU, Sleeman J, Fujii H, Herrlich P, Hotta H, Tanaka K, Chikuma S, Yagita H, Okumura K, Murakami M, Saiki I, Chambers AF and Uede T (2000) CD-44 variants but not CD-44s cooperate with beta-1 containing integrins to permit cells to bind to osteopontin independently of arginine-glycine-aspartic acid, thereby stimulating cell motility and chemotaxis. Cancer Res 59: 219–226
Liaw L, Skinner MP, Raines EW, Ross R, Cheresh DA, Schwartz SM and Giachelli CM (1995) The adhesive and migratory effects of osteopontin are mediated via distinct cell surface integrins: role of αvβ3in smooth muscle migration to osteopontin in vitro. J Clin Invest 95: 713–724
Liaw L, Birk DE, Ballas CB, Whitsitt JS, Davidson JM and Hogan BLM (1998) Altered wound healing in mice lacking a functional osteopontin gene (spp1). J Clin Invest 101: 1468–1478
Malyankar UM, Rittling SR, Connor A and Denhardt DT (1994) The mitogen-regulated protein/proliferin transcript is degraded in primary mouse embryo fibroblast but not 3T3 nuclei: altered RNA processing correlates with immortalization. Proc Natl Acad Sci USA 91: 335–339
Montgomery AM, Reisfeld RA and Cheresh DA (1994) Integrin alpha v beta 3 rescues melanoma cells from apoptosis in three- dimensional dermal collagen. Proc Natl Acad Sci USA 91: 8856–8860
Noiri E, Dickman K, Miller F, Romanov G, Romanov V, Shaw R, Rittling SR, Denhardt DT and Goligorsky MS (2000) Acute renal ischemia in mice with a targeted disruption of the osteopontin gene: link to nitric oxide-induced injury. Kidney Int 56: 74–83
Oates AJ, Barraclough R and Rudland PS (1996) The identification of osteopontin as a metastasis-related gene product in a rodent mammary tumour model. Oncogene 13: 97–104
Renshaw MW, Ren XD and Schwartz MA (1997) Growth factor activation of MAP kinase requires cell adhesion. EMBO J 18: 5592–5599
Rittling SR (1996) Clonal nature of spontaneously immortalized 3T3 cells. Exp Cell Res 229: 7–13
Rittling SR and Novick KW (1997) Osteopontin expression in mammary gland development and tumorigenesis. Cell Growth Differ 8: 1061–1069
Rittling SR and Feng F (1998) Detection of mouse osteopontin by western blotting. Biochem Biophys Res Commun 250: 287–292
Rittling SR and Denhardt DT (2000) Osteopontin (OPN) function in pathology: lessons from OPN-deficient mice. Experimental Nephrology 7: 103–113
Rittling SR, Matsumoto HN, McKee MD, Nanci A, An XR, Novick KE, Kowalski AJ, Noda M and Denhardt DT (1998) Mice lacking osteopontin show normal development and bone structure but display altered osteoclast formation in vitro. J Bone Miner Res 13: 1101–1111
Rollo EE, Laskin DL and Denhardt DT (1996) Osteopontin inhibits nitric oxide production and cytotoxicity by activated RAW 264.7 macrophages. J Leukoc Biol 60: 397–404
Ruoslahti E (1997) Integrins as signaling molecules and targets for tumour therapy. Kidney Int 51: 1413–1417
Scatena M, Almeida M, Chaisson ML, Fausto N, Nicosia RF and Giachelli CM (1998) NF-κB mediates αvβ3integrin-induced endothelial cell survival. J Cell Biol 141: 1083–1093
Schwartz MA (1997) Integrins, oncogenes and anchorage independence. J Cell Biol 139: 575–578
Senger DR, Asch BB, Smith BD, Perruzzi CA and Dvorak HF (1983) A secreted phosphoprotein marker for neoplastic transformation of both epithelial and fibroblastic cells. Nature 302: 714–715
Senger DR, Perruzzi CA, Gracey CF, Papadopoulous A and Tenen DG (1988) Secreted phospohoproteins associated with neoplastic transformation: close homology with plasma proteins cleaved during blood coagulation. Cancer Res 48: 5770–5774
Senger DR, Perruzzi CA, Papadopoulous-Sergiou A and Van De Water L (1994) Adhesive properties of osteopontin: regulation by a naturally occurring thrombin-cleavage in close proximity to the GRGDS cell-binding domain. Mol Biol Cell 5: 565–574
Senger DR, Ledbetter SR, Claffey KP, Papadopoulous-Sergiou A, Perruzzi CA and Detmar M (1996) Stimulation of endothelial cell migration by vascular permeability factor/vascular endothelial growth factor through cooperative mechanisms involving the αvβ3integrin, osteopontin, and thrombin. Am J Pathol 149: 293–305
Singh RP, Patarca R, Schwartz J, Singh P and Cantor H (1990) Definition of a specific interaction between the early T lymphocyte activation 1 (Eta-1) protein and murine macrophages in vitro and its effect upon macrophages in vivo. J Exp Med 171: 1931–1942
Smith LL, Cheung HK, Ling LE, Chen J, Sheppard D, Pytela R and Giachelli CM (1996) Osteopontin N-terminal domain contains a cryptic adhesive sequence recognized by α9β1integrin. J Biol Chem 271: 28485–28491
Somerman MJ, Prince CW, Sauk JJ, Foster RA and Butler WT (1987) Cell attachment activity of the 44 kilodalton bone phosphoprotein is not restricted to bone cells. Matrix 9: 49–54
Strömblad S, Becker JC, Yebra M, Brooks PC and Cheresh DA (1996) Suppression of p53 activity and p21WAF1/CIP1expression by vascular cell integrin αvβ3during angiogenesis. J Clin Invest 98: 426–433
Su L, Mukherjee AB and Mukherjee BB (1995) Expression of antisense osteopontin mRNA inhibits tumour promoter-induced neoplastic transformation of mouse JB6 epidermal cells. Oncogene 10: 2163–2169
Tuck AB, O’Malley FP, Singhal H, Tonkin KS, Kerkvliet N, Saad Z, Doig GS and Chambers AF (1998) Osteopontin expression in a group of lymph node negative breast cancer patients. Int J Cancer 79: 502–508
Weber GF, Ashkar S, Glimcher MJ and Cantor H (1996) Receptor-ligand interaction between CD44 and osteopontin (ETA-1). Science 271: 509–512
Zhang Z, Vuori K, Reed JC and Ruoslahti E (1995) The α5β1integrin supports survival of cells on fibronectin and up-regulates Bcl-2 expression. Proc Natl Acad Sci USA 92: 6161–6165
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Wu, Y., Denhardt, D. & Rittling, S. Osteopontin is required for full expression of the transformed phenotype by the ras oncogene. Br J Cancer 83, 156–163 (2000). https://doi.org/10.1054/bjoc.2000.1200
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1200
Keywords
This article is cited by
-
NF-κB signaling in neoplastic transition from epithelial to mesenchymal phenotype
Cell Communication and Signaling (2023)
-
The role of osteopontin in the progression of solid organ tumour
Cell Death & Disease (2018)
-
Suppression of tumour growth by orally administered osteopontin is accompanied by alterations in tumour blood vessels
British Journal of Cancer (2014)
-
Significance of Plasma Osteopontin Levels in Patients with Bladder Urothelial Carcinomas
Molecular Diagnosis & Therapy (2012)
-
Inhibition of osteopontin reduces liver metastasis of human pancreatic cancer xenografts injected into the spleen in a mouse model
Surgery Today (2010)