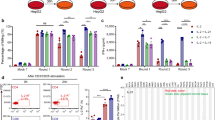
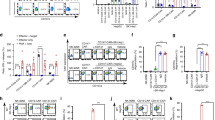
Abstract
Chimeric antigen receptor-expressing T (CAR-T) cells induce robust antitumor responses in patients with hematologic malignancies. However, CAR-T cells exhibit only limited efficacy against solid tumors such as hepatocellular carcinoma (HCC), partially due to their limited expansion and persistence. CD8+ T cells, as key components of the adaptive immune response, play a central role in antitumor immunity. Aerobic glycolysis is the main metabolic feature of activated CD8+ T cells. In the tumor microenvironment, however, the uptake of large amounts of glucose by tumor cells and other immunosuppressive cells can impair the activation of T cells. Only when tumor-infiltrating lymphocytes (TILs) in the tumor microenvironment have a glycolytic advantage might the effector function of T cells be activated. Glucose transporter type 1 (GLUT1) and acylglycerol kinase (AGK) can boost glycolytic metabolism and activate the effector function of CD8+ T cells, respectively. In this study, we generated GPC3-targeted CAR-T cells overexpressing GLUT1 or AGK for the treatment of HCC. GPC3-targeted CAR-T cells overexpressing GLUT1 or AGK specifically and effectively lysed GPC3-positive tumor cells in vitro in an antigen-dependent manner. Furthermore, GLUT1 or AGK overexpression protected CAR-T cells from apoptosis during repeated exposures to tumor cells. Compared with second-generation CAR-T cells, GPC3-targeted CAR-T cells overexpressing GLUT1 or AGK exhibited greater CD8+ T-cell persistence in vivo and better antitumor effects in HCC allograft mouse models. Finally, we revealed that GLUT1 or AGK maintained anti-apoptosis ability in CD8+ T cells via activation of the PI3K/Akt pathway. This finding might identify a therapeutic strategy for advanced HCC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The human sequence data generated in this study are not publicly available due to privacy requirements but are available upon reasonable request from the corresponding author. Other data generated in this study are available within the article and its supplementary data files.
References
Shi D, Shi Y, Kaseb AO, Qi X, Zhang Y, Chi J, et al. Chimeric antigen receptor-glypican-3 T-cell therapy for advanced hepatocellular carcinoma: results of phase I trials. Clin Cancer Res. 2020;26:3979–89.
Singh N, Orlando E, Xu J, Xu J, Binder Z, Collins MA, et al. Mechanisms of resistance to CAR T cell therapies. Semin Cancer Biol. 2020;65:91–8.
Li D, Li N, Zhang YF, Fu H, Feng M, Schneider D, et al. Persistent polyfunctional chimeric antigen receptor T cells that target glypican 3 eliminate orthotopic hepatocellular carcinomas in mice. Gastroenterology. 2020;158:2250–65.e2220.
Ruixin S, Yansha S, Chuanlong W, Yifan L, Min Z, Yiwei D, et al. Expressing CXCR4 in CAR-T cells suppresses MDSCs recruitment via STAT3/NF-kappaB/SDF-1alpha axis to enhance anti-tumor efficacy against pancreatic cancer. Mol Ther. 2023;31:3193–209.
Filmus J, Capurro M. Glypican-3: a marker and a therapeutic target in hepatocellular carcinoma. FEBS J. 2013;280:2471–6.
Gao H, Li K, Tu H, Pan X, Jiang H, Shi B, et al. Development of T cells redirected to glypican-3 for the treatment of hepatocellular carcinoma. Clin Cancer Res. 2014;20:6418–28.
Louis CU, Savoldo B, Dotti G, Pule M, Yvon E, Myers GD, et al. Antitumor activity and long-term fate of chimeric antigen receptor-positive T cells in patients with neuroblastoma. Blood. 2011;118:6050–6.
Ahmed N, Brawley VS, Hegde M, Robertson C, Ghazi A, Gerken C, et al. Human epidermal growth factor receptor 2 (HER2) -specific chimeric antigen receptor-modified T cells for the immunotherapy of HER2-positive sarcoma. J Clin Oncol. 2015;33:1688–96.
