Abstract
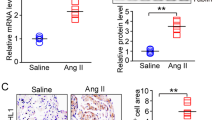
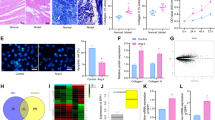
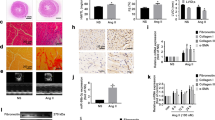
Atrial fibrillation (AF), the most common cardiac arrhythmia, is an important contributor to mortality and morbidity. Ubquitin-specific protease 7 (USP7), one of the most abundant ubiquitin-specific proteases (USP), participated in many cellular events, such as cell proliferation, apoptosis, and tumourigenesis. However, its role in AF remains unknown. Here, the mice were treated with Ang II infusion to induce the AF model. Echocardiography was used to measure the atrial diameter. Electrical stimulation was programmed to measure the induction and duration of AF. The changes in atrial remodeling were measured using routine histologic analysis. Here, a significant increase in USP7 expression was observed in Ang II-stimulated atrial cardiomyocytes and atrial tissues, as well as in atrial tissues from patients with AF. The administration of p22077, the inhibitor of USP7, attenuated Ang II-induced inducibility and duration of AF, atrial dilatation, connexin dysfunction, atrial fibrosis, atrial inflammation, and atrial oxidase stress, and then inhibited the progression of AF. Mechanistically, the administration of p22077 alleviated Ang II-induced activation of TGF-β/Smad2, NF-κB/NLRP3, NADPH oxidases (NOX2 and NOX4) signals, the up-regulation of CX43, ox-CaMKII, CaMKII, Kir2.1, and down-regulation of SERCA2a. Together, this study, for the first time, suggests that USP7 is a critical driver of AF and revealing USP7 may present a new target for atrial fibrillation therapeutic strategies.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Every single record generated in the current investigation remains accessible to the corresponding authors following reasonable request. All the data obtained in the current study were available from the corresponding authors on reasonable request. All data necessary for evaluating the conclusions of the paper are included in the paper. The authors of this paper will provide any additional data which are requested.
References
Dan GA. Rhythm control in AF: Have we reached the last frontier? Eur Cardiol. 2019;14:77–81.
Morin DP, Bernard ML, Madias C, Rogers PA, Thihalolipavan S, Estes NAM. The state of the art: Atrial fibrillation epidemiology, prevention, and treatment. Mayo Clin Proc. 2016;91:1778–810.
Arbelo E, Brugada J, Blomström-Lundqvist C, Laroche C, Kautzner J, Pokushalov E, et al. Contemporary management of patients undergoing atrial fibrillation ablation: in-hospital and 1-year follow-up findings from the ESC-EHRA atrial fibrillation ablation long-term registry. Eur Heart J. 2017;38:1303–16.
Demers J, Ton AT, Huynh F, Thibault S, Ducharme A, Paradis P, et al. Atrial electrical remodeling in mice with cardiac-specific overexpression of Angiotensin II Type 1 receptor. J Am Heart Assoc. 2022;11:e023974.
Chen Y, Chen X, Li H, Li Y, Cheng D, Tang Y, et al. Serum extracellular vesicles containing MIAT induces atrial fibrosis, inflammation and oxidative stress to promote atrial remodeling and atrial fibrillation via blockade of miR-485-5p-mediated CXCL10 inhibition. Clin Transl Med. 2021;11:e482.
Lu X, Rudemiller NP, Wen Y, Ren J, Hammer GE, Griffiths R, et al. A20 in myeloid cells protects against hypertension by inhibiting dendritic cell-mediated T-cell activation. Circ Res. 2019;125:1055–66.
Tang LJ, Zhou YJ, Xiong XM, Li NS, Zhang JJ, Luo XJ, et al. Ubiquitin-specific protease 7 promotes ferroptosis via activation of the p53/TfR1 pathway in the rat hearts after ischemia/reperfusion. Free Radic Biol Med. 2021;162:339–52.
Qin B, Zhou L, Wang F, Wang Y. Ubiquitin-specific protease 20 in human disease: Emerging role and therapeutic implications. Biochem Pharmacol. 2022;206:115352.
Lu X, Zhang Y, Zheng Y, Chen B. The miRNA-15b/USP7/KDM6B axis engages in the initiation of osteoporosis by modulating osteoblast differentiation and autophagy. J Cell Mol Med. 2021;25:2069–81.
Xu Q, Liu M, Gu J, Ling S, Liu X, Luo Z, et al. Ubiquitin-specific protease 7 regulates myocardial ischemia/reperfusion injury by stabilizing Keap1. Cell Death Discov. 2022;8:291.
