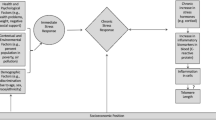
Abstract
Leukocyte telomere length, a marker of immune system function, is sensitive to exposures such as psychosocial stressors and health-maintaining behaviors. Past research has determined that stress experienced in adulthood is associated with shorter telomere length, but is limited to mostly cross-sectional reports. We test whether repeated reports of chronic psychosocial and financial burden is associated with telomere length change over a 5-year period (years 15 and 20) from 969 participants in the Coronary Artery Risk Development in Young Adults (CARDIA) Study, a longitudinal, population-based cohort, ages 18–30 at time of recruitment in 1985. We further examine whether multisystem resiliency, comprised of social connections, health-maintaining behaviors, and psychological resources, mitigates the effects of repeated burden on telomere attrition over 5 years. Our results indicate that adults with high chronic burden do not show decreased telomere length over the 5-year period. However, these effects do vary by level of resiliency, as regression results revealed a significant interaction between chronic burden and multisystem resiliency. For individuals with high repeated chronic burden and low multisystem resiliency (1 SD below the mean), there was a significant 5-year shortening in telomere length, whereas no significant relationships between chronic burden and attrition were evident for those at moderate and higher levels of resiliency. These effects apply similarly across the three components of resiliency. Results imply that interventions should focus on establishing strong social connections, psychological resources, and health-maintaining behaviors when attempting to ameliorate stress-related decline in telomere length among at-risk individuals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Richardson S, Shaffer JA, Falzon L, Krupka D, Davidson KW, Edmondson D. Meta-analysis of perceived stress and its association with incident coronary heart disease. Am J Cardiol. 2012;110:1711–6.
Cohen S, Janicki-Deverts D, Miller GE. Psychological stress and disease. J Am Med Assoc. 2007;298:1685–7.
Yusuf S, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case–control study. Lancet. 2004;364:937–52.
Aldwin CM, et al. Do stress trajectories predict mortality in older men? Longitudinal findings from the VA normative aging study. J Aging Res. 2011;2011:896109.
Miller GE, Chen E, Cole SW. Health psychology: developing biologically plausible models linking the social world and physical health. Annu Rev Psychol. 2009;60:501–24.
Gouin JP, Hantsoo L, Kiecolt-Glaser JK. Immune dysregulation and chronic stress among older adults: a review. Neuroimmunomodulation. 2008;15:251–9.
Blackburn EH. Telomere states and cell fates. Nature. 2000;408:53–56.
Campisi J, di Fagagna FD. Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol. 2007;8:729–40.
Sahin E, Depinho RA. Axis of ageing: telomeres, p53 and mitochondria. Nat Rev Mol Cell Biol. 2012;13:397–404.
Jaskelioff M, et al. Telomerase reactivation reverses tissue degeneration in aged telomerase-deficient mice. Nature. 2011;469:102–6.
Haycock PC, et al. Leucocyte telomere length and risk of cardiovascular disease: systematic review and meta-analysis. BMJ. 2014;349:g4227.
Codd V, et al. Identification of seven loci affecting mean telomere length and their association with disease. Nat Genet. 2013;45:422–7.
Wentzensen IM, Mirabello L, Pfeiffer RM, Savage SA. The association of telomere length and cancer: a meta-analysis. Cancer Epidemiol Biomark Prev. 2011;20:1238–50.
Ma H, et al. Shortened telomere length is associated with increased risk of cancer: a meta-analysis. PLoS ONE. 2011;6:e20466.
Zhao J, Miao K, Wang H, Ding H, Wang DW. Association between telomere length and type 2 diabetes mellitus: a meta-analysis. PLoS ONE. 2013;8:e79993.
Rode L, Bojesen SE, Weischer M, Vestbo J, Nordestgaard BG. Short telomere length, lung function and chronic obstructive pulmonary disease in 46,396 individuals. Thorax. 2013;68:429–35.
Zhan Y, et al. Telomere length shortening and Alzheimer disease—a Mendelian randomization study. JAMA Neurol. 2015;72:1202–3.
Cawthon RM, Smith KR, O’Brien E, Sivatchenko A, Kerber RA. Association between telomere length in blood and mortality in people aged 60 years or older. Lancet. 2003;361:393–5.
Epel ES, et al. The rate of leukocyte telomere shortening predicts mortality from cardiovascular disease in elderly men. Aging. 2009;1:81–88.