Ruixin S, Yifan L, Chuanlong W, Min Z, Hong L, Guoxiu D, et al. Expressing IL-15/IL-18 and CXCR2 improve infiltration and survival of EGFRvIII-targeting CAR-T cells in breast cancer. Biochem Pharmacol. 2023;212:115536.
Luo H, Su J, Sun R, Sun Y, Wang Y, Dong Y, et al. Coexpression of IL7 and CCL21 increases efficacy of CAR-T cells in solid tumors without requiring preconditioned lymphodepletion. Clin Cancer Res. 2020;26:5494–505.
Mishra D, Banerjee D. Metabolic interactions between tumor and stromal cells in the tumor microenvironment. Adv Exp Med Biol. 2021;1350:101–21.
Ou X, Lv W. Metabolic changes and interaction of tumor cell, myeloid-derived suppressor cell and T cell in hypoxic microenvironment. Fut Oncol. 2020;16:383–93.
Li S, Li J, Dai W, Zhang Q, Feng J, Wu L, et al. Genistein suppresses aerobic glycolysis and induces hepatocellular carcinoma cell death. Br J Cancer. 2017;117:1518–28.
Qiu J, Villa M, Sanin DE, Buck MD, O’Sullivan D, Ching R, et al. Acetate promotes T cell effector function during glucose restriction. Cell Rep. 2019;27:2063–74.e2065.
Chang CH, Curtis JD, Maggi LB Jr, Faubert B, Villarino AV, O’Sullivan D, et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell. 2013;153:1239–51.
Xu K, Yin N, Peng M, Stamatiades EG, Shyu A, Li P, et al. Glycolysis fuels phosphoinositide 3-kinase signaling to bolster T cell immunity. Science. 2021;371:405–10.
Cascone T, McKenzie JA, Mbofung RM, Punt S, Wang Z, Xu C, et al. Increased tumor glycolysis characterizes immune resistance to adoptive T cell therapy. Cell Metab. 2018;27:977–87 e974.
Jin R, Hao J, Yu J, Wang P, Sauter ER, Li B. Role of FABP5 in T cell lipid metabolism and function in the tumor microenvironment. Cancers (Basel). 2023;15:657.
Singh L, Nair L, Kumar D, Arora MK, Bajaj S, Gadewar M, et al. Hypoxia induced lactate acidosis modulates tumor microenvironment and lipid reprogramming to sustain the cancer cell survival. Front Oncol. 2023;13:1034205.
Li H, Zhao A, Li M, Shi L, Han Q, Hou Z. Targeting T-cell metabolism to boost immune checkpoint inhibitor therapy. Front Immunol. 2022;13:1046755.
O’Sullivan D, Pearce EL. Targeting T cell metabolism for therapy. Trends Immunol. 2015;36:71–80.
Palacio-Castaneda V, Kooijman L, Venzac B, Verdurmen WPR, Le Gac S. Metabolic switching of tumor cells under hypoxic conditions in a tumor-on-a-chip model. Micromachines (Basel). 2020;11:382.
Reinfeld BI, Madden MZ, Wolf MM, Chytil A, Bader JE, Patterson AR, et al. Cell-programmed nutrient partitioning in the tumour microenvironment. Nature. 2021;593:282–8.
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012;2:401–4.
Yoshihara K, Shahmoradgoli M, Martinez E, Vegesna R, Kim H, Torres-Garcia W, et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat Commun. 2013;4:2612.
Ge W, Tan SJ, Wang SH, Li L, Sun XF, Shen W, et al. Single-cell transcriptome profiling reveals dermal and epithelial cell fate decisions during embryonic hair follicle development. Theranostics. 2020;10:7581–98.
Zeng D, Ye Z, Shen R, Yu G, Wu J, Xiong Y, et al. IOBR: multi-omics immuno-oncology biological research to decode tumor microenvironment and signatures. Front Immunol. 2021;12:687975.
Wang Y, Zhang H, Du G, Luo H, Su J, Sun Y, et al. Enforced expression of Runx3 improved CAR-T cell potency in solid tumor via enhancing resistance to activation-induced cell death. Mol Ther. 2023;31:701–14.
Watson MJ, Vignali PDA, Mullett SJ, Overacre-Delgoffe AE, Peralta RM, Grebinoski S, et al. Metabolic support of tumour-infiltrating regulatory T cells by lactic acid. Nature. 2021;591:645–51.
