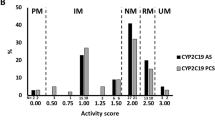
Abstract
Psychiatric pharmacotherapy with olanzapine is commonplace. We investigated the influence of CYP1A2*1F (−163A, rs762551) and serotonergic polymorphisms on olanzapine serum concentrations and clinical outcome in a naturalistic clinical setting. Included were 124 Caucasian psychiatric inpatients treated with olanzapine for at least 4 weeks with steady-state serum concentrations available for 73 patients. The CYP1A2*1F polymorphism was reported to affect the inducibility of CYP1A2. In our study population, CYP1A2*1F/*1F genotype alone resulted in a 22% reduction of dose-/body weight-normalized olanzapine serum concentrations compared to homo- and heterozygote carriers of CYP1A2*1A (both groups without inducers). This effect was independent of the well-known effect of inducing agents (here tobacco smoke and carbamazepine which led to on average 28% lower concentrations in CYP1A2*1A carriers and 26% lower concentrations in CYP1A2*1F/*1F carriers). Consistently, patients with the CYP1A2*1F/*1F genotype taking inducers had 22% lower concentrations compared to CYP1A2*1A carriers taking inducers. The influence of genotype alone remained significant after Bonferroni's post hoc test. Higher olanzapine concentrations were significantly correlated with better improvement of paranoid and depressive symptoms in patients with schizophrenic disorders (Spearman's r=0.5, P=0.026 and P=0.006, respectively). No relationship between serum concentrations and the side effects (DOTES) score was detected. However, patients with the 5-HTR2A intron 2 (rs7997012) AA genotype suffered from more pronounced side effects compared to carriers of the GA or GG genotype (P=0.018 and P=0.002). Short-term weight gain under olanzapine therapy was significantly lower for 5-HTR2C −759 T-allele carriers (P=0.011). Our data suggest that the CYP1A2*1F/*1F genotype exhibits a significant influence on olanzapine concentrations independent of other inducing factors. Thus, CYP1A2*1F genotyping may be useful for clinical treatment decisions given the fact that olanzapine serum concentrations correlated with treatment response. Side effects and weight gain, however, seem to be more influenced by serotonergic polymorphisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mauri MC, Volonteri LS, Colasanti A, Fiorentini A, De Gaspari IF, Bareggi SR . Clinical pharmacokinetics of atypical antipsychotics: a critical review of the relationship between plasma concentrations and clinical response. Clin Pharmacokinet 2007; 46: 359–388.
Bymaster F, Perry KW, Nelson DL, Wong DT, Rasmussen K, Moore NA et al. Olanzapine: a basic science update. Br J Psychiatry Suppl 1999; 37: 36–40.
Meltzer HY . The role of serotonin in antipsychotic drug action. Neuropsychopharmacology 1999; 21: 106S–115S.
Lane HY, Lee CC, Liu YC, Chang WH . Pharmacogenetic studies of response to risperidone and other newer atypical antipsychotics. Pharmacogenomics 2005; 6: 139–149.
Wilffert B, Zaal R, Brouwers JR . Pharmacogenetics as a tool in the therapy of schizophrenia. Pharm World Sci 2005; 27: 20–30.
Arranz MJ, Munro J, Sham P, Kirov G, Murray RM, Collier DA et al. Meta-analysis of studies on genetic variation in 5-HT2A receptors and clozapine response. Schizophr Res 1998; 32: 93–99.
Muller M . Pharmacogenomics and drug response. Int J Clin Pharmacol Ther 2003; 41: 231–240.
McMahon FJ, Buervenich S, Charney D, Lipsky R, Rush AJ, Wilson AF et al. Variation in the gene encoding the serotonin 2A receptor is associated with outcome of antidepressant treatment. Am J Hum Genet 2006; 78: 804–814.
Wang YC, Bai YM, Chen JY, Lin CC, Lai IC, Liou YJ . Polymorphism of the adrenergic receptor alpha 2a -1291C>G genetic variation and clozapine-induced weight gain. J Neural Transm 2005; 112: 1463–1468.
Muller DJ, Kennedy JL . Genetics of antipsychotic treatment emergent weight gain in schizophrenia. Pharmacogenomics 2006; 7: 863–887.
Bishop JR, Ellingrod VL, Moline J, Miller D . Pilot study of the G-protein beta3 subunit gene (C825T) polymorphism and clinical response to olanzapine or olanzapine-related weight gain in persons with schizophrenia. Med Sci Monit 2006; 12: BR47–BR50.
