Abstract
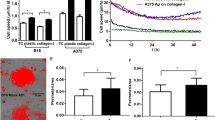
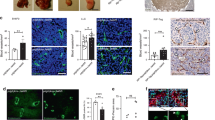
Angiogenesis of tumours might develop as a result of environmental conditions, such as hypoxia, and/or as a result of genetic alterations specific for tumour cells. The relative contributions of these mechanisms were investigated by comparing the in vivo expression of vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) to the hypoxic fraction, the angiogenic potential and the vascular density of four human melanoma lines (A-07, D-12, R-18, U-25) grown intradermally in Balb/c nu/nu mice. VEGF expression, bFGF expression and expression of pimonidazole, a marker of hypoxic cells, were investigated by immunohistochemistry. An association between high VEGF and bFGF expression and high angiogenic potential was detected, suggesting an important role for VEGF/bFGF in the angiogenesis of melanomas. High VEGF/bFGF expression was also related to low hypoxic fraction and high vascular density. Thus, the constitutive, genetically determined level of VEGF was probably more important than hypoxia-induced upregulation in the angiogenesis of the melanoma xenografts. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Chiarotto JA and Hill RP (1999) A quantitative analysis of the reduction in oxygen levels required to induce up-regulation of vascular endothelial growth factor (VEGF) mRNA in cervical cancer cell lines. Br J Cancer 80: 1518–1524
Claffey KP, Brown LF, del Aguila LF, Tognazzi K, Yeo KT, Manseau EJ and Dvorak HF (1996) Expression of vascular permeability factor/vascular endothelial growth factor by melanoma cells increases tumor growth, angiogenesis, and experimental metastasis. Cancer Res 56: 172–181
Cordon-Cardo C, Vlodavsky I, Haimovitz-Friedman A, Hicklin D and Fuks Z (1990) Expression of basic fibroblast growth factor in normal human tissues. Lab Invest 63: 832–840
D’Amore PA and Shima DT (1996) Tumor angiogenesis: a physiological process or genetically determined? Cancer Metastasis Rev 15: 205–212
Danielsen T and Rofstad EK (1998) VEGF, bFGF and EGF in the angiogenesis of human melanoma xenografts. Int J Cancer 76: 836–841
Danielsen T, Grønlund H, Hvidsten M, Smith-Sørensen B, Børresen-Dale A-L and Rofstad EK (1999) No association between radiosensitivity and TP53 status, G1arrest or protein levels of p53, myc, ras or raf in human melanoma lines. Int J Radiat Biol 75: 1149–1160
Fontanini G, Vignati S, Lucchi M, Mussi A, Calcinai A, Boldrini L, Chine S, Silvestri V, Angeletti CA, Basolo F and Bevilacqua G (1997) Neoangiogenesis and p53 protein in lung cancer: their prognostic role and their relation with vascular endothelial growth factor (VEGF) expression. Br J Cancer 75: 1295–1301
Goto F, Goto K, Weindel K and Folkman J (1993) Synergistic effects of vascular endothelial growth factor and basic fibroblast growth factor on the proliferation and cord formation of bovine capillary endothelial cells within collagen gels. Lab Invest 69: 508–517
Guidi AJ, Abu-Jawdeh G, Berse B, Jackman RW, Tognazzi K, Dvorak HF and Brown LF (1995) Vascular permeability factor (vascular endothelial growth factor) expression and angiogenesis in cervical neoplasia. J Natl Cancer Inst 87: 1237–1245
Hlatky L, Tsionou C, Hahnfeldt P and Coleman CN (1994) Mammary fibroblasts may influence breast tumor angiogenesis via hypoxia-induced vascular endothelial growth factor up-regulation and protein expression. Cancer Res 54: 6083–6086
Kim KJ, Li B, Winer J, Armanini M, Gillett N, Phillips HS and Ferrara N (1993) Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 362: 841–844
Lyng H, Sundfør K and Rofstad EK (1997) Oxygen tension in human tumours measured with polarographic needle electrodes and its relationship to vascular density, necrosis and hypoxia. Radiother Oncol 44: 163–169
Mattern J, Kallinowski F, Herfarth C and Volm M (1996) Association of resistance-related protein expression with poor vascularization and low levels of oxygen in human rectal cancer. Int J Cancer 67: 20–23
Mazure NM, Chen EY, Yeh P, Laderoute KR and Giaccia AJ (1996) Oncogenic transformation and hypoxia synergistically act to modulate vascular endothelial growth factor expression. Cancer Res 56: 3436–3440