Sun Y, Dong Y, Sun R, Liu Y, Wang Y, Luo H, et al. Chimeric anti-GPC3 sFv-CD3epsilon receptor-modified T cells with IL7 co-expression for the treatment of solid tumors,. Mol Ther Oncolytics. 2022;25:160–73.
Hu Z, Qu G, Yu X, Jiang H, Teng XL, Ding L, et al. Acylglycerol kinase maintains metabolic state and immune responses of CD8+ T cells. Cell Metab. 2019;30:290–302.e295.
Commander R, Wei C, Sharma A, Mouw JK, Burton LJ, Summerbell E, et al. Subpopulation targeting of pyruvate dehydrogenase and GLUT1 decouples metabolic heterogeneity during collective cancer cell invasion. Nat Commun. 2020;11:1533.
Tao J, McCourt C, Sultana H, Nelson C, Driver J, Hackmann TJ. Use of a fluorescent analog of glucose (2-NBDG) to identify uncultured rumen bacteria that take up glucose. Appl Environ Microbiol. 2019;85:e03018–18.
Manaharan T, Ming CH, Palanisamy UD. Syzygium aqueum leaf extract and its bioactive compounds enhances pre-adipocyte differentiation and 2-NBDG uptake in 3T3-L1 cells. Food Chem. 2013;136:354–63.
Zou C, Wang Y, Shen Z. 2-NBDG as a fluorescent indicator for direct glucose uptake measurement. J Biochem Biophys Methods. 2005;64:207–15.
Macintyre AN, Gerriets VA, Nichols AG, Michalek RD, Rudolph MC, Deoliveira D, et al. The glucose transporter Glut1 is selectively essential for CD4 T cell activation and effector function. Cell Metab. 2014;20:61–72.
Adachi K, Tamada K. Immune checkpoint blockade opens an avenue of cancer immunotherapy with a potent clinical efficacy. Cancer Sci. 2015;106:945–50.
Kumagai S, Koyama S, Itahashi K, Tanegashima T, Lin YT, Togashi Y, et al. Lactic acid promotes PD-1 expression in regulatory T cells in highly glycolytic tumor microenvironments. Cancer Cell. 2022;40:201–18.e209.
Li W, Tanikawa T, Kryczek I, Xia H, Li G, Wu K, et al. Aerobic glycolysis controls myeloid-derived suppressor cells and tumor immunity via a specific CEBPB isoform in triple-negative breast cancer. Cell Metab. 2018;28:87–103 e106.
Hong H, Xu G, Chen J, Zhang J, Chen C, Wu C, et al. LncRNA RMRP contributes to the development and progression of spinal cord injury by regulating miR-766-5p/FAM83A axis. Mol Neurobiol. 2022;59:6200–10.
Anderson KG, Stromnes IM, Greenberg PD. Obstacles posed by the tumor microenvironment to T cell activity: a case for synergistic therapies. Cancer Cell. 2017;31:311–25.
Kugelberg E. Innate lymphoid cells: nutrients direct immune balance. Nat Rev Immunol. 2014;14:137.
Chang CH, Pearce EL. Emerging concepts of T cell metabolism as a target of immunotherapy. Nat Immunol. 2016;17:364–8.
Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science. 2009;324:1029–33.
Liao W, Lin JX, Leonard WJ. Interleukin-2 at the crossroads of effector responses, tolerance, and immunotherapy. Immunity. 2013;38:13–25.
Ancey PB, Contat C, Meylan E. Glucose transporters in cancer—from tumor cells to the tumor microenvironment. FEBS J. 2018;285:2926–43.
Martinez-Outschoorn UE, Peiris-Pages M, Pestell RG, Sotgia F, Lisanti MP. Cancer metabolism: a therapeutic perspective. Nat Rev Clin Oncol. 2017;14:11–31.
Cohen S, Danzaki K, MacIver NJ. Nutritional effects on T-cell immunometabolism. Eur J Immunol. 2017;47:225–35.
Yao LX, Liu J, Xu L. MiR-610 functions as a tumor suppressor in oral squamous cell carcinoma by directly targeting AGK. Eur Rev Med Pharmacol Sci. 2019;23:187–97.
Zhou P, Chi H. AGK unleashes CD8(+) T cell glycolysis to combat tumor growth. Cell Metab. 2019;30:233–4.