Park YM, Chung YC, Lee SH, Lee KJ, Kim H, Byun YC et al. Weight gain associated with the alpha2a-adrenergic receptor -1,291 C/G polymorphism and olanzapine treatment. Am J Med Genet B Neuropsychiatr Genet 2006; 141: 394–397.
Wang YC, Bai YM, Chen JY, Lin CC, Lai IC, Liou YJ . C825T polymorphism in the human G protein beta3 subunit gene is associated with long-term clozapine treatment-induced body weight change in the Chinese population. Pharmacogenet Genomics 2005; 15: 743–748.
Templeman LA, Reynolds GP, Arranz B, San L . Polymorphisms of the 5-HT2C receptor and leptin genes are associated with antipsychotic drug-induced weight gain in Caucasian subjects with a first-episode psychosis. Pharmacogenet Genomics 2005; 15: 195–200.
Prior TI, Baker GB . Interactions between the cytochrome P450 system and the second-generation antipsychotics. J Psychiatry Neurosci 2003; 28: 99–112.
Callaghan JT, Bergstrom RF, Ptak LR, Beasley CM . Olanzapine. Pharmacokinetic and pharmacodynamic profile. Clin Pharmacokinet 1999; 37: 177–193.
Hartter S, Korhonen T, Lundgren S, Rane A, Tolonen A, Turpeinen M et al. Effect of caffeine intake 12 or 24 h prior to melatonin intake and CYP1A2*1F polymorphism on CYP1A2 phenotyping by melatonin. Basic Clin Pharmacol Toxicol 2006; 99: 300–304.
Sachse C, Brockmoller J, Bauer S, Roots I . Functional significance of a C → A polymorphism in intron 1 of the cytochrome P450 CYP1A2 gene tested with caffeine. Br J Clin Pharmacol 1999; 47: 445–449.
Ghotbi R, Christensen M, Roh HK, Ingelman-Sundberg M, Aklillu E, Bertilsson L . Comparisons of CYP1A2 genetic polymorphisms, enzyme activity and the genotype-phenotype relationship in Swedes and Koreans. Eur J Clin Pharmacol 2007; 63: 537–546.
Flockhart DA . Drug Interactions: Cytochrome P450 Drug Interaction Table (Version 4.0). [cited 2009; Available from: http://medicine.iupui.edu/flockhart/table.htm ].
Anttila SA, Leinonen EV . A review of the pharmacological and clinical profile of mirtazapine. CNS Drug Rev 2001; 7: 249–264.
Murphy Jr GM, Kremer C, Rodrigues HE, Schatzberg AF . Pharmacogenetics of antidepressant medication intolerance. Am J Psychiatry 2003; 160: 1830–1835.
Drieling T, Biedermann NC, Scharer LO, Strobl N, Langosch JM . [Psychotropic drug-induced change of weight: a review]. Fortschr Neurol Psychiatr 2007; 75: 65–80.
Theisen FM, Haberhausen M, Schulz E, Fleischhaker C, Clement HW, Heinzel-Gutenbrunner M et al. Serum levels of olanzapine and its N-desmethyl and 2-hydroxymethyl metabolites in child and adolescent psychiatric disorders: effects of dose, diagnosis, age, sex, smoking, and comedication. Ther Drug Monit 2006; 28: 750–759.
Weiss U, Marksteiner J, Kemmler G, Saria A, Aichhorn W . Effects of age and sex on olanzapine plasma concentrations. J Clin Psychopharmacol 2005; 25: 570–574.
Carrillo JA, Herraiz AG, Ramos SI, Gervasini G, Vizcaino S, Benitez J . Role of the smoking-induced cytochrome P450 (CYP)1A2 and polymorphic CYP2D6 in steady-state concentration of olanzapine. J Clin Psychopharmacol 2003; 23: 119–127.
Skogh E, Reis M, Dahl ML, Lundmark J, Bengtsson F . Therapeutic drug monitoring data on olanzapine and its N-demethyl metabolite in the naturalistic clinical setting. Ther Drug Monit 2002; 24: 518–526.
Gex-Fabry M, Balant-Gorgia AE, Balant LP . Therapeutic drug monitoring of olanzapine: the combined effect of age, gender, smoking, and comedication. Ther Drug Monit 2003; 25: 46–53.
Bergemann N, Frick A, Parzer P, Kopitz J . Olanzapine plasma concentration, average daily dose, and interaction with co-medication in schizophrenic patients. Pharmacopsychiatry 2004; 37: 63–68.
