Abstract
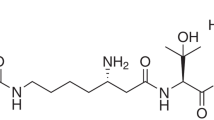
17-(5′-Isoxazolyl)androsta-4,16-dien-3-one (L-39), a novel androstene derivative, was synthesized and evaluated in vitro and in vivo. L-39 showed potent and non-competitive inhibition of human testicular microsomal 17α-hydroxylase/C17,20-lyase with an IC50 value of 59 nM and K i of 22 nM. L-39 also showed potent and competitive inhibition of 5α-reductase in human prostatic microsomes with IC50and K i values of 33 and 28 nM respectively. L-39 (5 μM) has also been shown to manifest anti-androgenic activity in cultures of human prostate cancer cell lines (LNCaP) by preventing the labelled synthetic androgen R1881 (5 nM) from binding to the androgen receptors. Androgen- dependent human próstate cancer xenografts (PC-82) were grown in nude mice and the effects of L-39 (50 mg kg–1day–1) on tumour growth and prostate-specific antigen (PSA) levels were determined after 28 days. L-39 significantly (P < 0.01) diminished tumour growth and wet weights to a similar extent as castration or flutamide treatment. L-39 also significantly (P < 0.01) reduced serum PSA levels by more than 80% in the mice bearing human prostate cancer xenografts. Pharmacokinetic studies were also conducted in male Balb/c mice. After subcutaneous administration of a single bolus dose, L-39 was rapidly absorbed into the systemic circulation. Peak plasma levels occurred at 0.75 h and then declined with a t 1/2 of 1.51 h. The bioavailability of L-39 after subcutaneous administration was 28.5%. These results demonstrate that L-39 is a potent inhibitor of androgen synthesis and is effective in reducing the growth of human prostate cancer xenografts in nude mice. Although improvements in the bioavailability are necessary, L-39 is a potential lead compound with this profile as an inhibitor of prostate cancer growth. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Angelastro MR, Laughlin ME, Schatzman GL, Bey P and Blohm TR (1989) 17α-(cyclopropylamino)-androsta-5-en-3α-ol, a selective mechanism based inhibitor of cytochrome P-45017α(steroid 17α-hydroxylase/C17,20lyase). Biochem Biophys Res Commun 162: 1571–1577
Ayub M and Levell MJ (1987) Inhibition of testicular 17α-hydroxylase and 17,20-lyase but not 3-hydroxysteroid dehydrogenase–isomerase or 17-hydroxysteroid oxidoreductase by ketoconazole and other imidazole drugs. J Steroid Biochem 28: 521–531
Bruchovsky N and Wilson JD (1968) The conversion of testosterone to 5α-androsta-17α-ol-3-one by rat prostate in vivo and in vitro. J Biol Chem 243: 2012–2021
Crawford ED, Eisenberger MA, McLeod DG, Spaulding JT, Benson R, Dorr FA, Blumenstein BA, Davies MA and Goodman PJ (1989) A controlled trial of leuprolide with and without flutamide in prostatic carcinoma. N Engl J Med 321: 419–424
Cunningham GR and Hirshkowitz M (1995) Inhibition of steroid 5α-reductase with finasteride: sleep-related erections, potency and libido in healthy men. J Clin Endocrinol Metab 80: 1934–1940
Dijkman GA and Debruyne FM (1996) Epidemiology of prostate cancer. Eur Urol 30: 281–295
Grigoryev DN, Long BJ, Nnane IP, Njar VCO, Liu Y and Brodie AMH (1999) Effects of new 17α-hydroxylase/C17,20-lyase inhibitors on LNCaP prostate cancer cell growth in vitro and in vivo. Br J Cancer 81: 622–630
Jarman M, Barrie SE, Deadman JJ, Houghton J and McCague R (1990) Novel inhibitors of enzymes of androgen biosynthesis. J Med Chem 33: 3050–3055
Klus GT, Nakamura J, Li J, Ling Y, Son C, Kemppainen JA, Wilson EM and Brodie AMH (1996) Growth inhibition of human prostate cells in vitro by novel inhibitors of androgen synthesis. Cancer Res 56: 4956–4964
Labrie F, Dupont A, Belanger A, Lacoursiere Y, Raynaud JP, Husson JM, Gareau J, Fazekas ATA, Sandow J, Monfette G, Girard JG, Emond J and Houle JG (1983) New approach in the treatment of prostate cancer: complete instead of partial withdrawal of androgens. Prostate 4: 579–594
