Abstract
Background
Multivitamin use is common among cancer patients. Whether post-diagnostic multivitamin supplementation is beneficial for prostate cancer survival is largely unknown, and some evidence even suggests potential harm.
Methods
We prospectively assessed post-diagnostic multivitamin use in relation to prostate cancer survival among 4756 men with nonmetastatic prostate cancer at diagnosis in the Health Professionals Follow-up Study (1986–2016). Cox regression models were used to evaluate the association between post-diagnostic multivitamin use and frequency and risk of lethal prostate cancer (distant metastases or prostate cancer-specific death) and all-cause mortality.
Results
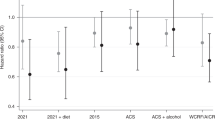
We observed 438 lethal prostate cancer and 2609 deaths during a median follow-up of 11 years. Compared to non-users, post-diagnostic multivitamin use was not associated with risk of lethal prostate cancer (HR [95% CI], 0.98 [0.74–1.30]) or all-cause mortality (1.00 [0.88–1.12]), after adjustment for potential confounders. Similarly, null associations were observed across various categories of use frequency. Compared to non-users, men who used multivitamins regularly (6–9 tablets/week) after cancer diagnosis had similar risk of lethal prostate cancer (0.96 [0.72–1.28]) and all-cause mortality (0.99 [0.88–1.12]).
Conclusions
We found no evidence that post-diagnostic multivitamin use among men with nonmetastatic prostate cancer was associated with better or worse survival in a well-nourished population.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
Investigators wishing to use HPFS data are asked to submit a brief description of the proposed project (go to https://www.nurseshealthstudy.org/researchers (contact email: nhsaccess@channing.harvard.edu) and https://sites.sph.harvard.edu/hpfs/for-collaborators/ for details). Other data that support the findings of this study are available from the corresponding author upon request.
References
Kantor ED, Rehm CD, Du M, White E, Giovannucci EL. Trends in dietary supplement use among US adults from 1999-2012. Jama. 2016;316:1464–74.
Bailey RL, Gahche JJ, Lentino CV, Dwyer JT, Engel JS, Thomas PR, et al. Dietary supplement use in the United States, 2003-2006. J Nutr. 2011;141:261–6. https://doi.org/10.3945/jn.110.133025.
Velicer CM, Ulrich CM. Vitamin and mineral supplement use among US adults after cancer diagnosis: a systematic review. J Clin Oncol. 2008;26:665–73. https://doi.org/10.1200/JCO.2007.13.5905.
Rock CL, Doyle C, Demark-Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL, et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin. 2012;62:243–74. https://doi.org/10.3322/caac.21142.
National Cancer Institute SEER Program. Cancer Stat Facts: Prostate Cancer. Accessed 24 Jan 2022, https://seer.cancer.gov/statfacts/html/prost.html.
Chan JM, Elkin EP, Silva SJ, Broering JM, Latini DM, Carroll PR. Total and specific complementary and alternative medicine use in a large cohort of men with prostate cancer. Urology. 2005;66:1223–8. https://doi.org/10.1016/j.urology.2005.06.003.
Wilkinson S, Gomella LG, Smith JA, Brawer MK, Dawson NA, Wajsman Z, et al. Attitudes and use of complementary medicine in men with prostate cancer. J Urol. 2002;168:2505–9. https://doi.org/10.1097/01.ju.0000037733.62447.50.
Wiygul JB, Evans BR, Peterson BL, Polascik TJ, Walther PJ, Robertson CN, et al. Supplement use among men with prostate cancer. Urology. 2005;66:161–6. https://doi.org/10.1016/j.urology.2005.01.035.
Zuniga KB, Zhao S, Kenfield SA, Cedars B, Cowan JE, Van Blarigan EL, et al. Trends in Complementary and Alternative Medicine Use among Patients with Prostate Cancer. J Urol. 2019;202:689–95. https://doi.org/10.1097/JU.0000000000000336.
Van Blarigan E, Kenfield SA, Cedars BE, Broering J, Cowan JE, Carroll P, et al. Multivitamin use and risk of prostate cancer recurrence: Data from the Cancer of the Prostate Strategic Urologic Research Endeavor (CaPSURE). Am Soc Clin Oncol. 2018. https://doi.org/10.1200/JCO.2018.36.6_suppl.70.
Gaziano JM, Sesso HD, Christen WG, Bubes V, Smith JP, MacFadyen J, et al. Multivitamins in the prevention of cancer in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2012;308:1871–80. https://doi.org/10.1001/jama.2012.14641.
