Abstract
Background
Previous studies have shown that TREM2 plays a protective role in acute lung injury (ALI). This prospective study aimed to investigate the role of sTREM2 as a forecasting factor for ALI in infants after pediatric cardiac surgery undergoing cardiopulmonary bypass (CPB).
Methods
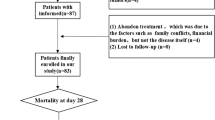
Seventy-five consecutive patients younger than 1 year who underwent cardiac surgery were enrolled in this study. Sixty-one fulfilled the inclusion criteria and had been divided into ALI and non-ALI groups. Children’s demographic characteristics and clinical data were collected. Perioperative sTREM2 levels were analyzed at five timepoints.
Results
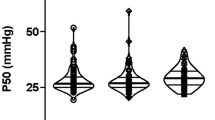
In this study, children in the ALI group were younger, lighter, with higher RACHS-1 scores and underwent significantly longer CPB time. Post-CPB ALI had an impact on clinical outcomes, which contributed to a longer duration of mechanical ventilation, ICU and hospital stay than non-ALI group. Significant differences were manifested off-CPB, 1 h/6 h after CPB, and day 1 after surgery between the two groups. Binary logistic models revealed that off-CPB sTREM2 was significantly associated with the incidence of post-CPB ALI after adjustment. ROC analysis showed that the AUC of off-CPB sTREM2 level was 0.791, and the optimal cutoff value was 788.6 pg/ml.
Conclusions
The off-CPB sTREM2 level was an independent prognostic factor for post-CPB ALI in infants.
Impact
-
Plasma sTREM2 works together with downstream TREM2 to regulate inflammation response by binding the receptor to other cells. Previous studies have shown that TREM2 plays a protective role in ischemia-reperfusion and has anti-inflammatory effects on acute lung injury (ALI).
-
This study analyzed the risk factors of post-cardiopulmonary bypass (CPB) ALI. We found that weight and off-CPB sTREM2 level were independent prognostic factors for post-CPB ALI.
-
Plasma sTREM2 may serve as an early biomarker in the prognostic evaluation of acute lung injury after cardiac surgery in infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Stephens, R. S., Shah, A. S. & Whitman, G. J. Lung injury and acute respiratory distress syndrome after cardiac surgery. Ann. Thorac. Surg. 95, 1122–1129 (2013).
Sanfilippo, F. et al. Acute respiratory distress syndrome in the perioperative period of cardiac surgery: predictors, diagnosis, prognosis, management options, and future directions. J. Cardiothorac. Vasc. Anesth. 36, 1169–1179 (2022).
Wang, D. et al. Incidence, risk factors, and outcomes of severe hypoxemia after cardiac surgery. Front. Cardiovasc. Med. 9, 934533 (2022).
Bellani, G. et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 315, 788–800 (2016).
Asimakopoulos, G., Smith, P. L., Ratnatunga, C. P. & Taylor, K. M. Lung injury and acute respiratory distress syndrome after cardiopulmonary bypass. Ann. Thorac. Surg. 68, 1107–1115 (1999).
Gaies, M. et al. Clinical epidemiology of extubation failure in the pediatric cardiac ICU: a report from the Pediatric Cardiac Critical Care Consortium. Pediatr. Crit. Care Med. 16, 837–845 (2015).
Rong, L. Q., Di Franco, A. & Gaudino, M. Acute respiratory distress syndrome after cardiac surgery. J. Thorac. Dis. 8, E1177–E1186 (2016).
Monteverde, E. et al. Characterization of pediatric patients receiving prolonged mechanical ventilation. Pediatr. Crit. Care Med. 12, e287–e291 (2011).
Asfari, A. et al. Biomarkers for adverse lung injury following pediatric cardiopulmonary bypass. Crit. Care Explor. 3, e0528 (2021).
Hou, J. et al. TREM2 sustains macrophage-hepatocyte metabolic coordination in nonalcoholic fatty liver disease and sepsis. J. Clin. Invest. 131, e135197 (2021).
Tanaka, M. et al. A potential novel pathological implication of serum soluble triggering receptor expressed on myeloid cell 2 in insulin resistance in a general Japanese population: The Hisayama study. Diabetes Res Clin. Pract. 146, 225–232 (2018).
