Abstract
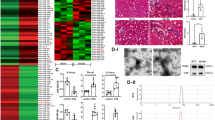
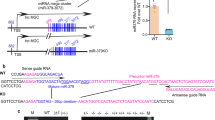
Renal tubulointerstitial fibrosis (TIF) is considered as the final convergent pathway of diabetic nephropathy (DN) without effective therapies currently. MiRNAs play a key role in fibrotic diseases and become promising therapeutic targets for kidney diseases, while miRNA clusters, formed by the cluster arrangement of miRNAs on chromosomes, can regulate diverse biological functions alone or synergistically. In this study, we developed clustered miR-23a/27a/26a-loaded skeletal muscle satellite cells–derived exosomes (Exos) engineered with RVG peptide, and investigated their therapeutic efficacy in a murine model of DN. Firstly, we showed that miR-23a-3p, miR-26a-5p and miR-27a-3p were markedly decreased in serum samples of DN patients using miRNA sequencing. Meanwhile, we confirmed that miR-23a-3p, miR-26a-5p and miR-27a-3p were primarily located in proximal renal tubules and highly negatively correlated with TIF in db/db mice at 20 weeks of age. We then engineered RVG-miR-23a/27a/26a cluster loaded Exos derived from muscle satellite cells, which not only enhanced the stability of miR-23a/27a/26a cluster, but also efficiently delivered more miR-23a/27a/26a cluster homing to the injured kidney. More importantly, administration of RVG-miR-23a/27a/26a-Exos (100 μg, i.v., once a week for 8 weeks) significantly ameliorated tubular injury and TIF in db/db mice at 20 weeks of age. We revealed that miR-23a/27a/26a-Exos enhanced antifibrotic effects by repressing miRNA cluster-targeting Lpp simultaneously, as well as miR-27a-3p-targeting Zbtb20 and miR-26a-5p-targeting Klhl42, respectively. Knockdown of Lpp by injection of AAV-Lpp-RNAi effectively ameliorated the progression of TIF in DN mice. Taken together, we established a novel kidney-targeting Exo-based delivery system by manipulating the miRNA-23a/27a/26a cluster to ameliorate TIF in DN, thus providing a promising therapeutic strategy for DN.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Xue R, Gui DK, Zheng LY, Zhai RN, Wang F, Wang NS. Mechanistic insight and management of diabetic nephropathy: recent progress and future perspective. J Diabetes Res. 2017;2017:1839809.
Mori Y, Ajay AK, Chang JH, Mou S, Zhao HP, Kishi SJ, et al. KIM-1 mediates fatty acid uptake by renal tubular cells to promote progressive diabetic kidney disease. Cell Metab. 2021;5:1042–61.
Zeni L, Norden AGW, Cancarini G, Unwin RJ. A more tubulocentric view of diabetic kidney disease. J Nephrol. 2017;30:701–17.
Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993;75:843–54.
Cerqueira DM, Tayeb M, Ho J. MicroRNAs in kidney development and disease. JCI Insight. 2022;7:e158277.
Wonnacott A, Denby L, Coward RJM, Fraser DJ, Bowen T. MicroRNAs and their delivery in diabetic fibrosis. Adv Drug Deliv Rev. 2022;182:114045.
Vinas JL, Spence M, Porter CJ, Douvris A, Gutsol A, Zimpelmann JA, et al. micro-RNA-486-5p protects against kidney ischemic injury and modifies the apoptotic transcriptome in proximal tubules. Kidney Int. 2021;100:597–612.
Bhaskaran V, Nowicki MO, Idriss M, Jimenez MA, Lugli G, Hayes JL, et al. The functional synergism of microRNA clustering provides therapeutically relevant epigenetic interference in glioblastoma. Nat Commun. 2019;10:442.
Diazzi S, Baeri A, Fassy J, Lecacheur M, Marin-Bejar O, Girard CA, et al. Blockade of the pro-fibrotic reaction mediated by the miR-143/145 cluster enhances the responses to targeted therapy in melanoma. EMBO Mol Med. 2022;14:e15295.
Nilsen A, Hillestad T, Skingen VE, Aarnes EK, Fjeldbo CS, Hompland T, et al. miR-200a/b/-429 downregulation is a candidate biomarker of tumor radioresistance and independent of hypoxia in locally advanced cervical cancer. Mol Oncol. 2022;16:1402–19.
Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367:eaau6977.
