Abstract
Background
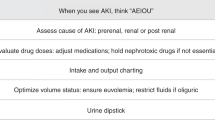
Daily serum creatinine monitoring protocols for acute kidney injury (AKI) are invasive and may lead to surveillance resistance. We aimed to understand if use of urine neutrophil gelatinase-associated lipocalin (uNGAL) could increase high-risk nephrotoxic medication (NTMx) associated AKI screening adherence in neonates.
Methods
Statistical process control methods prior to and post implementation were trended. The primary outcome, screening adherence, was defined as either daily serum creatinine or uNGAL assessment through 2 days post high-risk NTMx exposure.
Results
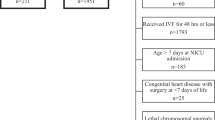
1291 monitoring days from the pre-implementation era (4/2020-6/2021) were compared to1377 monitoring days from the post-era (6/2021-10/2022). AKI screening adherence increased (81 to 92%) following implementation of optional uNGAL screening. Urine NGAL accounted for 35% of screening obtained. Use of uNGAL resulted in a 40% reduction in blood sampling for serum creatinine.
Conclusions
Incorporation of uNGAL as a complementary screening tool to serum creatinine demonstrated sustained increased AKI surveillance in our Baby NINJA monitoring program.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Rhone ET, Carmody JB, Swanson JR, Charlton JR. Nephrotoxic medication exposure in very low birth weight infants. J Matern Fetal Neonatal Med. 2014;27:1485–90.
Steflik HJ, Charlton JR, Briley M, Selewski DT, Gist KM, Hanna MH, et al. Neonatal nephrotoxic medication exposure and early acute kidney injury: results from the AWAKEN study. J Perinatol. 2023;43:1029–37.
Goldstein SL, Dahale D, Kirkendall ES, Mottes T, Kaplan H, Muething S, et al. A prospective multi-center quality improvement initiative (NINJA) indicates a reduction in nephrotoxic acute kidney injury in hospitalized children. Kidney Int. 2020;97:580–8.
Stoops C, Stone S, Evans E, Dill L, Henderson T, Griffin R, et al. Baby NINJA (nephrotoxic injury negated by just-in-time action): reduction of nephrotoxic medication-associated acute kidney injury in the neonatal intensive care unit. J Pediatr. 2019;215:223–8.e6.
Jakacka N, Snarski E, Mekuria S. Prevention of iatrogenic anemia in critical and neonatal care. Adv Clin Exp Med. 2016;25:191–7.
Askenazi D. Are we ready for the clinical use of novel acute kidney injury biomarkers? Pediatr Nephrol. 2012;27:1423–5.
Gavigan HW, Slagle CL, Krallman KA, Poindexter BB, Hooper DK, Goldstein SL. Blood transfusion rates in Baby NINJA (nephrotoxic injury negated by just-in-time action)-a single-center experience. Pediatr Nephrol. 2021;36:1901–5.
Askenazi DJ, Montesanti A, Hundley H, Koralkar R, Pawar P, Shuaib F, et al. Urine biomarkers predict acute kidney injury and mortality in very low birth weight infants. J Pediatr. 2011;159:907–12.e1.
Askenazi DJ, Koralkar R, Hundley HE, Montesanti A, Parwar P, Sonjara S, et al. Urine biomarkers predict acute kidney injury in newborns. J Pediatr. 2012;161:270–5.
Slagle CL, Goldstein SL, Gavigan HW, Rowe JA, Krallman KA, Kaplan HC, et al. Association between elevated urine neutrophil gelatinase-associated lipocalin and postoperative acute kidney injury in neonates. J Pediatr. 2021;238:193–201.e2.
Rumpel J, Spray BJ, Chock VY, Kirkley MJ, Slagle CL, Frymoyer A, et al. Urine biomarkers for the assessment of acute kidney injury in neonates with hypoxic ischemic encephalopathy receiving therapeutic hypothermia. J Pediatr. 2022;241:133–40.e3.
Bennett M, Dent CL, Ma Q, Dastrala S, Grenier F, Workman R, et al. Urine NGAL predicts severity of acute kidney injury after cardiac surgery: a prospective study. Clin J Am Soc Nephrol. 2008;3:665–73.
Lavery AP, Meinzen-Derr JK, Anderson E, Ma Q, Bennett MR, Devarajan P, et al. Urinary NGAL in premature infants. Pediatr Res. 2008;64:423–8.
Jansen D, Peters E, Heemskerk S, Koster-Kamphuis L, Bouw MP, Roelofs HM, et al. Tubular injury biomarkers to detect gentamicin-induced acute kidney injury in the neonatal intensive care unit. Am J Perinatol. 2016;33:180–7.
Goldstein SL, Krallman KA, Schmerge A, Dill L, Gerhardt B, Chodaparavu P, et al. Urinary neutrophil gelatinase-associated lipocalin rules out nephrotoxic acute kidney injury in children. Pediatr Nephrol. 2021;36:1915–21.
