Abstract
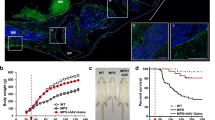
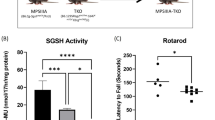
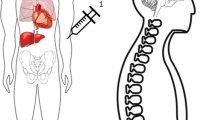
ABSTRACT: β-Glucuronidase injected i.v. into newborn mucopolysaccharidosis VII mice was cleared from the circulation in less than 1 h and taken up by tissues in a distribution corresponding to the location of the mannose 6-phosphate receptor. One h after a 3.5-mg/kg β-glucuronidase injection, β-glucuronidase levels were equal to or greater than normal in every organ examined with the exception of the brain, where 31% normal activity was present. Enzyme was detectable histochemically in the major sites of pathology for mucopolysaccharidosis VII including bone, brain, heart, and fixed tissue macrophages. The half-life of recombinant β-glucuronidase activity in various organs of injected mucopolysaccharidosis VII mice was 1.5 to 4.5 d. These studies show that recombinant β-glucuronidase administered to newborn mice reaches the sites of clinically important storage in murine mucopolysaccharidosis VII.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vogler, C., Sands, M., Higgins, A. et al. Enzyme Replacement with Recombinant β-Glucuronidase in the Newborn Mucopolysaccharidosis Type VII Mouse. Pediatr Res 34, 837–840 (1993). https://doi.org/10.1203/00006450-199312000-00028
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199312000-00028
This article is cited by
-
Association between postnatal second-hand smoke exposure and ADHD in children: a systematic review and meta-analysis
Environmental Science and Pollution Research (2021)
-
Brain transplantation of genetically modified bone marrow stromal cells corrects CNS pathology and cognitive function in MPS VII mice
Gene Therapy (2004)
-
Prevention of systemic clinical disease in MPS VII mice following AAV-mediated neonatal gene transfer
Gene Therapy (2001)
-
Elimination of lysosomal storage in brains of MPS VII mice treated by intrathecal administration of an adeno-associated virus vector
Gene Therapy (1999)
-
Long-term delivery of a lysosomal enzyme by genetically modified fibroblasts in dogs
Nature Medicine (1995)