Abstract
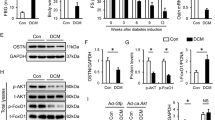
Mitochondrial dysfunction plays a critical role in the pathogenesis of diabetic cardiomyopathy. Translocase of mitochondrial outer membrane 70 (Tom70) primarily facilitates the import of mitochondrial preproteins that may be involved in the regulation of oxidative stress and mitochondrial function. This study aimed to investigate the role of Tom70 in the development of myocardial injury in leptin receptor-deficient (db/db) diabetic mice. Tom70 siRNA or an overexpressing lentivirus was intramuscularly injected into mouse hearts or used to treat cultured neonatal cardiomyocytes. We found that Tom70 was downregulated in the diabetic hearts compared with the level in the wild-type hearts and that knocking down Tom70 exacerbated cardiac hypertrophy, fibrosis, and ventricular dysfunction in the db/db mice. Similarly, the in vitro data demonstrated that silencing Tom70 enhanced high-glucose and high-fat (HGHF) medium treatment-induced mitochondrial superoxide production, decreased ATP production and the mitochondrial membrane potential, and enhanced cell apoptosis in neonatal cardiomyocytes. Importantly, overexpression of Tom70 alleviated HGHF medium-induced oxidative stress, mitochondrial dysfunction, and cell apoptosis. Furthermore, in vivo data confirmed that reconstitution of Tom70 ameliorated cardiac hypertrophy, interstitial fibrosis, and ventricular dysfunction in the db/db mice. In addition, Tom70 overexpression mitigated mitochondrial fragmentation and dysfunction in the hearts of the db/db mice. Taken together, these findings suggest that downregulation of Tom70 contributes to the development of diabetic cardiomyopathy and that reconstitution of Tom70 may be a new therapeutic strategy for the prevention and treatment of diabetic cardiomyopathy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Boudina S, Abel ED. Diabetic cardiomyopathy revisited. Circulation. 2007;115:3213–23.
Borghetti G, von Lewinski D, Eaton DM, Sourij H, Houser SR, Wallner M. Diabetic cardiomyopathy: current and future therapies. beyond glycemic control. Front Physiol. 2018;9:1514.
Teshima Y, Takahashi N, Nishio S, Saito S, Kondo H, Fukui A, et al. Production of reactive oxygen species in the diabetic heart. Roles of mitochondria and NADPH oxidase. Circ J. 2014;78:300–6.
Anderson EJ, Rodriguez E, Anderson CA, Thayne K, Chitwood WR, Kypson AP. Increased propensity for cell death in diabetic human heart is mediated by mitochondrial-dependent pathways. Am J Physiol Heart Circ Physiol. 2011;300:H118–124.
Anderson EJ, Kypson AP, Rodriguez E, Anderson CA, Lehr EJ, Neufer PD. Substrate-specific derangements in mitochondrial metabolism and redox balance in the atrium of the type 2 diabetic human heart. J Am Coll Cardiol. 2009;54:1891–8.
Backes S, Hess S, Boos F, Woellhaf MW, Godel S, Jung M, et al. Tom70 enhances mitochondrial preprotein import efficiency by binding to internal targeting sequences. J Cell Biol. 2018;217:1369–82.
Fan AC, Kozlov G, Hoegl A, Marcellus RC, Wong MJ, Gehring K, et al. Interaction between the human mitochondrial import receptors Tom20 and Tom70 in vitro suggests a chaperone displacement mechanism. J Biol Chem. 2011;286:32208–19.
Filadi R, Leal NS, Schreiner B, Rossi A, Dentoni G, Pinho CM, et al. TOM70 Sustains Cell Bioenergetics by Promoting IP3R3-Mediated ER to Mitochondria Ca(2+) Transfer. Curr Biol. 2018;28:369–82. e366
Li J, Qi M, Li C, Shi D, Zhang D, Xie D, et al. Tom70 serves as a molecular switch to determine pathological cardiac hypertrophy. Cell Res. 2014;24:977–93.
Pei HF, Hou JN, Wei FP, et al. Melatonin attenuates postmyocardial infarction injury via increasing Tom70 expression. J Pineal Res. 2017;62:1–13
Santos JM, Kowluru RA. Impaired transport of mitochondrial transcription factor A (TFAM) and the metabolic memory phenomenon associated with the progression of diabetic retinopathy. Diabetes Metab Res Rev. 2013;29:204–13.
Ehler E, Moore-Morris T, Lange S. Isolation and culture of neonatal mouse cardiomyocytes. J Vis Exp. 2013;6:50154.
