Abstract
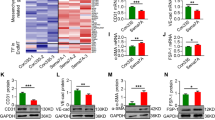
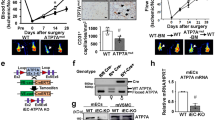
Hypertension is a prominent contributor to vascular injury. Deubiquinatase has been implicated in the regulation of hypertension-induced vascular injury. In the present study we investigated the specific role of deubiquinatase YOD1 in hypertension-induced vascular injury. Vascular endothelial endothelial-mesenchymal transition (EndMT) was induced in male WT and YOD1−/− mice by administration of Ang II (1 μg/kg per minute) via osmotic pump for four weeks. We showed a significantly increased expression of YOD1 in mouse vascular endothelial cells upon Ang II stimulation. Knockout of YOD1 resulted in a notable reduction in EndMT in vascular endothelial cells of Ang II-treated mouse; a similar result was observed in Ang II-treated human umbilical vein endothelial cells (HUVECs). We then conducted LC-MS/MS and co-immunoprecipitation (Co-IP) analyses to verify the binding between YOD1 and EndMT-related proteins, and found that YOD1 directly bound to β-catenin in HUVECs via its ovarian tumor-associated protease (OTU) domain, and histidine at 262 performing deubiquitination to maintain β-catenin protein stability by removing the K48 ubiquitin chain from β-catenin and preventing its proteasome degradation, thereby promoting EndMT of vascular endothelial cells. Oral administration of β-catenin inhibitor MSAB (20 mg/kg, every other day for four weeks) eliminated the protective effect of YOD1 deletion on vascular endothelial injury. In conclusion, we demonstrate a new YOD1-β-catenin axis in regulating Ang II-induced vascular endothelial injury and reveal YOD1 as a deubiquitinating enzyme for β-catenin, suggesting that targeting YOD1 holds promise as a potential therapeutic strategy for treating β-catenin-mediated vascular diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Mills KT, Stefanescu A, He J. The global epidemiology of hypertension. Nat Rev Nephrol. 2020;16:223–37.
Mills KT, Bundy JD, Kelly TN, Reed JE, Kearney PM, Reynolds K, et al. Global disparities of hypertension prevalence and control: A systematic analysis of population-based studies from 90 countries. Circulation. 2016;134:441–50.
Gerdts E, Sudano I, Brouwers S, Borghi C, Bruno RM, Ceconi C, et al. Sex differences in arterial hypertension. Eur Heart J. 2022;43:4777–88.
Harrison DG, Coffman TM, Wilcox CS. Pathophysiology of hypertension: The mosaic theory and beyond. Circ Res. 2021;128:847–63.
Eguchi S, Sparks MA, Sawada H, Lu HS, Daugherty A, Zhuo JL. Recent advances in understanding the molecular pathophysiology of angiotensin ii receptors; lessons from cell-selective receptor deletion in mice. Can J Cardiol. 2023;39:1795–807.
Forrester SJ, Booz GW, Sigmund CD, Coffman TM, Kawai T, Rizzo V, et al. Angiotensin ii signal transduction: An update on mechanisms of physiology and pathophysiology. Physiol Rev. 2018;98:1627–738.
Pan X, Shao Y, Wu F, Wang Y, Xiong R, Zheng J, et al. Fgf21 prevents angiotensin ii-induced hypertension and vascular dysfunction by activation of ace2/angiotensin-(1-7) axis in mice. Cell Metab. 2018;27:1323–37. e1325.
Xu S, Ilyas I, Little PJ, Li H, Kamato D, Zheng X, et al. Endothelial dysfunction in atherosclerotic cardiovascular diseases and beyond: From mechanism to pharmacotherapies. Pharmacol Rev. 2021;73:924–67.
Testai L, Brancaleone V, Flori L, Montanaro R, Calderone V. Modulation of endmt by hydrogen sulfide in the prevention of cardiovascular fibrosis. Antioxidants. 2021;10:910.
Markwald RR, Fitzharris TP, Manasek FJ. Structural development of endocardial cushions. Am J Anat. 1977;148:85–119.
