Abstract
Extract: Hyperglycinemia is a disorder of amino acid metabolism characterized by the presence of increased concentrations of glycine in the blood, urine, and cerebrospinal fluid. It is now recognized that there are two forms of hyperglycinemia each representing distinct diseases. These studies were designed to assess the metabolism of glycine in the nonketotic form of hyperglycinemia. Isotope content was assessed in respiratory CO2 and in glycine, serine and the β carbon of serine of plasma after the separate intravenous injections of glycine-I-14C and glycine-2-14C.
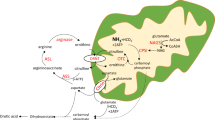
The specific activities of 14CO2 isolated from expired air after the injection of glycine-1-14C (fig. 2) declined in control subjects from peak values at 10 to 15 minutes in nearly linear fashion over a 2-hour period. In contrast, curves obtained in the patients were rather flat, rising slowly after injection to highest values at about 60 minutes. At 15 minutes, values for the control individuals exceeded those of the patients by a factor of 5− to 10-fold. These data indicate a defect in the formation of 14CO2 from the first carbon of glycine. When the control subjects were infused with nonisotopic glycine to produce pools comparable to those found in the patient, the specific activities of the serine isolated from plasma after the injection of glycine-1-14C (table II) were virtually the same in both groups. The rate of conversion of glycine-2-14C to serine (fig. 3) in the patients was, however, considerably slower than it was in the control subjects for at least the first 30 minutes, and the curves were flat throughout. Degradation of the serine isolated from plasma and precipitation of the β carbon as formaldemethone indicated that the incorporation of the α carbon of glycine into the β carbon of serine was much higher in the controls than in the patients (fig. 4). The curves for the patients approximated the abscissa indicating virtually no conversion.
These data indicate that in nonketotic hyperglycinemia there is a defect in the oxidation of carbon 1 of glycine to CO2 and in the conversion of carbon 2 of glycine to carbon 3 of serine. This is consistent with a defect in an enzyme catalyzing the transformation of glycine to CO2, NH3 and hydroxymethyltetrahydrofolate.
Speculation: The data obtained indicate that patients with nonketotic hyperglycinemia are unable in vivo to convert the first carbon of glycine directly to CO2 and the second carbon of glycine to the third carbon of serine. This is consistent with a genetic defect in an enzyme which catalyzes decarboxylation and formation of hydroxymethyltetrahydrofolate from glycine. It should be possible to document such a defect at a cellular and subcellular level.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ando, T., Nyhan, W., Gerritsen, T. et al. Metabolism of Glycine in the Nonketotic Form of Hyperglycinemia. Pediatr Res 2, 254–263 (1968). https://doi.org/10.1203/00006450-196807000-00004
Issue Date:
DOI: https://doi.org/10.1203/00006450-196807000-00004
Keywords
This article is cited by
-
Treatment of non‐ketotic hyperglycinaemia with diazepam, choline and folic acid
Journal of Inherited Metabolic Disease (1982)
-
Metabolism of glycine in the normal individual and in patients with non‐ketotic hyperglycinaemia
Journal of Inherited Metabolic Disease (1982)
-
Modification of fibroblast propionyl CoA carboxylase activity by valine as a possible diagnostic technique in non‐ketotic hyperglycinaemia
Journal of Inherited Metabolic Disease (1982)
-
Non‐ketotic hyperglycinaemia in a family with an unusual phenotype
Journal of Inherited Metabolic Disease (1978)
-
Propionyl‐CoA‐carboxylase determination: Study of enzyme parameters in cultured skin fibroblasts from enzyme‐deficient and normal subjects
Journal of Inherited Metabolic Disease (1978)