Glei DA, Goldman N, Weinstein M, Risques RA. Shorter ends, faster end? Leukocyte telomere length and mortality among older Taiwanese. J Gerontol A Biol Sci Med Sci. 2014; glu191. https://doi.org/10.1093/gerona/glu191.
Kimura M, et al. Telomere length and mortality: a study of leukocytes in elderly Danish twins. Am J Epidemiol. 2008;167:799–806.
Bakaysa SL, et al. Telomere length predicts survival independent of genetic influences. Aging Cell. 2007;6:769–74.
Deelen J, et al. Leukocyte telomere length associates with prospective mortality independent of immune-related parameters and known genetic markers. Int J Epidemiol. 2014;43:878–86.
Schaefer C, et al. Demographic and behavioral influences on telomere length and relationship with all-cause mortality: early results from the Kaiser Permanente Research Program on Genes, Environ, Health (RPGEH). Clin Med Res. 2013;11:146–146.
Bendix L, et al. Longitudinal changes in leukocyte telomere length and mortality in humans. J Gerontol A Biol Sci Med Sci. 2014;69:231–9.
Svensson J, et al. Leukocyte telomere length is not associated with mortality in older men. Exp Gerontol. 2014;57:6–12.
Duggan C, et al. Change in peripheral blood leukocyte telomere length and mortality in breast cancer survivors. J Natl Cancer Inst. 2014;106:dju035.
Njajou OT, et al. Association between telomere length, specific causes of death, and years of healthy life in health, aging, and body composition, a population-based cohort study. J Gerontol Ser A Biol Sci Med Sci. 2009;64:860–4.
Rode L, Nordestgaard BG, Bojesen SE. Peripheral blood leukocyte telomere length and mortality among 64,637 individuals from the general population. J Natl Cancer Inst. 2015;107:djv074.
Damjanovic AK, et al. Accelerated telomere erosion is associated with a declining immune function of caregivers of Alzheimer’s disease patients. J Immunol. 2007;179:4249–54.
Epel ES, et al. Accelerated telomere shortening in response to life stress. Proc Natl Acad Sci USA. 2004;101:17312–5.
Humphreys J, et al. Telomere shortening in formerly abused and never abused women. Biol Res Nurs. 2012;14:115–23.
Ala-Mursula L, et al. Long-term unemployment is associated with short telomeres in 31-year-old men: an observational study in the Northern Finland birth cohort 1966. PLoS ONE. 2013;8:e80094.
Litzelman K, et al. Association between informal caregiving and cellular aging in the survey of the health of Wisconsin: the role of caregiving characteristics, stress, and strain. Am J Epidemiol. 2014;179:1340–52.
Ahola K, et al. Work-related exhaustion and telomere length: a population-based study. PLoS ONE. 2012;7:e40186.
Pepper, GV, Bateson, M & Nettle, D. Telomeres as integrative markers of exposure to stress and adversity: a systematic review and meta-analysis. 2018 bioRxiv 320150. https://doi.org/10.1101/320150.
Puterman E, et al. Lifespan adversity and later adulthood telomere length in the nationally representative US Health and Retirement Study. Proc Natl Acad Sci USA. 2016;113:E6335–E6342.
Verhoeven JE, van Oppen P, Puterman E, Elzinga B, Penninx BWJH. The association of early and recent psychosocial life stress with leukocyte telomere length. Psychosom Med. 2015;77:882–91.
Puterman E, Lin J, Krauss J, Blackburn EH, Epel ES. Determinants of telomere attrition over 1 year in healthy older women: stress and health behaviors matter. Mol Psychiatry. 2015;20:529–35.
Taylor SE, Seeman TE. Psychosocial resources and the SES-health relationship. Ann N Y Acad Sci. 1999;896:210–25.
Taylor SE, Kemeny ME, Reed GM, Bower JE, Gruenewald TL. Psychological resources, positive illusions, and health. Am Psychol. 2000;55:99–109.
Cohen S, Wills TAS. Stress, social support, and the buffering hypothesis. Psychol Bull. 1985;98:310–57.
Hamer M. Psychosocial stress and cardiovascular disease risk: the role of physical activity. Psychosom Med. 2012;74:896–903.
Cohen S, Kamarck T, Mermelstein R. A global measure of perceived stress. J Health Soc Behav. 1983;24:385–96.
Puterman E, et al. The power of exercise: buffering the effect of chronic stress on telomere length. PLoS ONE. 2010;5:e10837.
Puterman E, et al. Aerobic exercise lengthens telomeres and reduces stress in family caregivers: a randomized controlled trial—Curt Richter Award Paper 2018. Psychoneuroendocrinology. 2018. https://doi.org/10.1016/J.PSYNEUEN.2018.08.002.