Pan JA, Zhang H, Lin H, Gao L, Zhang HL, Zhang JF, et al. Irisin ameliorates doxorubicin-induced cardiac perivascular fibrosis through inhibiting endothelial-to-mesenchymal transition by regulating ros accumulation and autophagy disorder in endothelial cells. Redox Biol. 2021;46:102120.
Liang G, Wang S, Shao J, Jin YJ, Xu L, Yan Y, et al. Tenascin-x mediates flow-induced suppression of endmt and atherosclerosis. Circ Res. 2022;130:1647–59.
Woo KV, Shen IY, Weinheimer CJ, Kovacs A, Nigro J, Lin CY, et al. Endothelial fgf signaling is protective in hypoxia-induced pulmonary hypertension. J Clin Invest. 2021;131:e141467.
Chang CJ, Lai YJ, Tung YC, Wu LS, Hsu LA, Tseng CN, et al. Osteopontin mediation of disturbed flow-induced endothelial mesenchymal transition through cd44 is a novel mechanism of neointimal hyperplasia in arteriovenous fistulae for hemodialysis access. Kidney Int. 2023;103:702–18.
Xiong J. To be endmt or not to be, that is the question in pulmonary hypertension. Protein Cell. 2015;6:547–50.
Tang RN, Lv LL, Zhang JD, Dai HY, Li Q, Zheng M, et al. Effects of angiotensin ii receptor blocker on myocardial endothelial-to-mesenchymal transition in diabetic rats. Int J Cardiol. 2013;162:92–99.
Lin K, Luo W, Yan J, Shen S, Shen Q, Wang J, et al. Tlr2 regulates angiotensin ii-induced vascular remodeling and endmt through nf-κb signaling. Aging. 2020;13:2553–74.
Souilhol C, Harmsen MC, Evans PC, Krenning G. Endothelial-mesenchymal transition in atherosclerosis. Cardiovasc Res. 2018;114:565–77.
Piera-Velazquez S, Jimenez SA. Endothelial to mesenchymal transition: Role in physiology and in the pathogenesis of human diseases. Physiol Rev. 2019;99:1281–324.
Cai C, Tang YD, Zhai J, Zheng C. The ring finger protein family in health and disease. Signal Transduct Target Ther. 2022;7:300.
Ye B, Zhou H, Chen Y, Luo W, Lin W, Zhao Y, et al. Usp25 ameliorates pathological cardiac hypertrophy by stabilizing serca2a in cardiomyocytes. Circ Res. 2023;132:465–80.
Ernst R, Mueller B, Ploegh HL, Schlieker C. The otubain yod1 is a deubiquitinating enzyme that associates with p97 to facilitate protein dislocation from the er. Mol Cell. 2009;36:28–38.
Han Z, Jia Q, Zhang J, Chen M, Wang L, Tong K, et al. Deubiquitylase yod1 regulates cdk1 stability and drives triple-negative breast cancer tumorigenesis. J Exp Clin Cancer Res. 2023;42:228.
Shao X, Chen Y, Wang W, Du W, Zhang X, Cai M, et al. Blockade of deubiquitinase yod1 degrades oncogenic pml/rarα and eradicates acute promyelocytic leukemia cells. Acta Pharm Sin B. 2022;12:1856–70.
Zhang Z, Zhao W, Li Y, Li Y, Cheng H, Zheng L, et al. Yod1 serves as a potential prognostic biomarker for pancreatic cancer. Cancer Cell Int. 2022;22:203.
Wang JM, Chen AF, Zhang K. Isolation and primary culture of mouse aortic endothelial cells. J Vis Exp. 2016;118:52965.
Tripathi M, Singh BK, Liehn EA, Lim SY, Tikno K, Castano-Mayan D, et al. Caffeine prevents restenosis and inhibits vascular smooth muscle cell proliferation through the induction of autophagy. Autophagy. 2022;18:2150–60.
Rusanov AL, Kozhin PM, Tikhonova OV, Zgoda VG, Loginov DS, Chlastáková A, et al. Proteome profiling of pmj2-r and primary peritoneal macrophages. Int J Mol Sci. 2021;22:6323.