Li J, Li Y, Son C, Banks P and Brodie A (1992) 4-Pregnen-3-one-20α-carboxyaldehyde: a potent inhibitor of 17α-hydroxylase/C17,20lyase and of 5α-reductase. J Steroid Biochem Mol Biol 42: 313–321
Li J, Li Y, Son C and Brodie AMH (1995) Inhibition of androgen synthesis by 22-hydroximino-23, 24-bisnor-4-cholen-3-one. Prostate 26: 140–150
Li JS, Li Y, Son C and Brodie A (1996) Synthesis and evaluation of pregnane derivatives as inhibitors of human testicular 17α-hydroxylase/C17,20lyase. J Med Chem 39: 4335–4339
Ling Y, Li J, Liu Y, Kato K, Klus GT and Brodie A (1997) 17-Imidazolyl, pyrazolyl and isoxazolyl androsteine derivatives. Novel steroidal inhibitors of human cytochrome C17,20-lyase (P-45017α). J Med Chem 40: 3297–3304
Long BJ, Grigoryev DN, Nnane IP, Liu Y, Ling Y, Wang X and Brodie A (1999) In vitro and in vivo inhibition of LNCaP prostate cancer cell growth by novel inhibitors of androgen synthesis. In: Proceedings of the 90th Annual Meeting of American Association for Cancer Research, Philadelphia 1999, vol. 40, p 64
Lowry OH, Roseborough NS, Farr AL and Randall RS (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193: 265–275
Nakajin S, Takahashi K and Shinoda M (1989) Inhibitory effects and interaction of stanozolol with pig testicular cytochrome P-450 (17α-hydroxylase/C17,20lyase). Chem Pharm Bull 7: 1855–1858
Njar VCO and Brodie AMH (1999) Inhibitor of 17α-hydroxylase/C17,20lyase (CYP17): potential agents for the treatment of prostate cancer. Current Pharm Design 5: 163–180
Njar VCO, Kato K, Nnane IP, Grigoryev DN, Long BJ and Brodie AMH (1988) Novel azolyl steroids; potent inhibitors of human cytochrome 17α-hydroxylase/C17,20-lyase (P-45017α): potent inhibitors for the treatment of prostate cancer. J Med Chem 41: 902–912
Nnane IP, Kato K, Liu Y, Lu Q, Wang X, Ling Y and Brodie A (1998) Effects of some novel inhibitors of C17,20-lyase and 5α-reductase in vitro and in vivo and their potential role in the treatment of prostate cancer. Cancer Res 58: 3826–3832
Nnane IP, Kato K, Liu Y, Long BJ, Lu Q, Wang X, Ling Y and Brodie A (1999) Inhibition of androgen synthesis in human testicular and prostatic microsomes and in male rats by novel steroidal compounds. Endocrinology 140: 2891–2897
Peters DH and Sorkin M (1993) Finasteride: a review of its potential in the treatment of benign prostatic hyperplasia. Drugs 46: 177–208
Roach M (1996) The role of PSA in the radiotherapy of prostate cancer. Oncology 10: 1143–1153
Rosenberg AG and von Eschenbach AC (1990) Hormonal therapy for prostate cancer. Semin Surg Oncol 6: 71–76
Saunders FJ (1963) Some aspects of relation of structure of steroids to their prostate stimulating effects. In: Prostate and Related Tissues, (National Cancer Institute monograph No 12), Vollmer EP (ed), pp. 139–159. US Government Printing Office: Washington, DC
Small EJ, Baron AD, Fippin L and Apodaca D (1997) Ketoconazole retains activity in advanced prostate cancer patients with progression despite flutamide withdrawal. J Urol 157: 204–1207
Trachtenberg J (1984) Ketoconazole therapy in advanced prostatic cancer. J Urol 132: 61–63
Wilson JD (1996) Role of dihydrotestosterone in androgen action. Prostate 6: 88–92
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Nnane, I., Long, B., Ling, YZ. et al. Anti-tumour effects and pharmacokinetic profile of 17-(5′-isoxazolyl)androsta-4,16-dien-3-one (L-39) in mice: an inhibitor of androgen synthesis. Br J Cancer 83, 74–82 (2000). https://doi.org/10.1054/bjoc.2000.1136
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1136
Keywords
This article is cited by
-
Pharmacokinetics of novel inhibitors of androgen synthesis after intravenous administration in mice
Cancer Chemotherapy and Pharmacology (2003)