Abstract
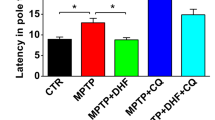
Parkinson’s disease (PD) is the second most common neurodegenerative disease, and its prevalence is increasing. Currently, no effective therapies for PD exist. Marine-derived natural compounds are considered important resources for the discovery of new drugs due to their distinctive structures and diverse activities. In this study, tetrahydroauroglaucin (TAG), a polyketide isolated from a marine sponge, was found to have notable neuroprotective effects on MPTP/MPP+-induced neurotoxicity. RNA sequencing analysis and metabolomics revealed that TAG significantly improved lipid metabolism disorder in PD models. Further investigation indicated that TAG markedly decreased the accumulation of lipid droplets (LDs), downregulated the expression of RUBCN, and promoted autophagic flux. Moreover, conditional knockdown of Rubcn notably attenuated PD-like symptoms and the accumulation of LDs, accompanied by blockade of the neuroprotective effect of TAG. Collectively, our results first indicated that TAG, a promising PD therapeutic candidate, could suppress the accumulation of LDs through the RUBCN-autophagy pathway, which highlighted a novel and effective strategy for PD treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Samii A, Nutt JG, Ransom BR. Parkinson’s disease. Lancet. 2004;363:1783–93.
Balestrino R, Schapira AHV. Parkinson disease. Eur J Neurol. 2020;27:27–42.
Shulman JM, De Jager PL, Feany MB. Parkinson’s disease: genetics and pathogenesis. Annu Rev Pathol. 2011;6:193–222.
Moustafa AA, Chakravarthy S, Phillips JR, Gupta A, Keri S, Polner B, et al. Motor symptoms in Parkinson’s disease: a unified framework. Neurosci Biobehav Rev. 2016;68:727–40.
Ko YF, Kuo PH, Wang CF, Chen YJ, Chuang PC, Li SZ, et al. Quantification analysis of sleep based on smartwatch sensors for Parkinson’s disease. Biosensors. 2022;12:74.
Schrag A, Horsfall L, Walters K, Noyce A, Petersen I. Prediagnostic presentations of Parkinson’s disease in primary care: a case-control study. Lancet Neurol. 2015;14:57–64.
Day JO, Mullin S. The genetics of Parkinson’s disease and implications for clinical practice. Genes. 2021;12:1006.
Barreto GE, Iarkov A, Moran VE. Beneficial effects of nicotine, cotinine and its metabolites as potential agents for Parkinson’s disease. Front Aging Neurosci. 2014;6:340.
Aryal B, Lee Y. Disease model organism for Parkinson disease: Drosophila melanogaster. BMB Rep. 2019;52:250–8.
Cacabelos R. Parkinson’s disease: from pathogenesis to pharmacogenomics. Int J Mol Sci. 2017;18:551.
Sagar S, Kaur M, Minneman KP. Antiviral lead compounds from marine sponges. Mar Drugs. 2010;8:2619–38.
Zhang B, Zhang T, Xu J, Lu J, Qiu P, Wang T, et al. Marine sponge-associated Fungi as potential novel bioactive natural product sources for drug discovery: a review. Mini Rev Med Chem. 2020;20:1966–2010.
Lin Z, Phadke S, Lu Z, Beyhan S, Abdel Aziz MH, Reilly C, et al. Onydecalins, fungal polyketides with anti- Histoplasma and anti-TRP activity. J Nat Prod. 2018;81:2605–11.
Shah M, Sun C, Sun Z, Zhang G, Che Q, Gu Q, et al. Antibacterial polyketides from Antarctica sponge-derived fungus Penicillium sp. HDN151272. Mar Drugs. 2020;18:71.
Liu Z, Qiu P, Liu H, Li J, Shao C, Yan T, et al. Identification of anti-inflammatory polyketides from the coral-derived fungus Penicillium sclerotiorin: In vitro approaches and molecular-modeling. Bioorg Chem. 2019;88:102973.
Zhang Q, Su G, Yang Q, Wei Z, Wang J, Zheng L, et al. Round scad-derived octapeptide WCPFSRSF confers neuroprotection by regulating Akt/Nrf2/NFκB signaling. J Agric Food Chem. 2021;69:10606–16.
Catanesi M, Caioni G, Castelli V, Benedetti E, d’Angelo M, Cimini A. Benefits under the sea: the role of marine compounds in neurodegenerative disorders. Mar Drugs. 2021;19:24.
Silva J, Alves C, Pinteus S, Susano P, Simões M, Guedes M, et al. Disclosing the potential of eleganolone for Parkinson’s disease therapeutics: Neuroprotective and anti-inflammatory activities. Pharmacol Res. 2021;168:105589.