Chen J, Zhuang R, Cheng HS, Jamaiyar A, Assa C, McCoy M, et al. Isolation and culture of murine aortic cells and rna isolation of aortic intima and media: Rapid and optimized approaches for atherosclerosis research. Atherosclerosis. 2022;347:39–46.
Yang L, Dai R, Wu H, Cai Z, Xie N, Zhang X, et al. Unspliced xbp1 counteracts β-catenin to inhibit vascular calcification. Circ Res. 2022;130:213–29.
Pang K, Park J, Ahn SG, Lee J, Park Y, Ooshima A, et al. Rnf208, an estrogen-inducible e3 ligase, targets soluble vimentin to suppress metastasis in triple-negative breast cancers. Nat Commun. 2019;10:5805.
Wu Y, Wang Y, Lin Y, Liu Y, Wang Y, Jia J, et al. Dub3 inhibition suppresses breast cancer invasion and metastasis by promoting snail1 degradation. Nat Commun. 2017;8:14228.
Lin X, Li AM, Li YH, Luo RC, Zou YJ, Liu YY, et al. Silencing myh9 blocks hbx-induced gsk3β ubiquitination and degradation to inhibit tumor stemness in hepatocellular carcinoma. Signal Transduct Target Ther. 2020;5:13.
Nanes BA, Grimsley-Myers CM, Cadwell CM, Robinson BS, Lowery AM, Vincent PA, et al. P120-catenin regulates ve-cadherin endocytosis and degradation induced by the kaposi sarcoma-associated ubiquitin ligase k5. Mol Biol Cell. 2017;28:30–40.
Huang Z, Shen S, Wang M, Li W, Wu G, Huang W, et al. Mouse endothelial otud1 promotes angiotensin ii-induced vascular remodeling by deubiquitinating smad3. EMBO Rep. 2023;24:e56135.
Liu Y, Chen Y, Ding C, Zhu X, Song X, Ren Y, et al. Trim56 positively regulates tnfα-induced nf-κb signaling by enhancing the ubiquitination of tak1. Int J Biol Macromol. 2022;219:571–8.
Mennerich D, Kubaichuk K, Kietzmann T. Dubs, hypoxia, and cancer. Trends Cancer. 2019;5:632–53.
Nandakumar P, Lee D, Richard MA, Tekola-Ayele F, Tayo BO, Ware E, et al. Rare coding variants associated with blood pressure variation in 15 914 individuals of african ancestry. J Hypertens. 2017;35:1381–9.
Song IK, Kim HJ, Magesh V, Lee KJ. Ubiquitin c-terminal hydrolase-l1 plays a key role in angiogenesis by regulating hydrogen peroxide generated by nadph oxidase 4. Biochem Biophys Res Commun. 2018;495:1567–72.
He S, Zhong W, Yin L, Wang Y, Qiu Z, Song G. High expression of ubiquitin-specific peptidase 39 is associated with the development of vascular remodeling. Mol Med Rep. 2017;15:2567–73.
Tanji K, Mori F, Miki Y, Utsumi J, Sasaki H, Kakita A, et al. Yod1 attenuates neurogenic proteotoxicity through its deubiquitinating activity. Neurobiol Dis. 2018;112:14–23.
Wang S, Wang Y, Wang S, Tong H, Tang Z, Wang J, et al. Long non-coding rna firre acts as a mir-520a-3p sponge to promote gallbladder cancer progression via mediating yod1 expression. Front Genet. 2021;12:674653.
Li Q, Sun H, Liu S, Tang J, Liu S, Yin P, et al. Ginsenoside rk1 inhibits hela cell proliferation through an endoplasmic reticulum signaling pathway. J Ginseng Res. 2023;47:645–53.
Zhu YM, Chen P, Shi L, Zhu T, Chen X. Mir-4429 suppresses the malignant development of ovarian cancer by targeting yod1. Eur Rev Med Pharm Sci. 2020;24:8722–30.
Han J, Shi X, Du Y, Shi F, Zhang B, Zheng Z, et al. Schisandrin c targets keap1 and attenuates oxidative stress by activating nrf2 pathway in ang ii-challenged vascular endothelium. Phytother Res. 2019;33:779–90.