Olesen OV, Linnet K . Olanzapine serum concentrations in psychiatric patients given standard doses: the influence of comedication. Ther Drug Monit 1999; 21: 87–90.
Linnet K, Olesen OV . Free and glucuronidated olanzapine serum concentrations in psychiatric patients: influence of carbamazepine comedication. Ther Drug Monit 2002; 24: 512–517.
Lucas RA, Gilfillan DJ, Bergstrom RF . A pharmacokinetic interaction between carbamazepine and olanzapine: observations on possible mechanism. Eur J Clin Pharmacol 1998; 54: 639–643.
Aitchison KJ, Gonzalez FJ, Quattrochi LC, Sapone A, Zhao JH, Zaher H et al. Identification of novel polymorphisms in the 5′ flanking region of CYP1A2, characterization of interethnic variability, and investigation of their functional significance. Pharmacogenetics 2000; 10: 695–704.
Han XM, Ouyang DS, Chen XP, Shu Y, Jiang CH, Tan ZR et al. Inducibility of CYP1A2 by omeprazole in vivo related to the genetic polymorphism of CYP1A2. Br J Clin Pharmacol 2002; 54: 540–543.
Nozawa M, Ohnuma T, Matsubara Y, Sakai Y, Hatano T, Hanzawa R et al. The relationship between the response of clinical symptoms and plasma olanzapine concentration, based on pharmacogenetics: Juntendo University Schizophrenia Projects (JUSP). Ther Drug Monit 2008; 30: 35–40.
Shirley KL, Hon YY, Penzak SR, Lam YW, Spratlin V, Jann MW . Correlation of cytochrome P450 (CYP) 1A2 activity using caffeine phenotyping and olanzapine disposition in healthy volunteers. Neuropsychopharmacology 2003; 28: 961–966.
Kootstra-Ros JE, Smallegoor W, van der Weide J . The cytochrome P450 CYP1A2 genetic polymorphisms *1F and *1D do not affect clozapine clearance in a group of schizophrenic patients. Ann Clin Biochem 2005; 42: 216–219.
Basile VS, Ozdemir V, Masellis M, Walker ML, Meltzer HY, Lieberman JA et al. A functional polymorphism of the cytochrome P450 1A2 (CYP1A2) gene: association with tardive dyskinesia in schizophrenia. Mol Psychiatry 2000; 5: 410–417.
Murray M . Altered CYP expression and function in response to dietary factors: potential roles in disease pathogenesis. Curr Drug Metab 2006; 7: 67–81.
Murray M . Role of CYP pharmacogenetics and drug-drug interactions in the efficacy and safety of atypical and other antipsychotic agents. J Pharm Pharmacol 2006; 58: 871–885.
Masellis M, Basile V, Meltzer HY, Lieberman JA, Sevy S, Macciardi FM et al. Serotonin subtype 2 receptor genes and clinical response to clozapine in schizophrenia patients. Neuropsychopharmacology 1998; 19: 123–132.
Anttila S, Kampman O, Illi A, Rontu R, Lehtimaki T, Leinonen E . Association between 5-HT2A, TPH1 and GNB3 genotypes and response to typical neuroleptics: a serotonergic approach. BMC Psychiatry 2007; 7: 22.
Allison DB, Mentore JL, Heo M, Chandler LP, Cappelleri JC, Infante MC et al. Antipsychotic-induced weight gain: a comprehensive research synthesis. Am J Psychiatry 1999; 156: 1686–1696.
Allison DB, Casey DE . Antipsychotic-induced weight gain: a review of the literature. J Clin Psychiatry 2001; 62 (Suppl 7): 22–31.
Muller DJ, Muglia P, Fortune T, Kennedy JL . Pharmacogenetics of antipsychotic-induced weight gain. Pharmacol Res 2004; 49: 309–329.
Popp J, Leucht S, Heres S, Steimer W . DRD4 48 bp VNTR but not 5-HT 2C Cys23Ser receptor polymorphism is related to antipsychotic-induced weight gain. Pharmacogenomics J 2009; 9: 71–77.
Ryu S, Cho EY, Park T, Oh S, Jang WS, Kim SK et al. 759 C/T polymorphism of 5-HT2C receptor gene and early phase weight gain associated with antipsychotic drug treatment. Prog Neuropsychopharmacol Biol Psychiatry 2007; 31: 673–677.
De Luca V, Mueller DJ, de Bartolomeis A, Kennedy JL . Association of the HTR2C gene and antipsychotic induced weight gain: a meta-analysis. Int J Neuropsychopharmacol 2007; 10: 697–704.