Vidyasekar P, Shyamsunder P, Santhakumar R, Arun R, Verma RS. A simplified protocol for the isolation and culture of cardiomyocytes and progenitor cells from neonatal mouse ventricles. Eur J Cell Biol. 2015;94:444–52.
Zhang Y, Zhang Y, Li W, et al. Uncoupling Protein 2 Inhibits Myointimal Hyperplasia in Preclinical Animal Models of Vascular Injury. J Am Heart Assoc. 2017;6:e002641.
Duncan JG. Mitochondrial dysfunction in diabetic cardiomyopathy. Biochim Biophys Acta. 2011;1813:1351–9.
Edmonson AM, Mayfield DK, Vervoort V, DuPont BR, Argyropoulos G. Characterization of a human import component of the mitochondrial outer membrane, TOMM70A. Cell Commun Adhes. 2002;9:15–27.
Sun J, Pu Y, Wang P, Chen S, Zhao Y, Liu C, et al. TRPV1-mediated UCP2 upregulation ameliorates hyperglycemia-induced endothelial dysfunction. Cardiovasc Diabetol 2013;12:69.
Xiong S, Wang P, Ma L, Gao P, Gong L, Li L, et al. Ameliorating endothelial mitochondrial dysfunction restores coronary function via transient receptor potential vanilloid 1-mediated protein kinase A/uncoupling protein 2 pathway. Hypertension. 2016;67:451–60.
Pierrat B, Ito M, Hinz W, Simonen M, Erdmann D, Chiesi M, et al. Uncoupling proteins 2 and 3 interact with members of the 14.3.3 family. Eur J Biochem. 2000;267:2680–7.
Young JC, Hoogenraad NJ, Hartl FU. Molecular chaperones Hsp90 and Hsp70 deliver preproteins to the mitochondrial import receptor Tom70. Cell. 2003;112:41–50.
Takano T, Kohara M, Kasama Y, Nishimura T, Saito M, Kai C, et al. Translocase of outer mitochondrial membrane 70 expression is induced by hepatitis C virus and is related to the apoptotic response. J Med Virol. 2011;83:801–9.
Xue Q, Pei H, Liu Q, Zhao M, Sun J, Gao E, et al. MICU1 protects against myocardial ischemia/reperfusion injury and its control by the importer receptor Tom70. Cell Death Dis. 2017;8:e2923.
Kato H, Lu Q, Rapaport D, Kozjak-Pavlovic V. Tom70 is essential for PINK1 import into mitochondria. PLoS One 2013;8:e58435.
Park J, Lee SB, Lee S, Kim Y, Song S, Kim S, et al. Mitochondrial dysfunction in Drosophila PINK1 mutants is complemented by parkin. Nature. 2006;441:1157–61.
Ji L, Liu F, Jing Z, Huang Q, Zhao Y, Cao H, et al. MICU1 alleviates diabetic cardiomyopathy through mitochondrial Ca(2+)-dependent antioxidant response. Diabetes 2017;66:1586–1600.
Jia G, Hill MA, Sowers JR. Diabetic cardiomyopathy: an update of mechanisms contributing to this clinical entity. Circ Res 2018;122:624–38.
Bugger H, Abel ED. Molecular mechanisms of diabetic cardiomyopathy. Diabetologia. 2014;57:660–71.
Liu F, Song R, Feng Y, Guo J, Chen Y, Zhang Y, et al. Upregulation of MG53 induces diabetic cardiomyopathy through transcriptional activation of peroxisome proliferation-activated receptor alpha. Circulation. 2015;131:795–804.
Battiprolu PK, Hojayev B, Jiang N, Wang ZV, Luo X, Iglewski M, et al. Metabolic stress-induced activation of FoxO1 triggers diabetic cardiomyopathy in mice. J Clin Invest. 2012;122:1109–18.
Shepherd DL, Hathaway QA, Nichols CE, Durr AJ, Pinti MV, Hughes KM, et al. Mitochondrial proteome disruption in the diabetic heart through targeted epigenetic regulation at the mitochondrial heat shock protein 70 (mtHsp70) nuclear locus. J Mol Cell Cardiol. 2018;119:104–15.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (81970262) and the Innovation Team Project from the Department of Education of Sichuan Province (18TD0030).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, P., Wang, D., Yang, Y. et al. Tom70 protects against diabetic cardiomyopathy through its antioxidant and antiapoptotic properties. Hypertens Res 43, 1047–1056 (2020). https://doi.org/10.1038/s41440-020-0518-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-020-0518-x
Keywords
This article is cited by
-
Annual reports on hypertension research 2020
Hypertension Research (2022)