Abstract
Background
Colder temperature exposure is a known trigger for pediatric asthma exacerbation. The induction of oxidative stress is a known pathophysiologic pathway for asthma exacerbation. However, the role of oxidative stress in linking colder temperature exposure and worsened pediatric asthma symptoms is poorly understood.
Methods
In a panel study involving 43 children with asthma, aged 5–13 years old, each child was visited 4 times with a 2-week interval. At each visit, nasal fluid, urine, and saliva samples were obtained and measured for biomarkers of oxidative stress in the nasal cavity (nasal malondialdehyde [MDA]), the circulatory system (urinary MDA), and the oral cavity (salivary MDA). Childhood Asthma-Control Test (CACT) was used to assess asthma symptoms.
Results
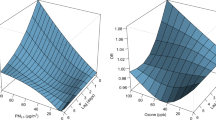
When ambient daily-average temperature ranged from 7 to 18 °C, a 2 °C decrement in personal temperature exposures were significantly associated with higher nasal MDA and urinary MDA concentrations by 47–77% and 6–14%, respectively. We estimated that, of the decrease in child-reported CACT scores (indicating worsened asthma symptoms and asthma control) associated with colder temperature exposure, 14–57% were mediated by nasal MDA.
Conclusion
These results suggest a plausible pathway that colder temperature exposure worsens pediatric asthma symptoms partly via inducing nasal oxidative stress.
Impact
-
The role of oxidative stress in linking colder temperature exposure and worsened asthma symptoms is still poorly understood.
-
Lower temperature exposure in a colder season was associated with higher nasal and systemic oxidative stress in children with asthma.
-
Nasal MDA, a biomarker of nasal oxidative stress, mediated the associations between colder temperature exposures and pediatric asthma symptoms.
-
The results firstly suggest a plausible pathway that colder temperature exposure worsens pediatric asthma symptoms partly via inducing oxidative stress in the nasal cavity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available to preserve individuals’ privacy but are available from the corresponding author on reasonable request.
References
Barrett, C. et al. Eat or Heat: Fuel Poverty and Childhood Respiratory Health. Lancet Respir. Med. 10, 229 (2022).
Hyrkäs-Palmu, H. et al. Cold Weather Increases Respiratory Symptoms and Functional Disability Especially among Patients with Asthma and Allergic Rhinitis. Sci. Rep. 8, 1–8 (2018).
He, L. et al. Associations between Personal Apparent Temperature Exposures and Asthma Symptoms in Children with Asthma. PLoS One 18, e0293603 (2023).
Xu, Z. et al. Extreme Temperatures and Emergency Department Admissions for Childhood Asthma in Brisbane, Australia. Occup. Environ. Med. 70, 730–735 (2013).
Chen, Y. et al. Associations between Ambient Temperature and Adult Asthma Hospitalizations in Beijing, China: A Time-Stratified Case-Crossover Study. Respir. Res. 23, 1–12 (2022).
Cohen, J., Agel, L., Barlow, M., Garfinkel, C. I. & White, I. Linking Arctic Variability and Change with Extreme Winter Weather in the United States. Science 373, 1116–1121 (2021).
Nadeem, A., Raj, H. G. & Chhabra, S. K. Increased Oxidative Stress in Acute Exacerbations of Asthma. J. Asthma 42, 45–50 (2005).
Strauss, R. H., McFadden, E. Jr, Ingram, R. Jr, Jaeger, J. J. & Stearns, D. R. Enhancement of Exercise-Induced Asthma by Cold Air. N. Engl. J. Med. 297, 743–747 (1977).
Kaminsky, D. A., Bates, J. H. & Irvin, C. G. Effects of Cool, Dry Air Stimulation on Peripheral Lung Mechanics in Asthma. Am. J. Respir. Crit. Care Med. 162, 179–186 (2000).
Deng, L. et al. High and Low Temperatures Aggravate Airway Inflammation of Asthma: Evidence in a Mouse Model. Environ. Pollut. 256, 113433 (2020).
Holguin, F. Oxidative Stress in Airway Diseases. Ann. Am. Thorac. Soc. 10, S150–S157 (2013).
D’Amato, M. et al. The Impact of Cold on the Respiratory Tract and Its Consequences to Respiratory Health. Clin. Transl. Allergy 8, 1–8 (2018).
Zeng, M. et al. Local and Systemic Oxidative Stress and Glucocorticoid Receptor Levels in Chronic Obstructive Pulmonary Disease Patients. Can. Respir. J. 20, 35–41 (2013).
He, L. et al. Malondialdehyde in Nasal Fluid: A Biomarker for Monitoring Asthma Control in Relation to Air Pollution Exposure. Environ. Sci. Technol. 54, 11405–11413 (2020).
Cui, X. et al. Association between Bedroom Particulate Matter Filtration and Changes in Airway Pathophysiology in Children with Asthma. JAMA Pediatrics 174, 533–542 (2020).
He, L. et al. Associations of Personal Exposure to Air Pollutants with Airway Mechanics in Children with Asthma. Environ. Int. 138, 105647 (2020).
Il’yasova, D., Scarbrough, P. & Spasojevic, I. Urinary Biomarkers of Oxidative Status. Clin. Chim. Acta 413, 1446–1453 (2012).
