Abstract
Background:
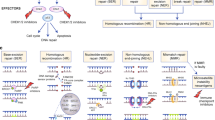
Recent advances in genetic sequencing and recent studies focusing on the broader use of Poly ADP Ribose polymerase (PARP) inhibitors have led to an upsurge of interest in DNA repair mutations as they relate to prostate cancer.
Methods:
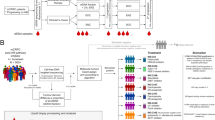
This review outlines ongoing studies and recent publications as they relate to the prevalence and detection of DNA repair mutations in metastatic prostate cancer. The detection of these mutations through multiple diagnostic approaches is discussed, including germline sequencing, somatic sequencing, cell-free DNA assays, and circulating tumor cell assays. The clinical course of patients with prostate cancer and DNA repair gene mutations is explored in addition to therapeutic strategies for this population.
Conclusions:
In men with metastatic prostate cancer, it is reasonable to obtain germline genetic sequencing as well as somatic tumor genomic sequencing to help guide further treatment decisions that may include PARP inhibitors or carboplatin in the future. In the future, in men with metastatic castrate resistant prostate cancer with progression on PARP inhibitors, cell-free DNA sequencing may help elucidate mechanisms of resistance, which include reversion mutations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kohler BA, Sherman RL, Howlader N, Jemal A, Ryerson AB, Henry KA, et al. Annual report to the nation on the status of cancer, 1975-2011, featuring incidence of breast cancer subtypes by race/ethnicity, poverty, and state. J Natl Cancer Inst. 2015;107:djv048.
Geethakumari PR, Cookson MS, Kelly WK, Prostate cancer clinical trials working group. The evolving biology of castration-resistant prostate cancer: review of recommendations from the prostate cancer clinical trials working group 3. Oncol (Williston Park). 2016;30:187–95.
Robinson D, Van Allen EM, Wu YM, Schultz N, Lonigro RJ, Mosquera JM, et al. Integrative clinical genomics of advanced prostate cancer. Cell 2015;161:1215–28.
Dantzer F, de La Rubia G, Menissier-De Murcia J, Hostomsky Z, de Murcia G, Schreiber V. Base excision repair is impaired in mammalian cells lacking Poly(ADP-ribose) polymerase-1. Biochemistry. 2000;39:7559–69.
Ame JC, Spenlehauer C, de Murcia G. The PARP superfamily. Bioessays. 2004;26:882–93.
Dhawan M, Ryan CJ, Ashwiorth A. DNA repair deficiency is common in advanced prostate cancer: new therapeutic opportunities. Oncologist. 2016;21:940–5.
Bryant HE, Schultz N, Thomas HD, et al. Specific killing of BRCA2-deficient tumours with inhibitor of poly(ADP-ribose) polymerase. Nature. 2005;434:913–7.
Farmer H, McCabe N, Lord CJ, et al. Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature. 2005;434:917–21.
Mateo J, Carreira S, Sandhu S, Miranda S, Mossop H, Perez-Lopez R, et al. DNA-repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697–708.
Barbieri CE, Baca SC, Lawrence MS, et al. Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nat Genet. 2012;44:685–689.
Cancer Genome Atlas Research Network. The molecular taxonomy of primary prostate cancer. Cell. 2015;163:1011–25.
Tannock IF, Hickman JA. Limits to personalized cancer medicine. N Engl J Med. 2016;375:1289–94.
Le Tourneau C, Delord JP, Goncalves A, Gavoille C, Dubot C, Isambert N, et al. Molecularly targeted therapy based on tumour molecular profiling versus conventional therapy for advanced cancer (SHIVA): a multicentre, open-label, proof-of-concept, randomised, controlled phase 2 trial. Lancet Oncol. 2015;16:1324–34.
Campone M, Arnedos M, Dieras V, et al. Comparative genomic hybridisation array and DNA sequencing to direct treatment of metastatic breast cancer: a multicentre, prospective trial (SAFIR01/UNICANCER). Lancet Oncol. 2014;15:267–74.
Iqbal J, Ragone A, Lubinski J, et al. The incidence of pancreatic cancer in BRCA1 and BRCA2 mutation carriers. Br J Cancer. 2012;107:2005.
Phelan CM, Iqbal J, Lynch HT, et al. Incidence of colorectal cancer in BRCA1 and BRCA2 mutation carriers: results from a follow-up study. Br J Cancer. 2014;110:530.
Breast Cancer Linkage C. Cancer risks in BRCA2 mutation carriers. J Natl Cancer Inst. 1999;91:1310–6.
Lord CJ, Ashworth A. BRCAness revisited. Nat Rev Cancer. 2016;16:110–20.
Lord CJ, Ashworth A. PARP inhibitors: synthetic lethality in the clinic. Science. 2017;355:1152–8.