Mukhopadhyay D, Tsiokas L and Sukhatme VP (1995) Wild-type p53 and v-Src exert opposing influences on human vascular endothelial growth factor gene expression. Cancer Res 55: 6161–6165
Obermair A, Kucera E, Mayerhofer K, Speiser P, Seifert M, Czerwenka K, Kaider A, Leodolter S, Kainz C and Zeillinger R (1997) Vascular endothelial growth factor (VEGF) in human breast cancer: correlation with disease-free survival. Int J Cancer 74: 455–458
Pötgens AJ, Lubsen NH, van Altena MC, Schoenmakers JG, Ruiter DJ and de Waal RM (1995) Vascular permeability factor expression influences tumor angiogenesis in human melanoma lines xenografted to nude mice. Am J Pathol 146: 197–209
Raleigh JA, Calkins-Adams DP, Rinker LH, Ballenger CA, Weissler MC, Fowler WC, Novotny DB and Varia MA (1998) Hypoxia and vascular endothelial growth factor expression in human squamous cell carcinomas using pimonidazole as a hypoxia marker. Cancer Res 58: 3765–3768
Rofstad EK (1994) Orthotopic human melanoma xenograft model systems for studies of tumour angiogenesis, pathophysiology, treatment sensitivity and metastatic pattern. Br J Cancer 70: 804–812
Rofstad EK and Danielsen T (1998) Hypoxia-induced angiogenesis and vascular endothelial growth factor secretion in human melanoma. Br J Cancer 77: 897–902
Rofstad EK and Danielsen T (1999) Hypoxia-induced metastasis of human melanoma cells: involvement of vascular endothelial growth factor-mediated angiogenesis. Br J Cancer 80: 1697–1707
Rofstad EK and Måseide K (1998) Fraction of radiobiologically hypoxic cells in human melanoma xenografts measured by using single-cell survival, tumour growth delay and local tumour control as end points. Br J Cancer 78: 893–898
Seghezzi G, Patel S, Ren CJ, Gualandris A, Pintucci G, Robbins ES, Shapiro RL, Galloway AC, Rifkin DB and Mignatti P (1998) Fibroblast growth factor-2 (FGF-2) induces vascular endothelial growth factor (VEGF) expression in the endothelial cells of forming capillaries: an autocrine mechanism contributing to angiogenesis. J Cell Biol 141: 1659–1673
Shweiki D, Itin A, Soffer D and Keshet E (1992) Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 359: 843–845
Shweiki D, Neeman M, Itin A and Keshet E (1995) Induction of vascular endothelial growth factor expression by hypoxia and by glucose deficiency in multicell spheroids: implications for tumor angiogenesis. Proc Natl Acad Sci USA 92: 768–772
Takahashi Y, Kitadai Y, Bucana CD, Cleary KR and Ellis LM (1995) Expression of vascular endothelial growth factor and its receptor, KDR, correlates with vascularity, metastasis, and proliferation of human colon cancer. Cancer Res 55: 3964–3968
Takahashi Y, Bucana CD, Cleary KR and Ellis LM (1998) p53, vessel count, and vascular endothelial growth factor expression in human colon cancer. Int J Cancer 79: 34–38
Tufto I, Lyng H and Rofstad EK (1998) Vascular density in human melanoma xenografts: relationship to angiogenesis, perfusion and necrosis. Cancer Lett 123: 159–165
Westphal JR, van’t Hullenaar RG, van der Laak JA, Cornelissen IM, Schalkwijk LJ, van Muijen GN, Wesseling P, de Wilde PC, Ruiter DJ and de Waal RM (1997) Vascular density in melanoma xenografts correlates with vascular permeability factor expression but not with metastatic potential. Br J Cancer 76: 561–570
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Danielsen, T., Rofstad, E. The constitutive level of vascular endothelial growth factor (VEGF) is more important than hypoxia-induced VEGF up-regulation in the angiogenesis of human melanoma xenografts. Br J Cancer 82, 1528–1534 (2000). https://doi.org/10.1054/bjoc.2000.1173
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1173
Keywords
This article is cited by
-
Hypoxia-associated spontaneous pulmonary metastasis in human melanoma xenografts: involvement of microvascular hot spots induced in hypoxic foci by interleukin 8
British Journal of Cancer (2002)
-
Influence of vascular endothelial growth factor single nucleotide polymorphisms on tumour development in cutaneous malignant melanoma
Genes & Immunity (2002)
-
Angiogenesis and melanoma
Current Oncology Reports (2001)