Kolesnikova SA, Lyakhova EG, Kalinovsky AI, Popov RS, Yurchenko EA, Stonik VA. Oxysterols from a marine sponge inflatella sp. and their action in 6-hydroxydopamine-induced cell model of Parkinson’s disease. Mar Drugs. 2018;16:458.
Letsiou S, Bakea A, Goff GL, Lopes P, Gardikis K, Weis M, et al. Marine fungus Aspergillus chevalieri TM2-S6 extract protects skin fibroblasts from oxidative stress. Mar Drugs. 2020;18:460.
Stern JH, Rutkowski JM, Scherer PE. Adiponectin, leptin, and fatty acids in the maintenance of metabolic homeostasis through adipose tissue crosstalk. Cell Metab. 2016;23:770–84.
Xu J, Huang X. Lipid metabolism at membrane contacts: dynamics and functions beyond lipid homeostasis. Front Cell Dev Biol. 2020;8:615856.
Tong Y, Sun Y, Tian X, Zhou T, Wang H, Zhang T, et al. Phospholipid transfer protein (PLTP) deficiency accelerates memory dysfunction through altering amyloid precursor protein (APP) processing in a mouse model of Alzheimer’s disease. Hum Mol Genet. 2015;24:5388–403.
Galper J, Kim WS, Dzamko N. LRRK2 and lipid pathways: implications for Parkinson’s disease. Biomolecules. 2022;12:1597.
Phillips GR, Hancock SE, Brown SHJ, Jenner AM, Kreilaus F, Newell KA, et al. Cholesteryl ester levels are elevated in the caudate and putamen of Huntington’s disease patients. Sci Rep. 2020;10:20314.
Xie J, Duan L, Qian X, Huang X, Ding J, Hu G. K(ATP) channel openers protect mesencephalic neurons against MPP+-induced cytotoxicity via inhibition of ROS production. J Neurosci Res. 2010;88:428–37.
Wei Y, Lu M, Mei M, Wang H, Han Z, Chen M, et al. Pyridoxine induces glutathione synthesis via PKM2-mediated Nrf2 transactivation and confers neuroprotection. Nat Commun. 2020;11:941.
Ye H, Robak LA, Yu M, Cykowski M, Shulman JM. Genetics and pathogenesis of Parkinson’s syndrome. Annu Rev Pathol. 2023;18:95–121.
Nicholatos JW, Francisco AB, Bender CA, Yeh T, Lugay FJ, Salazar JE, et al. Nicotine promotes neuron survival and partially protects from Parkinson’s disease by suppressing SIRT6. Acta Neuropathol Commun. 2018;6:120.
Davis S, Meltzer PS. GEOquery: a bridge between the Gene Expression Omnibus (GEO) and BioConductor. Bioinformatics. 2007;23:1846–7.
Lei Y, Xu X, Liu H, Chen L, Zhou H, Jiang J, et al. HBx induces hepatocellular carcinogenesis through ARRB1-mediated autophagy to drive the G1/S cycle. Autophagy. 2021;17:4423–41.
Ma X, McKeen T, Zhang J, Ding WX. Role and mechanisms of mitophagy in liver diseases. Cells. 2020;9:837.
Feng Y, Ning X, Wang J, Wen Z, Cao F, You Q, et al. Mace-like plasmonic Au-Pd heterostructures boost near-infrared photoimmunotherapy. Adv Sci. 2023;10:e2204842.
Guo JD, Zhao X, Li Y, Li GR, Liu XL. Damage to dopaminergic neurons by oxidative stress in Parkinson’s disease. Int J Mol Med. 2018;41:1817–25.
Sewastianik T, Straubhaar JR, Zhao JJ, Samur MK, Adler K, Tanton HE, et al. miR-15a/16-1 deletion in activated B cells promotes plasma cell and mature B-cell neoplasms. Blood. 2021;137:1905–19.
Castillo-Quan JI, Steinbaugh MJ, Fernández-Cárdenas LP, Pohl NK, Wu Z, Zhu F, et al. An antisteatosis response regulated by oleic acid through lipid droplet-mediated ERAD enhancement. Sci Adv. 2023;9:eadc8917.
Bezawork-Geleta A, Dimou J, Watt MJ. Lipid droplets and ferroptosis as new players in brain cancer glioblastoma progression and therapeutic resistance. Front Oncol. 2022;12:1085034.
Consortium TM. Single-cell transcriptomics of 20 mouse organs creates a Tabula Muris. Nature. 2018;562:367–72.