Park SY, Murphy SP, Wilkens LR, Henderson BE, Kolonel LN. Multivitamin use and the risk of mortality and cancer incidence: the multiethnic cohort study. Am J Epidemiol. 2011;173:906–14. https://doi.org/10.1093/aje/kwq447.
Zhang Y, Coogan P, Palmer JR, Strom BL, Rosenberg L. Vitamin and mineral use and risk of prostate cancer: the case-control surveillance study. Cancer Causes Control. 2009;20:691–8. https://doi.org/10.1007/s10552-008-9282-y.
Kristal AR, Stanford JL, Cohen JH, Wicklund K, Patterson RE. Vitamin and mineral supplement use is associated with reduced risk of prostate cancer. Cancer Epidemiol Biomark Prev. 1999;8:887–92.
Zhang Y, Song M, Mucci LA, Giovannucci EL. Regular, long-duration multivitamin use and risk of overall and aggressive prostate cancer in the health professionals follow-up study. J Urol. 2022;208:633–40. https://doi.org/10.1097/JU.0000000000002735.
Lawson KA, Wright ME, Subar A, Mouw T, Hollenbeck A, Schatzkin A, et al. Multivitamin use and risk of prostate cancer in the National Institutes of Health-AARP Diet and Health Study. J Natl Cancer Inst. 2007;99:754–64. https://doi.org/10.1093/jnci/djk177.
Stevens VL, McCullough ML, Diver WR, Rodriguez C, Jacobs EJ, Thun MJ, et al. Use of multivitamins and prostate cancer mortality in a large cohort of US men. Cancer Causes Control. 2005;16:643–50. https://doi.org/10.1007/s10552-005-0384-5.
Watkins ML, Erickson JD, Thun MJ, Mulinare J, Heath CW Jr. Multivitamin use and mortality in a large prospective study. Am J Epidemiol. 2000;152:149–62. https://doi.org/10.1093/aje/152.2.149.
Kim HJ, Giovannucci E, Rosner B, Willett WC, Cho E. Longitudinal and secular trends in dietary supplement use: nurses’ health study and health professionals follow-up study, 1986-2006. J Acad Nutr Dietetics. 2014;114:436–43.
Rimm EB, Giovannucci EL, Stampfer MJ, Colditz GA, Litin LB, Willett WC. Reproducibility and validity of an expanded self-administered semiquantitative food frequency questionnaire among male health professionals. Am J Epidemiol. 1992;135:1114–26. https://doi.org/10.1093/oxfordjournals.aje.a116211.
Rich-Edwards JW, Corsano KA, Stampfer MJ. Test of the National Death Index and Equifax Nationwide Death Search. Am J Epidemiol. 1994;140:1016–9. https://doi.org/10.1093/oxfordjournals.aje.a117191.
Cain KC, Harlow SD, Little RJ, Nan B, Yosef M, Taffe JR, et al. Bias due to left truncation and left censoring in longitudinal studies of developmental and disease processes. Am J Epidemiol. 2011;173:1078–84. https://doi.org/10.1093/aje/kwq481.
Giovannucci E, Liu Y, Platz EA, Stampfer MJ, Willett WC. Risk factors for prostate cancer incidence and progression in the health professionals follow-up study. Int J Cancer. 2007;121:1571–8. https://doi.org/10.1002/ijc.22788.
Zhang Y, Stopsack KH, Wu K, Song M, Mucci LA, Giovannucci E Post-diagnostic Zinc Supplement Use and Prostate Cancer Survival Among Men With Nonmetastatic Prostate Cancer. J Urol. 2022:101097JU0000000000003080. https://doi.org/10.1097/JU.0000000000003080.
Kenfield SA, Van Blarigan EL, DuPre N, Stampfer MJ, L Giovannucci E, Chan JM. Selenium supplementation and prostate cancer mortality. J Natl Cancer Inst. 2015;107:360 https://doi.org/10.1093/jnci/dju360.
Ladas EJ, Jacobson JS, Kennedy DD, Teel K, Fleischauer A, Kelly KM. Antioxidants and cancer therapy: a systematic review. J Clin Oncol. 2004;22:517–28. https://doi.org/10.1200/JCO.2004.03.086.
Margalit DN, Kasperzyk JL, Martin NE, Sesso HD, Gaziano JM, Ma J, et al. Beta-carotene antioxidant use during radiation therapy and prostate cancer outcome in the Physicians’ Health Study. Int J Radiat Oncol Biol Phys. 2012;83:28–32. https://doi.org/10.1016/j.ijrobp.2011.05.032.