Gong X, Li Y, He Y, Zhou F. USP7-SOX9-miR-96-5p-NLRP3 network regulates myocardial injury and cardiomyocyte pyroptosis in Sepsis. Hum Gene Ther. 2022;33:1073–90.
Huang YT, Cheng AC, Tang HC, Huang GC, Cai L, Lin TH, et al. USP7 facilitates SMAD3 autoregulation to repress cancer progression in p53-deficient lung cancer. Cell Death Dis. 2021;12:880.
Zhang N, Wang F, Zhang G, Zhang Q, Liu Y, Wang Q, et al. USP7 Promotes deubiquitination and stabilization of MyD88 to enhance immune responses. Front Immunol. 2022;13:900243.
Gao M, Qi Z, Deng M, Huang H, Xu Z, Guo G, et al. The deubiquitinase USP7 regulates oxidative stress through stabilization of HO-1. Oncogene. 2022;41:4018–27.
Gu YH, Ren KW, Wang Y, Wang SH, Yu XH, Xu LW, et al. Administration of USP7 inhibitor P22077 inhibited cardiac hypertrophy and remodeling in Ang II-induced hypertensive mice. Front Pharm. 2022;13:1021361.
Palazón-Riquelme P, Worboys JD, Green J, Valera A, Martín-Sánchez F, Pellegrini C, et al. USP7 and USP47 deubiquitinases regulate NLRP3 inflammasome activation. EMBO Rep. 2018;19:e44766.
Liu Y, Lv H, Tan R, An X, Niu X-H, Liu Y-J, et al. Platelets promote Ang II (Angiotensin II)-induced atrial fibrillation by releasing TGF-β1 (Transforming Growth Factor-β1) and interacting with fibroblasts. Hypertension. 2020;76:1856–67.
Bi H-L, Zhang X-L, Zhang Y-L, Xie X, Xia Y-L, Du J, et al. The deubiquitinase UCHL1 regulates cardiac hypertrophy by stabilizing epidermal growth factor receptor. Sci Adv. 2020;6:eaax4826.
Shi K-N, Li P-B, Su H-X, Gao J, Li H-H. MK-886 protects against cardiac ischaemia/reperfusion injury by activating proteasome-Keap1-NRF2 signalling. Redox Biol. 2023;62:102706.
Yao C, Veleva T, Scott L, Cao S, Li L, Chen G, et al. Enhanced Cardiomyocyte NLRP3 inflammasome signaling promotes atrial fibrillation. Circulation. 2018;138:2227–42.
Lv W, Zhang L, Cheng X, Wang H, Qin W, Zhou X, et al. Apelin inhibits angiotensin ii-induced atrial fibrosis and atrial fibrillation via TGF-β1/Smad2/α-SMA pathway. Front Physiol. 2020;11:583570.
Nattel S, Heijman J, Zhou L, Dobrev D. Molecular basis of atrial fibrillation pathophysiology and therapy: a translational perspective. Circ Res. 2020;127:51–72.
Zhang Y-L, Cao H-J, Han X, Teng F, Chen C, Yang J, et al. Chemokine receptor CXCR-2 initiates atrial fibrillation by triggering monocyte mobilization in mice. Hypertension. 2020;76:381–92.
Ao X, Ding W, Li X, Xu Q, Chen X, Zhou X, et al. Non-coding RNAs regulating mitochondrial function in cardiovascular diseases. J Mol Med. 2023;101:501–26.
De Jong AM, Maass AH, Oberdorf-Maass SU, Van Veldhuisen DJ, Van Gilst WH, Van Gelder IC. Mechanisms of atrial structural changes caused by stretch occurring before and during early atrial fibrillation. Cardiovasc Res. 2011;89:754–65.
Schotten U, Verheule S, Kirchhof P, Goette A. Pathophysiological mechanisms of atrial fibrillation: a translational appraisal. Physiol Rev. 2011;91:265–325.
Swaminathan PD, Purohit A, Soni S, Voigt N, Singh MV, Glukhov AV, et al. Oxidized CaMKII causes cardiac sinus node dysfunction in mice. J Clin Invest. 2011;121:3277–88.
Carlson CR, Aronsen JM, Bergan-Dahl A, Moutty MC, Lunde M, Lunde PK, et al. AKAP18δ anchors and regulates CaMKII activity at Phospholamban-SERCA2 and RYR. Circ Res. 2022;130:27–44.
Luo X, Yu W, Liu Z, Pu Z, Liu T, Li Y, et al. Ageing increases cardiac electrical remodelling in rats and mice via NOX4/ROS/CaMKII-mediated calcium signalling. Oxid Med Cell Longev. 2022;2022:8538296.