Zhang Y, Bi JY, Huang JY, Tang YN, Du SY, Li PY. Exosome: a review of its classification, isolation techniques, storage, diagnostic and targeted therapy applications. Int J Nanomed. 2020;15:6917–34.
Yim N, Ryu SW, Choi K, Lee KR, Lee S, Choi H, et al. Exosome engineering for efficient intracellular delivery of soluble proteins using optically reversible protein–protein interaction module. Nat Commun. 2016;7:12277.
Yang TZ, Martin P, Fogarty B, Brown A, Schurman K, Phipps R, et al. Exosome delivered anticancer drugs across the blood-brain barrier for brain cancer therapy in Danio rerio. Pharm Res. 2015;32:2003–14.
Tang TT, Wang B, Wu M, Li ZL, Feng Y, Cao JY, et al. Extracellular vesicle–encapsulated IL-10 as novel nanotherapeutics against ischemic AKI. Sci Adv. 2020;6:eaaz0748.
Kim S, Lee SA, Yoon H, Kim MY, Yoo JK, Ahn SH, et al. Exosome-based delivery of super-repressor IkBa ameliorates kidney ischemia-reperfusion injury. Kidney Int. 2021;100:570–84.
Wang B, Wang J, He W, Zhao YJ, Zhang AQ, Liu Y, et al. Exogenous miR-29a attenuates muscle atrophy and kidney fibrosis in unilateral ureteral obstruction mice. Hum Gene Ther. 2020;31:367–75.
Cao JY, Wang B, Tang TT, Wen Y, Li ZL, Feng ST, et al. Exosomal miR-125b-5p deriving from mesenchymal stem cells promotes tubular repair by suppression of p53 in ischemic acute kidney injury. Theranostics. 2021;11:5248–66.
An Y, Lin SY, Tan XJ, Zhu S, Nie FF, Zhen YH, et al. Exosomes from adipose-derived stem cells and application to skin wound healing. Cell Prolif. 2021;54:e12993.
Liu YJ, Guo Y, Bao SM, Huang HD, Liu WH, Guo WK. Bone marrow mesenchymal stem cell-derived exosomal microRNA-381-3p alleviates vascular calcification in chronic kidney disease by targeting NFAT5. Cell Death Dis. 2022;13:278.
Hu XM, Shen N, Liu AQ, Wang WD, Zhang LH, Sui ZG, et al. Bone marrow mesenchymal stem cell-derived exosomal miR-34c-5p ameliorates RIF by inhibiting the core fucosylation of multiple proteins. Mol Ther. 2022;30:763–81.
Sacco A, Doyonnas R, Kraft P, Vitorovic S, Blau HM. Self-renewal and expansion of single transplanted muscle stem cells. Nature. 2008;456:502–6.
Menasche P. Skeletal muscle satellite cell transplantation. Cardiovasc Res. 2003;58:351–7.
Wang HD, Wang B, Zhang AQ, Hassounah F, Seow YQ, Wood M, et al. Exosome-mediated miR-29 transfer reduces muscle atrophy and kidney fibrosis in mice. Mol Ther. 2019;27:571–83.
ADA. Chronic kidney disease and risk management: standards of medical care in diabetes-2022. Diabetes Care. 2022;45:175–84.
Lee SM, Bressler R. Prevention of diabetic nephropathy by diet control in the db/db mouse. Diabetes. 1981;30:106–11.
Sharma K, McCue P, Dunn SR. Diabetic kidney disease in the db/db mouse. Am J Physiol Ren Physiol. 2003;284:1138–44.
Luo CW, Zhou S, Zhou ZM, Liu YH, Yang L, Liu JF, et al. Wnt9a promotes renal fibrosis by accelerating cellular senescence in tubular epithelial cells. J Am Soc Nephrol. 2018;29:1238–56.
Qu GT, He TT, Dai AS, Zhao YJ, Guan D, Li SW, et al. miR‑199b‑5p mediates adriamycin‑induced podocyte apoptosis by inhibiting the expression of RGS10. Exp Ther Med. 2021;22:1469.
Zhang AQ, Wang HD, Wang B, Yuan YG, Klein JD, Wang XH. Exogenous miR-26a suppresses muscle wasting and renal fibrosis in obstructive kidney disease. FASEB J. 2019;33:13590–601.
Cui MY, Yao XX, Lin Y, Zhang D, Cui RJ, Zhang XW. Interactive functions of microRNAs in the miR-23a-27a-24-2 cluster and the potential for targeted therapy in cancer. J Cell Physiol. 2020;235:6–16.
