Abstract
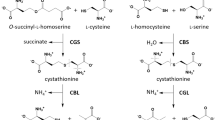
Extract: Cystathionine synthase activities are reported for extracts of fibroblasts grown from 39 control subjects, 47 homocystinuric individuals, and 10 parents of cystathionine synthase-deficient patients. Among the group with homocystinuria, fibroblast extracts from 38 had specific activities of cystathionine synthase below the control range. A number of considerations indicate that these 38 patients excrete homocystine because of cystathionine synthase deficiency. Fibroblasts from nine patients with homocystinuria had specific activities of cystathionine synthase within the control range. This group of nine was shown to be comprised of two individuals with cystathionine synthase deficiency, three with deficient activity of methylenetetrahydrofolate reductase, three with deficient activity of N5-methyltetrahydrofolate-homocysteine methyl-transferase, and one in whom homocystine excretion found by others in earlier studies could not be confirmed during the present investigation. The specific activities in fibroblasts of parents of cystathionine synthase deficient patients in most cases fall near the low end of the control range.
An assay of increased sensitivity was used to measure the cystathionine synthase activities in extracts of fibroblasts from cystathionine synthase-deficient patients at several concentrations of added pyridoxal phosphate. Of 25 cystathionine synthase-deficient patients judged to be clinically responsive to pyridoxine treatment, 24 had detectable cystathionine synthase activities in fibroblast extracts when the assays were performed without added pyridoxal phosphate. These activities ranged from approximately 0.1 % to 10% of the mean control value, and generally were stimulated no more by the addition of pyridoxal phosphate than were extracts from normal cells. Of 10 cystathionine synthase-deficient patients judged not to be responsive to pyridoxine, 9 had no cystathionine synthase activity in fibroblast extracts detected by the method employed. The cystathionine synthase in the cell extracts of the single nonresponsive patient with significant activity was stimulated much more by in vitro addition of pyridoxal phosphate than was control cystathionine synthase.
Genetic heterogeneity in cystathionine synthase-deficient patients and some of the genetic implications of the demonstrated α2β2 subunit structure of mammalian cystathionine synthase are discussed.
Speculation: Knowledge of enzyme activities, which includes cystathionine synthase, N5-methyl-tetrahydrofolate-homocysteine methyltransferase and methylenetetrahydrofolate reductase, in cultured fibroblasts is of diagnostic value in distinguishing between the several genetic abnormalities which may lead to excessive excretion of homocystine in the urine.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Uhlendorf, B., Conerly, E. & Mudd, S. Homocystinuria: Studies in Tissue Culture. Pediatr Res 7, 645–658 (1973). https://doi.org/10.1203/00006450-197307000-00008
Issue Date:
DOI: https://doi.org/10.1203/00006450-197307000-00008
Keywords
This article is cited by
-
Protein‐bound plasma homocyst(e)ine and identification of heterozygotes for cystathionine‐synthase deficiency
Journal of Inherited Metabolic Disease (1986)
-
Improved identification of heterozygotes for homocystinuria due to cystathionine synthase deficiency by the combination of methionine loading and enzyme determination in cultured fibroblasts
Human Genetics (1985)
-
Recent advances in the mechanism of pyridoxine‐responsive disorders
Journal of Inherited Metabolic Disease (1985)