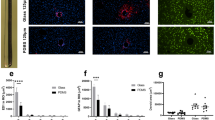
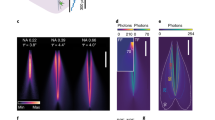
Abstract
Deciphering the neural patterns underlying brain functions is essential to understanding how neurons are organized into networks. This deciphering has been greatly facilitated by optogenetics and its combination with optoelectronic devices to control neural activity with millisecond temporal resolution and cell type specificity. However, targeting small brain volumes causes photoelectric artefacts, in particular when light emission and recording sites are close to each other. We take advantage of the photonic properties of tapered fibres to develop integrated ‘fibertrodes’ able to optically activate small brain volumes with abated photoelectric noise. Electrodes are positioned very close to light emitting points by non-planar microfabrication, with angled light emission allowing the simultaneous optogenetic manipulation and electrical read-out of one to three neurons, with no photoelectric artefacts, in vivo. The unconventional implementation of two-photon polymerization on the curved taper edge enables the fabrication of recoding sites all around the implant, making fibertrodes a promising complement to planar microimplants.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding authors on reasonable request and are available in the Zenodo repository (https://doi.org/10.5281/zenodo.6477861). Data on the optrode fabrication and in vitro optical measurements are available from B.S., A.B., L.S., F. Pisanello and M.D.V. Data on the in vivo use of tapered fibres are available from B.S., R.T.P., B.L.S. and F. Pisanello.
Code availability
The scripts related to the Monte Carlo simulations used in the current study are available from the corresponding authors on reasonable request and are available in the Zenodo repository (https://doi.org/10.5281/zenodo.6477861).
References
Camporeze, B. et al. Optogenetics: the new molecular approach to control functions of neural cells in epilepsy, depression and tumors of the central nervous system. Am. J. Cancer Res. 8, 1900–1918 (2018).
Grosenick, L., Marshel, J. H. & Deisseroth, K. Closed-loop and activity-guided optogenetic control. Neuron 86, 106–139 (2015).
Segev, E. et al. Patterned photostimulation via visible-wavelength photonic probes for deep brain optogenetics. Neurophotonics 4, 11002 (2016).
Zorzos, A. N., Boyden, E. S. & Fonstad, C. G. Multiwaveguide implantable probe for light delivery to sets of distributed brain targets. Opt. Lett. 35, 4133–4135 (2010).
Wu, F. et al. An implantable neural probe with monolithically integrated dielectric waveguide and recording electrodes for optogenetics applications. J. Neural Eng. 10, 56012 (2013).
Deubner, J., Coulon, P. & Diester, I. Optogenetic approaches to study the mammalian brain. Curr. Opin. Struct. Biol. 57, 157–163 (2019).
Nakamura, T. et al. Increased intracellular Ca2+ concentration in the hippocampal CA1 area during global ischemia and reperfusion in the rat: a possible cause of delayed neuronal death. Neuroscience 88, 57–67 (1999).
Bradley, P. M. J., Murphy, D., Kasparov, S., Croker, J. & Paton, J. F. R. A micro-optrode for simultaneous extracellular electrical and intracellular optical recording from neurons in an intact oscillatory neuronal network. J. Neurosci. Methods 168, 383–395 (2008).
LeChasseur, Y. et al. A microprobe for parallel optical and electrical recordings from single neurons in vivo. Nat. Methods 8, 319–325 (2011).
Anikeeva, P. et al. Optetrode: a multichannel readout for optogenetic control in freely moving mice. Nat. Neurosci. 15, 163–170 (2012).
Kim, E. G. R. et al. 3D silicon neural probe with integrated optical fibers for optogenetic modulation. Lab Chip 15, 2939–2949 (2015).
Sileo, L. et al. Tapered fibers combined with a multi-electrode array for optogenetics in mouse medial prefrontal cortex. Front. Neurosci. https://doi.org/10.3389/fnins.2018.00771 (2018).
Kozai, T. D. Y. & Vazquez, A. L. Photoelectric artefact from optogenetics and imaging on microelectrodes and bioelectronics: new challenges and opportunities. J. Mater. Chem. B 3, 4965–4978 (2015).
Kampasi, K. et al. Dual color optogenetic control of neural populations using low-noise, multishank optoelectrodes. Microsyst. Nanoeng. 4, 10 (2018).
Park, S. et al. One-step optogenetics with multifunctional flexible polymer fibers. Nat. Neurosci. 20, 612–619 (2017).
Lee, J., Ozden, I., Song, Y.-K. & Nurmikko, A. V. Transparent intracortical microprobe array for simultaneous spatiotemporal optical stimulation and multichannel electrical recording. Nat. Methods 12, 1157–1162 (2015).