Ellingrod VL, Perry PJ, Ringold JC, Lund BC, Bever-Stille K, Fleming F et al. Weight gain associated with the −759C/T polymorphism of the 5HT2C receptor and olanzapine. Am J Med Genet B Neuropsychiatr Genet 2005; 134: 76–78.
Ascher-Svanum H, Stensland MD, Kinon BJ, Tollefson GD . Weight gain as a prognostic indicator of therapeutic improvement during acute treatment of schizophrenia with placebo or active antipsychotic. J Psychopharmacol 2005; 19: 110–117.
Basson BR, Kinon BJ, Taylor CC, Szymanski KA, Gilmore JA, Tollefson GD . Factors influencing acute weight change in patients with schizophrenia treated with olanzapine, haloperidol, or risperidone. J Clin Psychiatry 2001; 62: 231–238.
Zerssen D . Göttingen: Beltz Test GmbH. In: Collegium Internationale Psychiatrae Scalarum (Hrsg.). Internationale Skalen für Psychiatrie (4. Auflage), 1996.
Guy W . ECDEU Assessment Manual for Psychopharmacology. US National Institute of Mental Health, DHEW publication no. 76-338: Bethesda (MD), 1976.
Kirchherr H, Kuhn-Velten WN . Quantitative determination of forty-eight antidepressants and antipsychotics in human serum by HPLC tandem mass spectrometry: a multi-level, single-sample approach. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 843: 100–113.
Popp J, Messner B, Steimer W . High-speed genotyping of CYP1A2*1F mutation with fluorescent hybridization probes using the LightCycler. Pharmacogenomics 2003; 4: 643–646.
Belfer I, Buzas B, Hipp H, Phillips G, Taubman J, Lorincz I . et al. Haplotype-based analysis of alpha 2A, 2B, and 2C adrenergic receptor genes captures information on common functional loci at each gene. J Hum Genet 2005; 50: 12–20.
Arranz MJ, Erdmann J, Kirov G, Rietschel M, Sodhi M, Albus M et al. 5-HT2A receptor and bipolar affective disorder: association studies in affected patients. Neurosci Lett 1997; 224: 95–98.
Lane HY, Lin CC, Huang CH, Chang YC, Hsu SK, Chang WH . Risperidone response and 5-HT6 receptor gene variance: genetic association analysis with adjustment for nongenetic confounders. Schizophr Res 2004; 67: 63–70.
Arinami T, Gao M, Hamaguchi H, Toru M . A functional polymorphism in the promoter region of the dopamine D2 receptor gene is associated with schizophrenia. Hum Mol Genet 1997; 6: 577–582.
Grandy DK, Zhang Y, Civelli O . PCR detection of the TaqA RFLP at the DRD2 locus. Hum Mol Genet 1993; 2: 2197.
Siffert W, Rosskopf D, Siffert G, Busch S, Moritz A, Erbel R et al. Association of a human G-protein beta3 subunit variant with hypertension. Nat Genet 1998; 18: 45–48.
Cohen J . Statistical Power Analysis for the Behavioral Sciences, 2nd edn, Lawrence Erlbaum Associates: Hillsdale, NJ, 1988.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laika, B., Leucht, S., Heres, S. et al. Pharmacogenetics and olanzapine treatment: CYP1A2*1F and serotonergic polymorphisms influence therapeutic outcome. Pharmacogenomics J 10, 20–29 (2010). https://doi.org/10.1038/tpj.2009.32
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2009.32
Keywords
This article is cited by
-
CYP1A2 expression rather than genotype is associated with olanzapine concentration in psychiatric patients
Scientific Reports (2023)
-
The association of genetic polymorphisms in CYP1A2, UGT1A4, and ABCB1 with autonomic nervous system dysfunction in schizophrenia patients treated with olanzapine
BMC Psychiatry (2020)
-
Interaction Between Variations in Dopamine D2 and Serotonin 2A Receptor is Associated with Short-Term Response to Antipsychotics in Schizophrenia
Neuroscience Bulletin (2019)
-
Gini Index-Based Maximum Concentration and Area Under the Curve Split Points for Analysing Adverse Event Occurrence in Bioequivalence Studies
Pharmaceutical Medicine (2018)
-
Genetic Variation in Melatonin Pathway Enzymes in Children with Autism Spectrum Disorder and Comorbid Sleep Onset Delay
Journal of Autism and Developmental Disorders (2015)