Yang, J. et al. TREM2 ectodomain and its soluble form in Alzheimer’s disease. J. Neuroinflammation 17, 204 (2020).
Hendrikx, T. et al. Soluble TREM2 levels reflect the recruitment and expansion of TREM2(+) macrophages that localize to fibrotic areas and limit NASH. J. Hepatol. 77, 1373–1385 (2022).
Bernard, G. R. et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am. J. Respir. Crit. Care Med. 149, 818–824 (1994).
Klesney-Tait, J., Turnbull, I. R. & Colonna, M. The TREM receptor family and signal integration. Nat. Immunol. 7, 1266–1273 (2006).
Daws, M. R. et al. Pattern recognition by TREM-2: binding of anionic ligands. J. Immunol. 171, 594–599 (2003).
Liang, F. et al. Role and regulatory mechanism of triggering receptor expressed on myeloid cells 2 in mice lung ischemia/reperfusion injury. Zhonghua Wei Zhong Bing. Ji Jiu Yi Xue 33, 933–937 (2021).
Takahashi, K., Rochford, C. D. & Neumann, H. Clearance of apoptotic neurons without inflammation by microglial triggering receptor expressed on myeloid cells-2. J. Exp. Med. 201, 647–657 (2005).
Li, C., Zhao, B., Lin, C., Gong, Z. & An, X. TREM2 inhibits inflammatory responses in mouse microglia by suppressing the PI3K/NF-kappaB signaling. Cell Biol. Int. 43, 360–372 (2019).
Liu, D. et al. Impact of TREM-2 gene silencing on inflammatory response of endotoxin-induced acute lung injury in mice. Mol. Cell Biochem. 394, 155–161 (2014).
Nakao, T. et al. DNAX activating protein of 12 kDa/triggering receptor expressed on myeloid cells 2 expression by mouse and human liver dendritic cells: functional implications and regulation of liver ischemia-reperfusion injury. Hepatology 70, 696–710 (2019).
Wu, R. et al. TREM2 protects against cerebral ischemia/reperfusion injury. Mol. Brain 10, 20 (2017).
Zhong, L. et al. Soluble TREM2 induces inflammatory responses and enhances microglial survival. J. Exp. Med. 214, 597–607 (2017).
Rodriguez-Vieitez, E. & Ashton, N. J. Plasma sTREM2: a potential marker of cerebrovascular injury in neurodegenerative disorders. Brain 144, 3283–3285 (2021).
Satoh-Asahara, N. et al. Soluble TREM2 and Alzheimer-related biomarker trajectories in the blood of patients with diabetes based on their cognitive status. Diabetes Res Clin. Pract. 193, 110121 (2022).
Filipello, F. et al. Soluble TREM2: innocent bystander or active player in neurological diseases? Neurobiol. Dis. 165, 105630 (2022).
Szykowska, A. et al. Selection and structural characterization of anti-TREM2 scFvs that reduce levels of shed ectodomain. Structure 29, 1241–1252.e1245 (2021).
Acknowledgements
My deepest gratitude goes first and foremost to Professor Qiang Shu, for his constant encouragement and guidance. Second, I would like to express my thanks to my colleagues who have contributed to this article.
Funding
This research was supported by the Health Science and Technology Program of Zhejiang Province, China (2022KY865); the Fundamental Research Funds for the Central Universities, China (226-2022-00060).
Author information
Authors and Affiliations
Contributions
L.Y. and M.S. jointly supervised this work. All authors contributed to the study conception and design, material preparation, data collection and interpretation of data. L. Yang performed analysis and wrote the first draft of the manuscript. All the authors commented on previous versions of the manuscript and revised it critically for important intellectual content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was in line with the principles of the Declaration of Helsinki. The study was approved by Zhejiang University School of Medicine Children’s Hospital Ethic Committee on Clinical Investigation (2022-IRB-052). Informed consent was obtained from each participating individual’s guardian.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, L., Sun, M., Ying, L. et al. sTREM2 in the prognostic evaluation of acute lung injury after cardiac surgery in infants. Pediatr Res 95, 770–774 (2024). https://doi.org/10.1038/s41390-023-02915-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02915-5