Aravanis, A. M. et al. An optical neural interface: in vivo control of rodent motor cortex with integrated fiberoptic and optogenetic technology. J. Neural Eng. 4, S143 (2007).
Yizhar, O., Fenno, L. E., Davidson, T. J., Mogri, M. & Deisseroth, K. Optogenetics in neural systems. Neuron 71, 9–34 (2011).
Mohanty, A. et al. Reconfigurable nanophotonic silicon probes for sub-millisecond deep-brain optical stimulation. Nat. Biomed. Eng. 4, 223–231 (2020).
Klein, E., Gossler, C., Paul, O. & Ruther, P. High-density μLED-based optical cochlear implant with improved thermomechanical behavior. Front. Neurosci. https://doi.org/10.3389/fnins.2018.00659 (2018).
Wu, F. et al. Monolithically integrated μLEDs on silicon neural probes for high-resolution optogenetic studies in behaving animals. Neuron 88, 1136–1148 (2015).
Kim, K. et al. Artifact-free and high-temporal-resolution in vivo opto-electrophysiology with microLED optoelectrodes. Nat. Commun. 11, 2063 (2020).
McAlinden, N. et al. Thermal and optical characterization of micro-LED probes for in vivo optogenetic neural stimulation. Opt. Lett. 38, 992–994 (2013).
Scharf, R. et al. Depth-specific optogenetic control in vivo with a scalable, high-density μLED neural probe. Sci. Rep. 6, 28381 (2016).
Pisanello, F. et al. Dynamic illumination of spatially restricted or large brain volumes via a single tapered optical fiber. Nat. Neurosci. 20, 1180–1188 (2017).
Pisanello, M. et al. Tailoring light delivery for optogenetics by modal demultiplexing in tapered optical fibers. Sci. Rep. 8, 4467 (2018).
Pisano, F. et al. Depth-resolved optical monitoring of neural activity in freely moving animals. In 22nd International Conference on Transparent Optical Networks (ICTON 2020), Publisher: IEEE, Page number Tu.D7.4, https://doi.org/10.1109/ICTON51198.2020.9203401
Pisanello, M., Pisanello, F., Sileo, L. and De Vittorio, M. Photonic technologies for optogenetics. In 16th International Conference on Transparent Optical Networks (ICTON 2014). Publisher: IEEE, Page number Mo.C5.3, https://doi.org/10.1109/ICTON.2014.6876293
Warden, M. R., Cardin, J. A. & Deisseroth, K. Optical neural interfaces. Annu. Rev. Biomed. Eng. 16, 103–129 (2014).
Dufour, S. & De Koninck, Y. Optrodes for combined optogenetics and electrophysiology in live animals. Neurophotonics 2, 031205 (2015).
Kostovski, G., Stoddart, P. R. & Mitchell, A. The optical fiber tip: an inherently light‐coupled microscopic platform for micro‐and nanotechnologies. Adv. Mater. 26, 3798–3820 (2014).
Pisanello, M. et al. Modal demultiplexing properties of tapered and nanostructured optical fibers for in vivo optogenetic control of neural activity. Biomed. Opt. Express 6, 4014–4026 (2015).
Pisanello, F. et al. Multipoint-emitting optical fibers for spatially addressable in vivo optogenetics. Neuron 82, 1245–1254 (2014).
Sileo, L. et al. Fabrication of multipoint light emitting optical fibers for optogenetics. Proc. SPIE https://doi.org/10.1117/12.2075819 (2015).
Song, J. S., Lee, S., Jung, S. H., Cha, G. C. & Mun, M. S. Improved biocompatibility of parylene‐C films prepared by chemical vapor deposition and the subsequent plasma treatment. J. Appl. Polym. Sci. 112, 3677–3685 (2009).
Pisano, F. et al. Focused ion beam nanomachining of tapered optical fibers for patterned light delivery. Microelectron. Eng. 195, 41–49 (2018).
Abouzari, M. R. S., Berkemeier, F., Schmitz, G. & Wilmer, D. On the physical interpretation of constant phase elements. Solid State Ion. 180, 922–927 (2009).
Rocha, P. R. F. et al. Electrochemical noise and impedance of Au electrode/electrolyte interfaces enabling extracellular detection of glioma cell populations. Sci. Rep. 6, 34843 (2016).
Hazubski, S., Soekadar, S. R., Hoppe, H. & Otte, A. Neuroprosthetics 2.0. EBioMedicine 48, 22 (2019).
Stujenske, J. M., Spellman, T. & Gordon, J. A. Modeling the spatiotemporal dynamics of light and heat propagation for in vivo optogenetics. Cell Rep. 12, 525–534 (2015).
Maglie, E. et al. Ray tracing models for estimating light collection properties of microstructured tapered optical fibers for optical neural interfaces. Opt. Lett. 45, 3856–3859 (2020).