Khoubnasabjafari, M., Ansarin, K. & Jouyban, A. Salivary Malondialdehyde as an Oxidative Stress Biomarker in Oral and Systemic Diseases. J. Dent. Res. Dent. Clin. Dent. Prospects 10, 71 (2016).
Gong, J. et al. Malondialdehyde in Exhaled Breath Condensate and Urine as a Biomarker of Air Pollution Induced Oxidative Stress. J. Exposure Sci. Environ. Epidemiol. 23, 322 (2013).
Valavanidis, A., Vlachogianni, T. & Fiotakis, C. 8-Hydroxy-2′-Deoxyguanosine (8-Ohdg): A Critical Biomarker of Oxidative Stress and Carcinogenesis. J. Environ. Sci. Health Part C. 27, 120–139 (2009).
He, L. et al. Effects of Personal Air Pollutant Exposure on Oxidative Stress: Potential Confounding by Natural Variation in Melatonin Levels. Int. J. Hyg. Environ. Health 223, 116–123 (2020).
He, L., Liu, X. L. & Zhang, J. J. Simultaneous Quantification of Urinary 6‑Sulfatoxymelatonin and 8‑Hydroxy‑2′‑Deoxyguanosine Using Liquid Chromatography-Tandem Mass Spectrometry. J. Chromatogr. B 1095, 119–126 (2018).
Liu, A. H. et al. Development and Cross-Sectional Validation of the Childhood Asthma Control Test. J. Allergy Clin. Immunol. 119, 817–825 (2007).
Koolen, B. et al. Comparing Global Initiative for Asthma (Gina) Criteria with the Childhood Asthma Control Test (C-Act) and Asthma Control Test (Act). Eur. Respir. J. 38, 561–566 (2011).
Schoemann, A. M., Boulton, A. J. & Short, S. D. Determining Power and Sample Size for Simple and Complex Mediation Models. Soc. Psychological Personal. Sci. 8, 379–386 (2017).
Cruz, A. A., Naclerio, R. M., Proud, D. & Togias, A. Epithelial Shedding Is Associated with Nasal Reactions to Cold, Dry Air. J. Allergy Clin. Immunol. 117, 1351–1358 (2006).
Huang, D. et al. Cold Exposure Impairs Extracellular Vesicle Swarm–Mediated Nasal Antiviral Immunity. J. Allergy Clin. Immunol. 151, 509–525.e508 (2023).
Lester, S. N. & Li, K. Toll-Like Receptors in Antiviral Innate Immunity. J. Mol. Biol. 426, 1246–1264 (2014).
Novak, N., Haberstok, J., Bieber, T. & Allam, J.-P. The Immune Privilege of the Oral Mucosa. Trends Mol. Med. 14, 191–198 (2008).
Lin, Z., Sun, L., Li, L. & Jiang, S. Nasal Mucosa Is Much More Susceptible Than Oral Mucosa to Infection of Sars‐Cov‐2 Omicron Subvariants: Wearing Nasal Masks Where Facial Masks Cannot Be Used. J. Med. Virol. 95, e28577 (2023).
Blagojevic, D. P., Grubor-Lajsic, G. N. & Spasic, M. B. Cold Defence Responses: The Role of Oxidative Stress. Front. Biosci. Scholar 3, 416–427 (2011).
Saadeh, C., Saadeh, C., Cross, B., Gaylor, M. & Griffith, M. Advantage of Impulse Oscillometry over Spirometry to Diagnose Chronic Obstructive Pulmonary Disease and Monitor Pulmonary Responses to Bronchodilators: An Observational Study. SAGE Open. Medicine 3, 2050312115578957 (2015).
Guan, W.-j et al. Impulse Oscillometry in Adults with Bronchiectasis. Ann. Am. Thorac. Soc. 12, 657–665 (2015).
Nadeem, A. et al. Proteinase Activated Receptor‐2‐Mediated Dual Oxidase‐2 up‐Regulation Is Involved in Enhanced Airway Reactivity and Inflammation in a Mouse Model of Allergic Asthma. Immunology 145, 391–403 (2015).
Acknowledgements
We thank Xiaoxing Cui, Lin Fang, Yanbo Teng, Lili Lin, Xiaojian Zhou, Jianguo Hong, and Qian Wang for their valuable contributions to the collection of field data.
Funding
This study received funding from NSFC (No. 51521005 & 51420105010) and Underwriters Laboratories Inc.
Author information
Authors and Affiliations
Contributions
Drs. He and J. Zhang had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data: He, Norris, Barkjohn, Z. Li, F.Li, Y. Zhang, Black, Bergin, J. Zhang, Drafting the article or revising it critically for important intellectual content: He, Norris, Palaguachi-Lopez, Barkjohn, Bergin, and J. Zhang., Final approval of the version to be published: He, Norris, Palaguachi-Lopez, Barkjohn, Z. Li, F.Li, Y. Zhang, Black, Bergin, J. Zhang.
Corresponding authors
Ethics declarations
Competing interests
All the authors declare no competing interests.
Consent statement
Oral assent and written consent were obtained from the participants and their caregivers, respectively.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, L., Norris, C., Palaguachi-Lopez, K. et al. Nasal oxidative stress mediating the effects of colder temperature exposure on pediatric asthma symptoms. Pediatr Res (2024). https://doi.org/10.1038/s41390-024-03196-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41390-024-03196-2