Askenazi DJ, Koralkar R, Patil N, Halloran B, Ambalavanan N, Griffin R. Acute kidney injury urine biomarkers in very low-birth-weight infants. Clin J Am Soc Nephrol. 2016;11:1527–35.
Tabel Y, Elmas A, Ipek S, Karadag A, Elmas O, Ozyalin F. Urinary neutrophil gelatinase-associated lipocalin as an early biomarker for prediction of acute kidney injury in preterm infants. Am J Perinatol. 2014;31:167–74.
Goldstein SL, Kirkendall E, Nguyen H, Schaffzin JK, Bucuvalas J, Bracke T, et al. Electronic health record identification of nephrotoxin exposure and associated acute kidney injury. Pediatrics. 2013;132:e756–67.
Jetton JG, Askenazi DJ. Acute kidney injury in the neonate. Clin Perinatol. 2014;41:487–502.
Selewski DT, Cornell TT, Heung M, Troost JP, Ehrmann BJ, Lombel RM, et al. Validation of the KDIGO acute kidney injury criteria in a pediatric critical care population. Intensive Care Med. 2014;40:1481–8.
Benneyan JC, Lloyd RC, Plsek PE. Statistical process control as a tool for research and healthcare improvement. Qual Saf Health Care. 2003;12:458–64.
R Developement Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2020.
Ree IMC, Lopriore E. Updates in neonatal hematology: causes, risk factors, and management of anemia and thrombocytopenia. Hematol Oncol Clin North Am 2019;33:521–32.
Stoops C, Gavigan H, Krallman K, Anderson N, Griffin R, Slagle C, et al. The Utility of Urinary NGAL as an alternative for serum creatinine to detect acute kidney injury in infants exposed to nephrotoxic medications in the neonatal intensive care unit. Neonatology. 2023:1–10. https://doi.org/10.1159/000535322. Epub ahead of print.
Askenazi DJ, Koralkar R, Levitan EB, Goldstein SL, Devarajan P, Khandrika S, et al. Baseline values of candidate urine acute kidney injury biomarkers vary by gestational age in premature infants. Pediatr Res. 2011;70:302–6.
Haase M, Devarajan P, Haase-Fielitz A, Bellomo R, Cruz DN, Wagener G, et al. The outcome of neutrophil gelatinase-associated lipocalin-positive subclinical acute kidney injury: a multicenter pooled analysis of prospective studies. J Am Coll Cardiol. 2011;57:1752–61.
Haase M, Kellum JA, Ronco C. Subclinical AKI–an emerging syndrome with important consequences. Nat Rev Nephrol. 2012;8:735–9.
Marcello M, Virzi GM, Mucino-Bermejo MJ, Milan Manani S, Giavarina D, Salvador L, et al. Subclinical AKI and clinical outcomes in elderly patients undergoing cardiac surgery: diagnostic utility of NGAL versus standard creatinine increase criteria. Cardiorenal Med. 2022;12:94–105.
Koyner JL. Subclinical acute kidney injury is acute kidney injury and should not be ignored. Am J Respir Crit Care Med. 2020;202:786–7.
Sellmer A, Bech BH, Bjerre JV, Schmidt MR, Hjortdal VE, Esberg G, et al. Urinary neutrophil gelatinase-associated lipocalin in the evaluation of patent ductus arteriosus and AKI in very preterm neonates: a cohort study. BMC Pediatr. 2017;17:7.
Acknowledgements
Previous presentations from various information included in this manuscript include an oral abstract presented at the American Society of Nephrology Kidney Week, on November 3rd, 2022, in Orlando, FL as well as a poster presentation at the AKI & CRRT conference, in San Diego, CA, and Pediatric Academic Societies Conference, Washington D.C, 2023.
Author information
Authors and Affiliations
Contributions
CL Slagle was involved in conceptualization, methodology, data curation and analysis, and the writing of the original draft. TH was involved in conceptualization, methodology, data curation and analysis, and review of the original draft. HWG was involved in the conceptualization, methodology, data curation, and review of the original draft. KAK was involved in the conceptualization, methodology, data curation, and review of the original draft. SLG was involved in the conceptualization, methodology, and review of the original draft.
Corresponding author
Ethics declarations
Competing interests
Cara Slagle, MD is a consultant for AM Pharma and Medtronic and has received honorariums from Medtronic, the University of Alabama, and the AKI Critical Care Foundation, all unrelated to the work above. Stuart Goldstein, MD and Cincinnati Children’s Hospital receive royalties from Vigilanz Corporation (Minneapolis, MN) for the NINJA application licensed to Vigilanz from Cincinnati Children’s Hospital Medical Center. Dr. Goldstein consults and receives research funding from BioPorto Diagnostics, Inc, which markets The ProNephroAKI NGAL Test™.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author has moved institutions since time of initial submission: Cara Slagle.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Slagle, C.L., Hemmelgarn, T., Gavigan, H.W. et al. Use of urine neutrophil gelatinase-associated lipocalin for nephrotoxic medication acute kidney injury screening in neonates. J Perinatol (2024). https://doi.org/10.1038/s41372-024-01922-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41372-024-01922-6