Zhang AQ, Li M, Wang B, Klein JD, Price SR, Wang XH. miRNA-23a/27a attenuates muscle atrophy and renal fibrosis through muscle-kidney crosstalk. J Cachexia Sarcopenia Muscle. 2018;9:755–70.
Wang B, Zhang C, Zhang AQ, Cai H, Price SR, Wang XH. MicroRNA-23a and MicroRNA-27a mimic exercise by ameliorating CKD-Induced muscle atrophy. J Am Soc Nephrol. 2017;28:2631–40.
Wang B, Zhang AQ, Wang HD, Klein JD, Tan L, Du J, et al. miR-26a limits muscle wasting and cardiac fibrosis through exosome-mediated microRNA transfer in chronic kidney disease. Theranostics. 2019;9:1864–77.
Dragomir MP, Knutsen E, Calin GA. Classical and noncanonical functions of miRNAs in cancers. Trends Genet. 2022;38:379–94.
Dragomir MP, Knutsen E, Calin GA. SnapShot: unconventional miRNA functions. Cell. 2018;174:1038.
Hooper CL, Dash PR, Boateng SY. Lipoma preferred partner is a mechanosensitive protein regulated by nitric oxide in the heart. FEBS Open Bio. 2012;2:135–44.
Li FF, Du MM, Yang YM, Wang Z, Zhang H, Wang XY, et al. Zinc finger and BTB domain-containing protein 20 aggravates angiotensin II-induced cardiac remodeling via the EGFR-AKT pathway. J Mol Med (Berl). 2022;100:427–38.
Lear TB, Lockwood KC, Larsen M, Tuncer F, Kennerdell JR, Morse C, et al. Kelch-like protein 42 is a profibrotic ubiquitin E3 ligase involved in systemic sclerosis. J Biol Chem. 2020;295:4171–80.
He L, Thomson JM, Hemann MT, Mu D, Goodson S, Powers S, et al. A microRNA polycistron as a potential human oncogene. Nature. 2005;435:828–33.
Gao ZQ, Zhu XL, Dou YX. The miR-302/367 cluster: a comprehensive update on its evolution and functions. Open Biol. 2015;5:1501–38.
Yang JL, Zhang XF, Chen XJ, Wang L, Yang GD. Exosome mediated delivery of miR-124 promotes neurogenesis after ischemia. Mol Ther Nucleic Acids. 2017;7:278–87.
Cai GF, Cai GL, Zhou HC, Zhuang Z, Liu K, Pei SY, et al. Mesenchymal stem cell-derived exosome miR-542-3p suppresses inflammation and prevents cerebral infarction. Stem Cell Res Ther. 2021;12:2.
Seow YQ, Wood MJ. Biological gene delivery vehicles: beyond viral vectors. Mol Ther. 2009;17:767–77.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81970664, 82000648, 82070735); the Natural Science Foundation of Jiangsu Province (No. BK20211385, BK20200363); the 789 Outstanding Talent Program of the Second Affiliated Hospital of Nanjing Medical University (No. 789ZYRC202080119, 789ZYRC202090251); the Science and Technology Development Foundation of Nanjing Medical University (No. NMUB2020049) and the Fundamental Research Funds for the Central Universities (No. 2242023K40046). We are grateful for the help provided by Lab Center, the Second Affiliated Hospital of Nanjing Medical University and Institute of Nephrology, Southeast University.
Author information
Authors and Affiliations
Contributions
AQZ and BW designed the experiments. JLJ analyzed and interpreted the data and wrote the paper. JLJ, HMS, and ZLL conducted the experiments and analyzed the data. RJ drew the graphical abstract. GTQ, HZ, EW, YYQ, XYL, LD, LCD, and WHG contributed to the data analysis. DFD provided serum samples of patients.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ji, Jl., Shi, Hm., Li, Zl. et al. Satellite cell-derived exosome-mediated delivery of microRNA-23a/27a/26a cluster ameliorates the renal tubulointerstitial fibrosis in mouse diabetic nephropathy. Acta Pharmacol Sin 44, 2455–2468 (2023). https://doi.org/10.1038/s41401-023-01140-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41401-023-01140-4
Keywords
This article is cited by
-
The translational potential of miR-26 in atherosclerosis and development of agents for its target genes ACC1/2, COL1A1, CPT1A, FBP1, DGAT2, and SMAD7
Cardiovascular Diabetology (2024)
-
Nanocarrier-Mediated Delivery of MicroRNAs for Fibrotic Diseases
Molecular Diagnosis & Therapy (2024)