Abstract
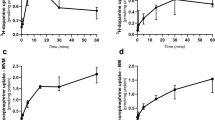
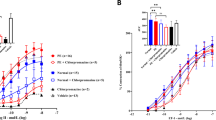
Summary: Somatostatin is a known inhibitor of hormone secretion and of nutrient transport. Because somatostatin-like immunoreactivity has been detected in amniotic fluid and the placenta has both hormone secretory and nutrient transport functions, we investigated the possible existence of somatostatin receptors on placenta cell membranes. Binding of 125I-Tyr1-and125I-Tyr11-somatostatin (5-21%) to solubilized placenta cell membranes was observed. Binding was time-, temperature-, and pH-dependent and occurred maximally with incubation at concentrations of 25 μg of membrane protein. Displacement of binding of 125I-Tyr1 and Tyr11 somatostatin by cold cyclic and linear somatostatin and somatostatin analogs Ala-5, Ala-8, and Ala-11 was observed. Scatchard analysis of data revealed high capacity of (Ro 0.44 mol/μg × 10−12) but low affinity (Kd 1.8 M × 10−7) binding sites similar to that reported in other tissues. Binding was not reversible under our experimental conditions. The significance of this low affinity binding of somatostatin to placenta cell membranes remains to be determined.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsalikian, E., Foley, T. & Becker, D. Characterization of Somatostatin Specific Binding in Plasma Cell Membranes of Human Placenta. Pediatr Res 18, 953–957 (1984). https://doi.org/10.1203/00006450-198410000-00009
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-198410000-00009
This article is cited by
-
Management of pituitary tumors in pregnancy
Nature Reviews Endocrinology (2011)
-
Effects of alloxan-induced diabetes on somatostatin binding to cytosolic components of rabbit gastric fundic mucosa
Bioscience Reports (1988)