Wong E, Cuervo AM. Autophagy gone awry in neurodegenerative diseases. Nat Neurosci. 2010;13:805–11.
Raudenska M, Balvan J, Masarik M. Crosstalk between autophagy inhibitors and endosome-related secretory pathways: a challenge for autophagy-based treatment of solid cancers. Mol Cancer. 2021;20:140.
Nakamura S, Yoshimori T. New insights into autophagosome-lysosome fusion. J Cell Sci. 2017;130:1209–16.
Wang X, Zhang X, Chu ESH, Chen X, Kang W, Wu F, et al. Defective lysosomal clearance of autophagosomes and its clinical implications in nonalcoholic steatohepatitis. FASEB J. 2018;32:37–51.
Tian X, Zhang G, Shao Y, Yang Z. Towards enhanced metabolomic data analysis of mass spectrometry image: multivariate curve resolution and machine learning. Anal Chim Acta. 2018;1037:211–9.
Castellanos DB, Martín-Jiménez CA, Rojas-Rodríguez F, Barreto GE, González J. Brain lipidomics as a rising field in neurodegenerative contexts: perspectives with machine learning approaches. Front Neuroendocrinol. 2021;61:100899.
Fahy E, Subramaniam S, Brown HA, Glass CK, Merrill AH, Murphy RC, et al. A comprehensive classification system for lipids. J Lipid Res. 2005;46:839–61.
Farooqui AA. Lipid mediators in the neural cell nucleus: their metabolism, signaling, and association with neurological disorders. Neuroscientist. 2009;15:392–407.
Shamim A, Mahmood T, Ahsan F, Kumar A, Bagga P. Lipids: an insight into the neurodegenerative disorders. Clin Nutr Exp. 2018;20:1–19.
Farese RV, Walther TC. Lipid droplets finally get a little R-E-S-P-E-C-T. Cell. 2009;139:855–60.
Liu L, Zhang K, Sandoval H, Yamamoto S, Jaiswal M, Sanz E, et al. Glial lipid droplets and ROS induced by mitochondrial defects promote neurodegeneration. Cell. 2015;160:177–90.
Marschallinger J, Iram T, Zardeneta M, Lee SE, Lehallier B, Haney MS, et al. Lipid-droplet-accumulating microglia represent a dysfunctional and proinflammatory state in the aging brain. Nat Neurosci. 2020;23:194–208.
Guo Y, Walther TC, Rao M, Stuurman N, Goshima G, Terayama K, et al. Functional genomic screen reveals genes involved in lipid-droplet formation and utilization. Nature. 2008;453:657–61.
Tanaka S, Hikita H, Tatsumi T, Sakamori R, Nozaki Y, Sakane S, et al. Rubicon inhibits autophagy and accelerates hepatocyte apoptosis and lipid accumulation in nonalcoholic fatty liver disease in mice. Hepatology. 2016;64:1994–2014.
Nakamura S, Oba M, Suzuki M, Takahashi A, Yamamuro T, Fujiwara M, et al. Suppression of autophagic activity by Rubicon is a signature of aging. Nat Commun. 2019;10:847.
Wong SW, Sil P, Martinez J. Rubicon: LC3-associated phagocytosis and beyond. FEBS J. 2018;285:1379–88.
Parzych KR, Klionsky DJ. An overview of autophagy: morphology, mechanism, and regulation. Antioxid Redox Signal. 2014;20:460–73.
Singh R, Kaushik S, Wang Y, Xiang Y, Novak I, Komatsu M, et al. Autophagy regulates lipid metabolism. Nature. 2009;458:1131–5.
Bae EJ, Lee HJ, Jang YH, Michael S, Masliah E, Min DS, et al. Phospholipase D1 regulates autophagic flux and clearance of α-synuclein aggregates. Cell Death Differ. 2014;21:1132–41.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 81991523, 82003725) and the National Key R&D Programme of China (No. 2021ZD0202901). We thank Prof. RXT from Nanjing University for supplying the marine-derived natural compounds.
Author information
Authors and Affiliations
Contributions
GH and RXT conceived and designed the study. The natural compounds were provided by ZWT and RXT. PY, YL, QHH, XHX and SYM performed the experiments and analysed the data. PY and YL wrote the manuscript. JHD provided technical support. GH and ML revised the manuscript. All the authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, P., Liu, Y., Tong, Zw. et al. The marine-derived compound TAG alleviates Parkinson’s disease by restoring RUBCN-mediated lipid metabolism homeostasis. Acta Pharmacol Sin (2024). https://doi.org/10.1038/s41401-024-01259-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41401-024-01259-y