Abstract
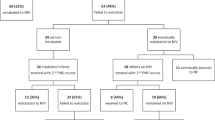
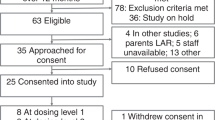
ABSTRACT: Bronchopulmonary dysplasia is an important complication of ventilation in babies for which treatment with steroids has been advocated. We report the results of a phase I study of early i.v. dexamethasone to prevent the development of bronchopulmonary dysplasia in a high-risk population of ventilated premature babies, < 30 wk gestation, with surfactant-treated respiratory distress syndrome. This study used a limited dexamethasone dosing regimen to minimize toxicity but used administration early in the course of acute lung disease to interrupt the injury cycle. Forty babies were enrolled; 19 were randomized to receive dexamethasone (0.5 mg/kg birth weight at 12–18 h of age and a second dose 12 h later) and 21 were randomized to receive placebo (i.v. saline). The dexamethasone group required less ventilatory support (mean airway, peak in-spiratory and end expiratory pressures, and intermittent mandatory ventilation) and supplemental oxygen after study d 4 (all p < 0.05, repeated measures analysis of variance). Improved tidal volume in the dexamethasone group, as measured by pulmonary function testing of infants who remained intubated, was seen on study d 7 (p = 0.02, t test). The dexamethasone group required shorter hospitalizations (median of 95 d versus 106 d, p = 0.01) (proportional hazards regression). Survival in the dexa methasone group was 89% versus 67% in the placebo group (p = 0.08, x2 analysis). Survival without bronchopulmonary dysplasia, diagnosed at 36 wk corrected gestational age, was 68% in the dexamethasone group versus 43% in the placebo group (p = 0.14). Mean blood pressure was elevated on study d 4 through 7 in the dexamethasone group, but this difference resolved by study d 10 without pharmacologic intervention. No differences in hyperglycemia, incidence of intraventricular hemorrhage (or its severity), or days to regain birth weight were seen. Early administration of dexamethasone resulted in short-term and suggested long-term benefits without significant complications. The results of this trial justify a large scale, broader-based (phase II) trial in premature babies with respiratory distress syndrome to determine the limits of effectiveness and the incidence of less-frequent potential side effects.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sanders, R., Cox, C., Phelps, D. et al. Two Doses of Early Intravenous Dexamethasone for the Prevention of Bronchopulmonary Dysplasia in Babies with Respiratory Distress Syndrome. Pediatr Res 36 (Suppl 1), 122–128 (1994). https://doi.org/10.1203/00006450-199407001-00022
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199407001-00022
This article is cited by
-
A systematic review of randomized controlled trials for the prevention of bronchopulmonary dysplasia in infants
Journal of Perinatology (2014)
-
Postnatale dexamethason en neurologische uitkomst: een meta-analyse
Tijdschrift voor kindergeneeskunde (2005)
-
Early neonatal dexamethasone treatment for prevention of bronchopulmonary dysplasia. Randomised trial and meta-analysis evaluating the duration of dexamethasone therapy
European Journal of Pediatrics (2005)
-
Indications for high-dose dexamethasone therapy in respiratory distress syndrome
European Journal of Pediatrics (1996)
-
Dexamethasone therapy in chronic lung disease
The Indian Journal of Pediatrics (1996)