Bailey RL, Gahche JJ, Miller PE, Thomas PR, Dwyer JT. Why US adults use dietary supplements. JAMA Intern Med. 2013;173:355–61. https://doi.org/10.1001/jamainternmed.2013.2299.
Macpherson H, Pipingas A, Pase MP. Multivitamin-multimineral supplementation and mortality: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2013;97:437–44. https://doi.org/10.3945/ajcn.112.049304.
Davies AA, Davey Smith G, Harbord R, Bekkering GE, Sterne JA, Beynon R, et al. Nutritional interventions and outcome in patients with cancer or preinvasive lesions: systematic review. J Natl Cancer Inst. 2006;98:961–73. https://doi.org/10.1093/jnci/djj263.
Saquib J, Rock CL, Natarajan L, Saquib N, Newman VA, Patterson RE, et al. Dietary intake, supplement use, and survival among women diagnosed with early-stage breast cancer. Nutr Cancer. 2011;63:327–33. https://doi.org/10.1080/01635581.2011.535957.
Kwan ML, Greenlee H, Lee VS, Castillo A, Gunderson EP, Habel LA, et al. Multivitamin use and breast cancer outcomes in women with early-stage breast cancer: the life after cancer epidemiology study. Breast Cancer Res Treat. 2011;130:195–205. https://doi.org/10.1007/s10549-011-1557-4.
Ng K, Meyerhardt JA, Chan JA, Niedzwiecki D, Hollis DR, Saltz LB, et al. Multivitamin use is not associated with cancer recurrence or survival in patients with stage III colon cancer: findings from CALGB 89803. J Clin Oncol. 2010;28:4354–63. https://doi.org/10.1200/JCO.2010.28.0362.
Zuniga KB, Chan JM, Ryan CJ, Kenfield SA. Diet and lifestyle considerations for patients with prostate cancer. Urol Oncol. 2020;38:105–17. https://doi.org/10.1016/j.urolonc.2019.06.018.
Wang P, Giovannucci EL. Are exposure-disease relationships assessed in cohorts of health professionals generalizable?: a comparative analysis based on WCRF/AICR systematic literature reviews. Cancer Causes Control. 2022; https://doi.org/10.1007/s10552-022-01633-3.
Acknowledgements
The authors would like to acknowledge the contribution to this study from central cancer registries supported through the Centers for Disease Control and Prevention’s National Program of Cancer Registries (NPCR) and/or the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) Program. Central registries may also be supported by state agencies, universities, and cancer centers. Participating central cancer registries include the following: Alabama, Alaska, Arizona, Arkansas, California, Colorado, Connecticut, Delaware, Florida, Georgia, Hawaii, Idaho, Indiana, Iowa, Kentucky, Louisiana, Massachusetts, Maine, Maryland, Michigan, Mississippi, Montana, Nebraska, Nevada, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, Puerto Rico, Rhode Island, Seattle SEER Registry, South Carolina, Tennessee, Texas, Utah, Virginia, West Virginia, Wyoming. We are grateful to the participants and research staff of the Health Professionals Follow-up Study and to the Channing Division of Network Medicine, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA.
Funding
The Health Professionals Follow-up Study is supported by the National Cancer Institute (U01 CA167552 and administrative supplements). KHS and LAM are Prostate Cancer Foundation Young Investigators. EG is funded as an American Cancer Society Clinical Research Professor (grant CRP-23-1014041).
Author information
Authors and Affiliations
Contributions
YZ had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study conception and design: YZ and EG Acquisition of data: YZ, LM and EG Statistical analysis: YZ, KS and EG. Interpretation of data: all authors. Drafting of the paper: YZ Critical revision of the paper for important intellectual content: all authors. Administrative, technical, or material support: none. Supervision: EG.
Corresponding author
Ethics declarations
Competing interests
LAM received research funding from Astra Zeneca and Janssen; was a consultant to Bayer; serves on the SAB and has equity in Convergent Therapeutics. None of these activities were related to the work under review.
Ethics approval and consent to participate
Our study was approved by the institutional review boards of Harvard T.H. Chan School of Public Health, and those of participating registries as required. Completion of the questionnaire was considered to imply informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Stopsack, K.H., Wu, K. et al. Multivitamin use after diagnosis and prostate cancer survival among men with nonmetastatic prostate cancer. Br J Cancer (2024). https://doi.org/10.1038/s41416-024-02651-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41416-024-02651-5