Liu JJW, Reed M, Girard TA. Advancing resilience: an integrative, multi-system model of resilience. Personal Individ Differ. 2017;111:111–8.
Puterman E, Epel ES. An intricate dance: life experience, multisystem resiliency, and rate of telomere decline throughout the lifespan. Soc Personal Psychol Compass. 2012;6:807–25.
Puterman E, et al. Multisystem resiliency moderates the major depression-telomere length association: findings from the heart and soul study. Brain Behav Immun. 2013;33:65–73.
Friedman GD, et al. CARDIA: study design, recruitment, and some characteristics of the examined subjects. J Clin Epidemiol. 1988;41:1105–16.
Cohen S, et al. Socioeconomic status, race, and diurnal cortisol decline in the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Psychosom Med. 2006;68:41–50.
Scheier MF, Carver CS, Bridges MW. Distinguishing optimism from neuroticism (and trait anxiety, self-mastery, and self-esteem): a reevaluation of the Life Orientation Test. J Personal Soc Psychol. 1994;67:1063.
Pearlin LI, Schooler C. The structure of coping. J Health Soc Behav. 1978;19:2–212.
Cawthon RM. Telomere measurement by quantitative PCR. Nucleic Acids Res. 2002;30:e47.
Lin J, et al. Analyses and comparisons of telomerase activity and telomere length in human T and B cells: insights for epidemiology of telomere maintenance. J Immunol Methods. 2010;352:71–80.
Enders CK. A primer on the use of modern missing-data methods in psychosomatic medicine research. Psychosom Med. 2006;68:427–36.
Schlomer GL, Bauman S, Card NA. Best practices for missing data management in counseling psychology. J Couns Psychol. 2010;57:1–10.
Glickman ME, Rao SR, Schultz MR. False discovery rate control is a recommended alternative to Bonferroni-type adjustments in health studies. J Clin Epidemiol. 2014;67:850–7.
Seeman TE, McEwen B, Rowe J, Singer B. Allostatic load as a marker of cumulative biological risk: MacArthur studies of successful aging. Proc Natl Acad Sci USA. 2001;98:4770–5.
Geronimus AT. The weathering hypothesis and the health of African-American women and infants: evidence and speculations. Ethn Dis. 1992;2:207–21.
Tomiyama AJ, et al. Does cellular aging relate to patterns of allostasis? An examination of basal and stress reactive HPA axis activity and telomere length. Physiol Behav. 2011. https://doi.org/10.1016/j.physbeh.2011.11.016.
Epel ES, et al. Cell aging in relation to stress arousal and cardiovascular disease risk factors. Psychoneuroendocrinology. 2006;31:277–87.
Parks CG, et al. Telomere length, current perceived stress, and urinary stress hormones in women. Cancer Epidemiol Biomark Prev. 2009;18:551–60.
Steptoe A, Hamer M, Lin J, Blackburn EH, Erusalimsky JD. The longitudinal relationship between cortisol responses to mental stress and leukocyte telomere attrition. J Clin Endocrinol Metab. 2016; 2016–3035. https://doi.org/10.1210/jc.2016-3035.
Charles ST, Piazza JR, Mogle J, Sliwinski MJ, Almeida DM. The wear and tear of daily stressors on mental health. Psychol Sci. 2013;24:733–41.
Piazza JR, Charles ST, Sliwinski MJ, Mogle J, Almeida DM. Affective reactivity to daily stressors and long-term risk of reporting a chronic physical health condition. Ann Behav Med. 2013;45:110–20.
Mroczek DK, et al. Emotional reactivity and mortality: longitudinal findings from the VA normative aging study. J Gerontol B Psychol Sci Soc Sci. 2013. https://doi.org/10.1093/geronb/gbt107.
Epel ES, et al. More than a feeling: a unified view of stress measurement for population science. Front Neuroendocrinol. 2018. https://doi.org/10.1016/j.yfrne.2018.03.001.
Puterman E, An EE. Intricate dance: life experience, multisystem resiliency, and rate of telomere decline throughout the lifespan. Soc Personal Psychol Compass. 2012;6:807–25.
Mezuk B, et al. “White Box” epidemiology and the social neuroscience of health behaviors. Soc Ment Health. 2013;3:79–95.
Schafer MH, Ferraro KF, Mustillo SA. Children of misfortune: early adversity and cumulative inequality in perceived life trajectories. AJS. 2011;116:1053.