Yang HY, Bian YF, Zhang HP, Gao F, Xiao CS, Liang B, et al. Angiotensin-(1-7) treatment ameliorates angiotensin ii-induced apoptosis of human umbilical vein endothelial cells. Clin Exp Pharmacol Physiol. 2012;39:1004–10.
Barhoumi T, Fraulob-Aquino JC, Mian MOR, Ouerd S, Idris-Khodja N, Huo KG, et al. Matrix metalloproteinase-2 knockout prevents angiotensin ii-induced vascular injury. Cardiovasc Res. 2017;113:1753–62.
You S, Qian J, Wu G, Qian Y, Wang Z, Chen T, et al. Schizandrin b attenuates angiotensin ii induced endothelial to mesenchymal transition in vascular endothelium by suppressing nf-κb activation. Phytomedicine. 2019;62:152955.
Li L, Chen L, Zang J, Tang X, Liu Y, Zhang J, et al. C3a and c5a receptor antagonists ameliorate endothelial-myofibroblast transition via the wnt/β-catenin signaling pathway in diabetic kidney disease. Metabolism. 2015;64:597–610.
Zhu Y, Hart GW. Dual-specificity rna aptamers enable manipulation of target-specific o-glcnacylation and unveil functions of o-glcnac on β-catenin. Cell. 2023;186:428–45. e427
Liang S, Zhou Z, Zhou Z, Liang J, Lin W, Zhang C, et al. Blockade of cbx4-mediated β-catenin sumoylation attenuates airway epithelial barrier dysfunction in asthma. Int Immunopharmacol. 2022;113:109333.
You H, Li Q, Kong D, Liu X, Kong F, Zheng K, et al. The interaction of canonical wnt/β-catenin signaling with protein lysine acetylation. Cell Mol Biol Lett. 2022;27:7.
Chen X, Wang C, Jiang Y, Wang Q, Tao Y, Zhang H, et al. Bcl-3 promotes wnt signaling by maintaining the acetylation of β-catenin at lysine 49 in colorectal cancer. Signal Transduct Target Ther. 2020;5:52.
Dao KH, Rotelli MD, Petersen CL, Kaech S, Nelson WD, Yates JE, et al. Fancl ubiquitinates β-catenin and enhances its nuclear function. Blood. 2012;120:323–34.
Sabbineni H, Verma A, Artham S, Anderson D, Amaka O, Liu F, et al. Pharmacological inhibition of β-catenin prevents endmt in vitro and vascular remodeling in vivo resulting from endothelial akt1 suppression. Biochem Pharmacol. 2019;164:205–15.
Huo YB, Gao X, Peng Q, Nie Q, Bi W. Dihydroartemisinin alleviates angii-induced vascular smooth muscle cell proliferation and inflammatory response by blocking the fto/nr4a3 axis. Inflamm Res. 2022;71:243–53.
Kröller-Schön S, Jansen T, Tran TLP, Kvandová M, Kalinovic S, Oelze M, et al. Endothelial α1ampk modulates angiotensin ii-mediated vascular inflammation and dysfunction. Basic Res Cardiol. 2019;114:8.
Acknowledgements
We are grateful to Meng-xin Zhang, Wen-ting Wang and Jian-song Lin from the Scientific Research Center of Wenzhou Medical University for their help in immunofluorescence experiment. This study was supported by the National Natural Science Foundation of China (82271347 to GJW) and the Major project of Wenzhou Science and Technology Bureau (ZY2020016 to GJW).
Author information
Authors and Affiliations
Contributions
GL, WJH, and BZY contributed to the literature search and study design. WTL, YCJ, YLM, YHC, ZZZ, XH carried out the experiments. BZY contributed to data collection and analysis. WTL, BZY, and GJW participated in the drafting of the article. GL and WJH revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, Wt., Jiang, Yc., Mei, Yl. et al. Endothelial deubiquinatase YOD1 mediates Ang II-induced vascular endothelial-mesenchymal transition and remodeling by regulating β-catenin. Acta Pharmacol Sin (2024). https://doi.org/10.1038/s41401-024-01278-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41401-024-01278-9