Zuo K, Fang C, Liu Z, Fu Y, Liu Y, Liu L, et al. Commensal microbe-derived SCFA alleviates atrial fibrillation via GPR43/NLRP3 signaling. Int J Biol Sci. 2022;18:4219–32.
Mighiu AS, Recalde A, Ziberna K, Carnicer R, Tomek J, Bub G, et al. Inducibility, but not stability, of atrial fibrillation is increased by NOX2 overexpression in mice. Cardiovasc Res. 2021;117:2354–64.
Liu Y, Ding W, Wang J, Ao X, Xue J. Non-coding RNA-mediated modulation of ferroptosis in cardiovascular diseases. Biomed Pharmacother. 2023;164:114993.
Liu G, Liu Q, Yan B, Zhu Z, Xu Y. USP7 inhibition alleviates H2O2-induced injury in chondrocytes via inhibiting NOX4/NLRP3 pathway. Front Pharm. 2020;11:617270.
Xue Q, Yang D, Zhang J, Gan P, Lin C, Lu Y, et al. USP7, negatively regulated by miR-409-5p, aggravates hypoxia-induced cardiomyocyte injury. APMIS. 2021;129:152–62.
Dong X, Yang C, Luo Y, Dong W, Xu X, Wu Y, et al. USP7 attenuates endoplasmic reticulum stress and NF-κB signaling to modulate chondrocyte proliferation, apoptosis, and inflammatory response under inflammation. Oxid Med Cell Longev. 2022;2022:1835900.
Lu H, Yuan P, Ma X, Jiang X, Liu S, Ma C, et al. Angiotensin-converting enzyme inhibitor promotes angiogenesis through Sp1/Sp3-mediated inhibition of notch signaling in male mice. Nat Commun. 2023;14:731.
Li J, Wang S, Bai J, Yang X-L, Zhang Y-L, Che Y-L, et al. Novel role for the immunoproteasome subunit PSMB10 in Angiotensin II–induced atrial fibrillation in mice. Hypertension. 2018;71:866–76.
Afonina IS, Zhong Z, Karin M, Beyaert R. Limiting inflammation—the negative regulation of NF-κB and the NLRP3 inflammasome. Nat Immunol. 2017;18:861–9.
Cau SB, Bruder-Nascimento A, Silva MB, Ramalho FNZ, Mestriner F, Alves-Lopes R, et al. Angiotensin-II activates vascular inflammasome and induces vascular damage. Vasc Pharmacol. 2021;139:106881.
Colleran A, Collins PE, O’Carroll C, Ahmed A, Mao X, McManus B, et al. Deubiquitination of NF-κB by Ubiquitin-Specific Protease-7 promotes transcription. Proc Natl Acad Sci USA. 2013;110:618–23.
Nie L, Wang C, Liu X, Teng H, Li S, Huang M, et al. USP7 substrates identified by proteomics analysis reveal the specificity of USP7. Genes Dev. 2022;36:1016–30.
Gao G, Dudley SC. Redox regulation, NF-kappaB, and atrial fibrillation. Antioxid Redox Signal. 2009;11:2265–77.
Funding
This study’s completion was funded by grants from the National Natural Science Foundation of China (82170320 to H.-L.B.); the Natural Science Foundation of Liaoning Provincial (2021-MS-279 to H.-L.B.); the Dalian Science Fund for Distinguished Young Scholars (2023RJ020 to H.-L.B.; 2022RJ13 to X.-L.Y.); Dalian Medical University Interdisciplinary Research Cooperation Project Team Funding (JCHZ2023023 to H.-L.B.).
Author information
Authors and Affiliations
Contributions
H.-L. B. and X.-L. Y. conceived the project. Y. W., Y.-H. G., K.-W. R., and X. X. performed the experiments and analyzed the data, S.-H. W., X.-X. Z. and L. W. were responsible for human clinical studies and analyses. H.-L. B., X.-L. Y. and Y. W. wrote the paper with input from all authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics
The Animal Care and Use Committee of Dalian Medical University assessed and authorized the laboratory study, which was conducted out in accordance with the US National Institutes of Health Studies involving human participants have been authorized by the First Affiliated Hospital of Dalian Medical University (PJ-KS-KY-2021-229).The animal study was assessed and authorised by the Animal Care and Use Committee of Dalian Medical University and carried out in accordance with the U.S. National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals (publication no. 85-23, 1996). Studies involving human participants were approved by the First Affiliated Hospital of Dalian Medical University (PJ-KS-KY-2021-229).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Gu, YH., Ren, KW. et al. Administration of USP7 inhibitor p22077 alleviates Angiotensin II (Ang II)-induced atrial fibrillation in Mice. Hypertens Res 47, 1309–1322 (2024). https://doi.org/10.1038/s41440-024-01581-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-024-01581-2