Surtees PG, et al. Life stress, emotional health, and mean telomere length in the European prospective investigation into cancer (EPIC)—Norfolk population study. J Gerontol Ser a-Biol Sci Med Sci. 2011;66:1152–62.
Jodczyk S, Fergusson DM, Horwood LJ, Pearson JF, Kennedy MA. No association between mean telomere length and life stress observed in a 30 year birth cohort. PLoS ONE. 2014;9:e97102.
Steenstrup T, Hjelmborg JVB, Kark JD, Christensen K, Aviv A. The telomere lengthening conundrum-artifact or biology? Nucleic Acids Res. 2013;41:e131.
Verhoeven JE, Lin J, Révész D, Wolkowitz OM, Penninx BWJH, et al. Unresolved issues in longitudinal telomere length research: response to susser. Am J Psychiatry. 2016;173:1147–9.
Svenson U, et al. Blood cell telomere length is a dynamic feature. PLoS ONE. 2011;6:e21485.
Chen W, et al. Longitudinal versus cross-sectional evaluations of leukocyte telomere length dynamics: age-dependent telomere shortening is the rule. J Gerontol Ser A Biol Sci Med Sci. 2011;66:312–9.
Verhoeven JE, et al. Depression, telomeres and mitochondrial DNA: between- and within-person associations from a 10-year longitudinal study. Mol Psychiatry. 2017. https://doi.org/10.1038/mp.2017.48.
Révész D, et al. Associations between cellular aging markers and metabolic syndrome: findings from the cardia study. J Clin Endocrinol Metab. 2018;103:148–57.
Lauderdale DS, Knutson KL, Yan LL, Liu K, Rathouz PJ. Self-reported and measured sleep duration: how similar are they? Epidemiology. 2008;19:838–45.
Booth M. Assessment of physical activity: an international perspective. Res Q Exerc Sport. 2000;71:S114–20.
Adam TC, Epel ES. Stress, eating, and the reward system. Physiol Behav. 2007;91:449–58.
Schuit AJ, van Loon AJM, Tijhuis M, Ocké M. Clustering of lifestyle risk factors in a general adult population. Prev Med. 2002;35:219–24.
Poortinga W. The prevalence and clustering of four major lifestyle risk factors in an English adult population. Prev Med. 2007;44:124–8.
Sun Q, et al. Healthy lifestyle and leukocyte telomere length in U.S. women. PloS ONE. 2012;7:e38374.
Acknowledgements
The Coronary Artery Risk Development in Young Adults Study (CARDIA) is conducted and supported by the National Heart, Lung, and Blood Institute (NHLBI) in collaboration with the University of Alabama at Birmingham (HHSN268201300025C & HHSN268201300026C), Northwestern University (HHSN268201300027C), University of Minnesota (HHSN268201300028C), Kaiser Foundation Research Institute (HHSN268201300029C), and Johns Hopkins University School of Medicine (HHSN268200900041C). CARDIA is also partially supported by the Intramural Research Program of the National Institute on Aging (NIA) and an intra-agency agreement between NIA and NHLBI (AG0005). This paper has been reviewed by CARDIA for scientific content.
Funding
This research was undertaken, in part, thanks to funding to EP from the Canada Research Chairs program and, in part, from the National Heart, Lung and Blood Institute of the National Institutes of Health under award number K99/R00 HL 109247. Cell aging assays were supported by a grant to EP and EE by the John & Catherine MacArthur Foundation Research Network on Socioeconomic Status and Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies, including the National Institutes of Health. TCDB was supported by a National Institute of Mental Health grant T32MH019391.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cabeza de Baca, T., Prather, A.A., Lin, J. et al. Chronic psychosocial and financial burden accelerates 5-year telomere shortening: findings from the Coronary Artery Risk Development in Young Adults Study. Mol Psychiatry 25, 1141–1153 (2020). https://doi.org/10.1038/s41380-019-0482-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-019-0482-5
This article is cited by
-
Social support and telomere length: a meta-analysis
Journal of Behavioral Medicine (2023)
-
Molecular Psychiatry, August 2020: new impact factor, and highlights of recent advances in psychiatry, including an overview of the brain’s response to stress during infection with the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
Molecular Psychiatry (2020)
-
Advances in research on stress and behavior: special issue, 2020
Molecular Psychiatry (2020)
-
Advances in depression research: second special issue, 2020, with highlights on biological mechanisms, clinical features, co-morbidity, genetics, imaging, and treatment
Molecular Psychiatry (2020)