Matsuura K, Canfield K, Feng W, Kurokawa M. Metabolic regulation of apoptosis in cancer. Int Rev Cell Mol Biol. 2016;327:43–87.
Kilic M, Kasperczyk H, Fulda S, Debatin KM. Role of hypoxia inducible factor-1 alpha in modulation of apoptosis resistance. Oncogene. 2007;26:2027–38.
Li C, Zhang G, Zhao L, Ma Z, Chen H. Metabolic reprogramming in cancer cells: glycolysis, glutaminolysis, and Bcl-2 proteins as novel therapeutic targets for cancer. World J Surg Oncol. 2016;14:15.
Mason EF, Zhao Y, Goraksha-Hicks P, Coloff JL, Gannon H, Jones SN, et al. Aerobic glycolysis suppresses p53 activity to provide selective protection from apoptosis upon loss of growth signals or inhibition of BCR-Abl. Cancer Res. 2010;70:8066–76.
Schiliro C, Firestein BL. Mechanisms of metabolic reprogramming in cancer cells supporting enhanced growth and proliferation. Cells. 2021;10:1056.
DeFrancesco L. CAR-T cell therapy seeks strategies to harness cytokine storm. Nat Biotechnol. 2014;32:604.
Chmielewski M, Abken H. CAR T cells transform to trucks: chimeric antigen receptor-redirected T cells engineered to deliver inducible IL-12 modulate the tumour stroma to combat cancer. Cancer Immunol Immunother. 2012;61:1269–77.
Hu B, Ren J, Luo Y, Keith B, Young RM, Scholler J, et al. Augmentation of antitumor immunity by human and mouse CAR T cells secreting IL-18. Cell Rep. 2017;20:3025–33.
Lanitis E, Rota G, Kosti P, Ronet C, Spill A, Seijo B, et al. Optimized gene engineering of murine CAR-T cells reveals the beneficial effects of IL-15 coexpression. J Exp Med. 2021;218:e20192203.
Du D, Liu C, Qin M, Zhang X, Xi T, Yuan S, et al. Metabolic dysregulation and emerging therapeutical targets for hepatocellular carcinoma. Acta Pharm Sin B. 2022;12:558–80.
van Bruggen JAC, Martens AWJ, Fraietta JA, Hofland T, Tonino SH, Eldering E, et al. Chronic lymphocytic leukemia cells impair mitochondrial fitness in CD8+ T cells and impede CAR T-cell efficacy. Blood. 2019;134:44–58.
Acknowledgements
Funding
This study was supported by the National Natural Science Foundation of China (ID 82073358 to Hua Jiang, ID 82073359 to Zonghai Li, 82272920 to Min Zhou), the Shanghai Municipal Health and Health Commission (ID 20234Y0193 to Sun Ruixin), the Shanghai Oriental Talent Youth Project (to Sun Ruixin), the Shanghai “Rising Stars of Medical Talents” Youth Development Program-Clinical Laboratory Practitioner Program (ID 2023-JY to Sun Ruixin) and the “Gan Quan Xin Xing” talent training program of Shanghai Tongji Hospital (HRBC 2307).
Author information
Authors and Affiliations
Contributions
SRX, JH, and LZH designed the study. RXS, LYF, SYS, WY, and SBZ performed the experiments and analyzed the data. SRX and LYF performed the animal experiments. SRX wrote the paper. JH and LZH reviewed and edited the manuscript. SRX, JH, ZM, and LZH supervised the project.
Corresponding authors
Ethics declarations
Competing interests
Dr. Zonghai Li has ownership interests in CAR-T cells related to this work. The remaining authors declare no competing interests.
Ethics approval and consent to participate
The Ethics Committee of Renji Hospital, Shanghai Jiaotong University School of Medicine (RJ2022-1115) approved this study. All animal experiments were performed according to protocols approved by the Shanghai Cancer Institute Experimental Animal Care Commission.
Additional information
Consent for publication Not applicable.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, Rx., Liu, Yf., Sun, Ys. et al. GPC3-targeted CAR-T cells expressing GLUT1 or AGK exhibit enhanced antitumor activity against hepatocellular carcinoma. Acta Pharmacol Sin (2024). https://doi.org/10.1038/s41401-024-01287-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41401-024-01287-8