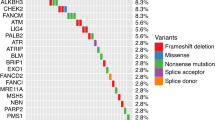
Pritchard CC, Mateo J, Walsh MF, De Sarkar N, Abida W, Beltran H, et al. Inherited DNA- repair gene mutations in men with metastatic prostate cancer. N Engl J Med. 2016;375:443–53.
The Cancer Genome Atlas (TCGA) Research Network. Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609–15.
Pennington KP, Walsh T, Harrell MI, et al. Germline and somatic mutations in homologous recombination genes predict platinum response and survival in ovarian, fallopian tube, and peritoneal carcinomas. Clin Cancer Res. 2014;20:764–75.
Mohler J, et al: NCCN Guidelines: Prostate cancer (Version 1.2018). J Natl Compr Cancer Network Feb. 2018;11.
Offit K, Bradbury A, Storm C, Merz JF, Noonan KE, Spence R. Gene patents and personalized cancer care: impact of the Myriad case on clinical oncology. J Clin Oncol. 2013;31:2743–8.
Jahr S, Hentze H, Englisch S, Hardt D, Fackelmayer FO, Hesch RD, et al. DNA fragments in the blood plasma of cancer patients: quantitations and evidence for their origin from apoptotic and necrotic cells. Cancer Res. 2001;61:1659–65.
Villaflor V, Won B, Nagy R, Banks K, Lanman RB, Talasaz A, et al. Biopsy-free circulating tumor DNA assay identifies actionable mutations in lung cancer. Oncotarget. 2016;7:66880–91.
Newman AM, Bratman SV, To J, Wynne JF, Eclov NC, Modlin LA, et al. An ultrasensitive method for quantitating circulating tumor DNA with broad patient coverage. Nat Med. 2014;20:548–54.
Stroun M, Lyautey J, Lederrey C, Olson-Sand A, Anker P. About the possible origin and mechanism of circulating DNA apoptosis and active DNA release. Clin Chim Acta. 2001;313:139–42.
Lanman RB, Mortimer SA, Zill OA, Sebisanovic D, Lopez R, Blau S, et al. Analytical and clinical validation of a digital sequencing panel for quantitative, highly accurate evaluation of cell-free circulating tumor DNA. PLoS ONE. 2015;10:e0140712.
Zhang T, Armstrong A. Clinical utility of circulating tumor cells in advanced prostate cancer. Curr Onco Rep. 2016;18:3
de Bono JS, Scher HI, Montgomery RB, Parker C, Miller MC, Tissing H, et al. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2008;14:6302–9.
Goldkorn A, Ely B, Quinn DI, Tangen CM, Fink LM, Xu T, et al. Circulating tumor cell counts are prognostic of overall survival in SWOG S0421: a phase III trial of docetaxel with or without atrasentan for metastatic castration-resistant prostate cancer. J Clin Oncol. 2014;32:1136–42.
Vansant G, A single cell genomic signature to detect homologous recombination deficiency (HRD) and PARP inhibitors sensitivity using patient’s circulating tumor cells (CTCs). J Clin Oncol. 2016;34 (suppl; abstre23015).
Castro E, Goh C, Olmos D, Saunders E, Leongamornlert D, Tymrakiewicz M, et al. Germline BRCA mutations are associated with higher risk of nodal involvement, distant metastasis, and poor survival outcomes in prostate cancer. J Clin Oncol. 2013;31:1748–57.
Castro E, Goh C, Leongamornlert D, Saunders E, Tymrakiewicz M, Dadaev T, et al. Effect of BRCA mutations on metastatic relapse and cause-specific survival after radical treatment for localised prostate cancer. Eur Urol. 2015;68:186–93.
Risbridger GP, Taylor RA, Clouston D, Sliwinski A, Thorne H, Hunter S, et al. Patient-derived xenografts reveal that intraductal carcinoma of the prostate is a prominent pathology in BRCA2 mutation carriers with prostate cancer and correlates with poor prognosis. Eur Urol. 2015;67:496–503.
McNeal JE, Yemoto CE. Spread of adenocarcinoma within prostatic ducts and acini. Morphol Clin Correl Am J Surg Pathol. 1996;20:802–14.
Zhao S, Chang S, Erho N, Yu M, Lehrer J, et al. Associations of luminal and basal subtyping of prostate cancer with prognosis and response to androgen deprivation therapy. JAMA Oncol. 2017;3:p.1663.
US National Library of Medicine. Clinicatrials.gov available at http://www.clinicaltrials.gov. Accessed Feb 2018.
Mirza MR, Monk BJ, Herrstedt J, Oza AM, Mahner S, Redondo A, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375:2154–64.
Swisher EM, Lin KK, Oza AM, Scott CL, Giordano H, Sun J, et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 Part 1): an international, multicentre, open-label, phase 2 trial. Lancet Oncol. 2017;18:75–87.
Kaufman B, Shapira-Frommer R, Schmutzler RK, Audeh MW, Friedlander M, Balmana J, et al. Olaparib monotherapy in patients with advanced cancer and a germline BRCA1/2 mutation. J Clin Oncol. 2015;33:244–50.