Steenland, H. W. & McNaughton, B. L. in Analysis and Modeling of Coordinated Multi-neuronal Activity (ed. Tatsuno, M.) 41–61 (Springer, 2015).
Saleh, B. E. A. & Teich, M. C. Fundamentals of Photonics (John Wiley & Sons, 2019).
Baldacchini, T. Three-Dimensional Microfabrication Using Two-Photon Polymerization: Fundamentals, Technology, and Applications (William Andrew, 2015).
Nieh, E. H. et al. Decoding neural circuits that control compulsive sucrose seeking. Cell 160, 528–541 (2015).
Brown, J. et al. Expanding the optogenetics toolkit by topological inversion of rhodopsins. Cell 175, 1131–1140 (2018).
Stark, E. et al. Pyramidal cell-interneuron interactions underlie hippocampal ripple oscillations. Neuron 83, 467–480 (2014).
Pisanello, F., Sileo, L. & De Vittorio, M. Micro- and nanotechnologies for optical neural interfaces. Front. Neurosci. https://doi.org/10.3389/fnins.2016.00070 (2016).
Sigurdsson, T., Stark, K. L., Karayiorgou, M., Gogos, J. A. & Gordon, J. A. Impaired hippocampal–prefrontal synchrony in a genetic mouse model of schizophrenia. Nature 464, 763–767 (2010).
Schmitzer-Torbert, N. 1, Jackson, J., Henze, D., Harris, K. & Redish, A. D. Quantitative measures of cluster quality for use in extracellular recordings. Neuroscience 131, 1–11 (2005).
Dhawale, A. K. et al. Automated long-term recording and analysis of neural activity in behaving animals. Elife 6, e27702 (2017).
Acknowledgements
B.S., A.B., M.B., F. Pisano and F. Pisanello acknowledge funding from the European Research Council under the European Union’s Horizon 2020 research and innovation programme (no. 677683); M.P. and M.D.V. acknowledge funding from the European Research Council under the European Union’s Horizon 2020 research and innovation programme (no. 692943). M.B., M.D.V. and F.Pisanello acknowledge funding from the European Research Council under the European Union’s Horizon 2020 research and innovation programme (no. 966674). F. Pisano, M.D.V. and F. Pisanello acknowledge funding from the European Union’s Horizon 2020 research and innovation programme (no. 101016787). L.S., M.D.V. and B.L.S. are funded by the US National Institutes of Health (U01NS094190). M.P., L.S., F. Pisanello, M.D.V. and B.L.S. are funded by the US National Institutes of Health (1UF1NS108177-01). A.B., F. Pisanello and M.D.V. also acknowledge funding from the European Union’s Horizon 2020 research and innovation programme (no. 828972). We also acknowledge J. Lee for help setting up the optrode fibre launch system.
Author information
Authors and Affiliations
Contributions
A.B., B.S., M.P. and R.T.P. equally contributed to this work. A.B., M.P., M.B., A.R., M.D.V., F. Pisano and F. Pisanello developed the 2PP system and the related fibertrode. L.S., B.S., M.P., A.Q., F. Pisano, M.D.V. and F. Pisanello developed the FIB fabrication protocol and the related fibertrode. B.S., A.B., M.P., M.B. and F. Pisanello performed the optoelectrical characterization of the probes. B.S. and R.T.P. performed the in vivo experiments. B.S., A.B., M.P., F. Pisano, F. Pisanello, R.T.P., B.L.S., J.A.A. and M.D.V. analysed and discussed the in vivo data. B.L.S., R.T.P., B.S., D.D.L., F.D.N. and F. Pisanello developed the in vivo experiment protocols. A.B., B.S., L.S., F. Pisanello, M.D.V., B.L.S. and J.A.A. wrote the manuscript and prepared the figures with contributions from all authors. M.D.V., B.L.S., J.A.A. and F. Pisanello conceived the study and jointly supervised the work.
Corresponding authors
Ethics declarations
Competing interests
L.S., B.L.S., M.D.V. and F. Pisanello are founders of and hold private equity in Optogenix, a company that develops, produces and sells technologies to deliver light into the brain. Tapered fibres commercially available from Optogenix were used as tools in the research. The remaining authors declare no competing interests. F. Pisano and M.P. have been employed by OptogeniX. OptogeniX did not fund the research described in this work.
Peer review
Peer review information
Nature Materials thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–9 and Table 1.
Rights and permissions
About this article
Cite this article
Spagnolo, B., Balena, A., Peixoto, R.T. et al. Tapered fibertrodes for optoelectrical neural interfacing in small brain volumes with reduced artefacts. Nat. Mater. 21, 826–835 (2022). https://doi.org/10.1038/s41563-022-01272-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-022-01272-8
This article is cited by
-
A deeper understanding of the brain with photonic–electronic integration
Nature Electronics (2023)