Robson M, Im SA, Senkus E, Xu B, Domchek SM, Masuda N, et al. Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. N Engl J Med. 2017;377:523–33.
Fong PC, Boss DS, Yap TA, Tutt A, Wu P, Mergui-Roelvink M, et al. Inhibition of poly(ADP- ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med. 2009;361:123–34.
Rajan A, Carter CA, Kelly RJ, Gutierrez M, Kummar S, Szabo E, et al. A phase I combination study of olaparib with cisplatin and gemcitabine in adults with solid tumors. Clin Cancer Res. 2012;18:2344–51.
Dhawan M, Bartelink I. et al. Differential toxicity in patients with and without DNA repair mutations: Phase I Study of Carboplatin and Talazoparib in advanced solid tumors. Clin Res Res. 2017;23:6400–10.
Corn PGTS, Zurita AJ, et al. A multi-institutional randomized phase II study (NCT01505868) of cabazitaxel (CAB) plus or minus carboplatin (CARB) in men with metastatic castration- resistant prostate cancer (mCRPC). J Clin Oncol 33, 2015 (suppl; abstr 5010).
Sternberg CN, Petrylak DP, Sartor O, et al. Multinational, doubleblind, phase III study of prednisone and either satraplatin or placebo in patients with castrate-refractory prostate cancer progressing after prior chemotherapy: the SPARC trial. J Clin Oncol. 2009;27:5431–5438.
Pomerantz M, Spisák S, Jia L, Cronin A, Csabai I, Ledet E. et al. The association between germline BRCA2 variants and sensitivity to platinum-based chemotherapy among men with metastatic prostate cancer. Cancer . 2017;123:3532–3539.
Hussain M, Daignault-Newton S, Twardowski PW, Albany C, Stein MN, Kunju LP. et al.Targeting androgen receptor and DNA repair in metastatic castration-resistant prostate cancer: results from NCI 9012.J Clin Oncol. 2018;36:991–9.
Chi K, Annala M, et al. A randomized phase II cross-over study of abiraterone+prednisone (ABI) vs enzalutamide (ENZ) for patients (pts) with metastatic, castration-resistant prostate cancer (mCRPC). J Clin Oncol 35, 2017 (suppl; abstr 5002).
Yip S, Chi K. et al. Outcomes in patients with advanced prostate cancer and inactivating germline mutations in BRCA2 or ATM. J Clin Oncol. 2018;36:242
James ND, de Bono JS, Spears MR, Clarke NW, Mason MD, Dearnaley DP, et al. Abiraterone for Prostate Cancer Not Previously Treated with Hormone Therapy. N Engl J Med. 2017;377:338–51.
Hussain M, Fizazi K, Saad F. et al.PROSPER: A phase 3, randomized, double-blind, placebo (PBO)-controlled study of enzalutamide (ENZA) in men with nonmetastatic castration-resistant prostate cancer (M0 CRPC). J Clin Oncol.2018;36:3
Fizazi K, Tran N, Fein L, Matsubara N, Rodriguez-Antolin A, Alekseev BY, et al. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N Engl J Med. 2017;377:352–60.
Ashworth A. Drug resistance caused by reversion mutation. Cancer Res. 2008;68:10021–3.
Norquist B, Wurz KA, Pennil CC, Garcia R, Gross J, Sakai W, et al. Secondary somatic mutations restoring BRCA1/2 predict chemotherapy resistance in hereditary ovarian carcinomas. J Clin Oncol. 2011;29:3008–15.
Quigley D, Alumkal JJ, Wyatt AW, Kothari V, Foye A, Lloyd P. et al. Analysis of circulating cell-free DNA identifies multi-clonal heterogeneity of BRCA2 reversion mutations associated with resistance to PARP inhibitors. Cancer Discov. 2017;7:999–1005.
Karzai F, et al. A phase 2 study of olaparib and durvalumab in metastatic castrate-resistant prostate cancer (mCRPC) in an unselected population, J Clin Oncol 36, 2018 (suppl 6S; abstr 163)
Acknowledgements
Dr. Dhawan has received funding from 2016–2017 from the Prescient grant NIH TL1, Grant Number UL1 TR001872.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Ryan has received research support from Clovis Oncology and has a consultant/advisory role with Janssen. Dr. Dhawan has served as a consultant for Driver.
Rights and permissions
About this article
Cite this article
Dhawan, M., Ryan, C.J. BRCAness and prostate cancer: diagnostic and therapeutic considerations. Prostate Cancer Prostatic Dis 21, 488–498 (2018). https://doi.org/10.1038/s41391-018-0069-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-018-0069-2
This article is cited by
-
Unveiling the vulnerabilities of synthetic lethality in triple-negative breast cancer
Clinical and Translational Oncology (2023)
-
Role of the DNA damage response in prostate cancer formation, progression and treatment
Prostate Cancer and Prostatic Diseases (2020)