Abstract
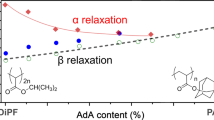
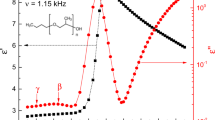
In the enthalpy relaxation of poly(cyanobiphenyl hexylacrylate) (PCBA6), the decrease in enthalpy was measured as a function of ageing time and ageing temperature in order to analyse it using activation energy spectrum (AES) model. In AES model the decrease in enthalpy is considered to be controlled by molecular processes whose energies are distributed over a continuous spectrum. The relaxation functions were derived from the enthalpy data for four PCBA6 samples of different molecular weights(Mw), then they were verified to obey stretched exponential forms. The relaxation times showed Arrhenius type temperature dependences to give Eapp, apparent activation energies, which became plateau around 200 kJ/mol as the rise in Mw. A plot of reduced ageing times was constructed after the manner of time–temperature reducibility. The fraction of free volume and the thermal expansion coefficient of glass transition were calculated by the method of WLF plot. Then, it was found that these values were relatively large, which is attributable to a property of a side-chain type polymer. AES showed bell-shaped curves; the beginning and the end of the peak were considered to coincide with the lower and the upper limits of activation energies of the relaxation processes, respectively. AES results, such as the area, the height of the peak, the upper and the lower limits, were discussed in terms of the molecular weight and the ageing temperature.
Similar content being viewed by others
Article PDF
References
Y. Tanaka, S. Takahashi, R. Kishi, and H. Ichijo, Mol. Cryst. Liq. Cryst., 339, 45 (2000).
Y. Tanaka, R. Kishi, and H. Ichijo, Mol. Cryst. Liq. Cryst., 348, 179 (2000).
P. Frubing, A. Kremmer, R. Gerhard-Multhaupt, A. Spanoudaki, and P. Pissis, J. Chem. Phys., 125, 214701 (2006).
W. C. Tsen and F. S. Chuang, J. Appl. Polym. Sci., 101, 4242 (2006).
D. Banu, A. El-Aghoury, and D. Feldman, J. Appl. Polym. Sci., 101, 2732 (2006).
M. R. J. Gibbs, J. E. Evetts, and J. A. Leake, J. Mater. Sci., 18, 278 (1983).
O. S. Narayanaswamy, J. Am. Ceram. Soc., 54, 491 (1971).
C. T. Moynihan, A. J. Easteal, M. A. DeBolt, and J. Tucker, J. Am. Ceram. Soc., 59, 12 (1976).
G. W. Scherer, J. Am. Ceram. Soc., 67, 504 (1984).
I. M. Hodge, Macromolecules, 20, 2897 (1987).
I. M. Hodge, J. Res. Natl. Inst. Stand. Technol., 102, 195 (1997).
J. M. G. Cowie and R. Ferguson, Macromolecules, 22, 2307 (1989).
J. L. Gomez Ribelles and M. Monleon Pradas, Macromolecules, 28, 5867 (1995).
J. M. Meseguer Duenas, A. V. Garayo, F. Romero Colomer, J. Mas Estelles, J. L. Gomez Ribelles, and M. Monleon Pradas, J. Polym. Sci., Polym. Phys. Ed., 35, 2201 (1997).
L. Andreozzi, M. Faetti, M. Giordano, and D. Palazzuoli, Macromolecules, 35, 9049 (2002).
V. Lorenzo, J. M. Perena, J. M. Perez, R. Benavente, and A. Bello, J. Mater. Sci., 32, 3601 (1997).
C. Alvarez, N. T. Correia, J. J. Moura Ramos, and A. C. Fernandes, Polymer, 41, 2907 (2000).
V. P. Shivaev, S. G. Kostromin, and N. A. Plate, Eur. Polym. J., 18, 651 (1982).
T. I. Gubina, S. Kise, S. G. Kostromin, R. V. Talroze, V. P. Shivaev, and N. A. Plate, Liq. Cryst., 4, 197 (1989).
A. S. Marshall and S. E. B. Petrie, J. Appl. Phys., 46, 4223 (1975).
F. L. Cumbrera, F. Sanchez-bajo, F. Guiberteau, J. D. Solier, and A. Munoz, J. Mater. Sci., 28, 5387 (1993).
G. Williams and D. C. Watts, Trans. Faraday Soc., 66, 80 (1970).
G. Williams, D. C. Watts, S. B. Dev, and A. M. North, Trans. Faraday Soc., 67, 1323 (1971).
J. M. G. Cowie and R. Ferguson, Polym. Commun., 27, 258 (1986).
I. M. Hodge, J. Non-Cryst. Solids, 169, 211 (1994).
S. E. B. Petrie, J. Polym. Sci., A2, 10, 1255 (1972).
S. Montserrat, P. Colomer, and J. Belana, J. Mater. Chem., 2, 217 (1992).
M. J. Richardson and N. G. Savill, Polymer, 16, 753 (1975).
M. J. Richardson, J. Polym. Sci., Part C, 38, 251 (1972).
D. Lacey, G. Nestor, and M. J. Richardson, Thermochim. Acta, 238, 99 (1994).
E. A. Collins and A. P. Metzger, Polym. Eng. and Sci., 10, 57 (1970).
G. V. Vinogradov, A. Ya. Malkin, and V. G. Kulichikhin, J. Polym. Sci., Polym. Phys. Ed., 8, 333 (1970).
V. A. Harmandaris, M. Doxastakis, V. G. Mavrantzas, and D. N. Theodorou, J. Chem. Phys., 116, 436 (2002).
E. von Meerwall, J. Grisby, D. Tomich, and R. van Antwerp, J. Polym. Sci., Polym. Phys. Ed., 20, 1037 (1982).
C. P. Lindsey and G. D. Patterson, J. Chem. Phys., 73, 3348 (1980).
M. L. Williams, R. F. Landel, and J. D. Ferry, J. Am. Chem. Soc., 77, 3701 (1955).
J. D. Ferry, in “Viscoelastic Properties of Polymers,” John Wiley & Sons, Inc., 1960.
S. Montserrat and P. Cortes, Makromole. Chem. Macromol. Symp., 20/21, 389 (1988).
A. Munoz, F. L. Cumbrera, and R. Marquez, Mater. Chem. Phys., 21, 279 (1989).
W. Rupp, H. P. Grossmann, and B. Stoll, Liq. Cryst., 3, 583 (1988).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tanaka, Y. Enthalpy Relaxation of Liquid Crystalline Polymer with Cyanobiphenyl Group in the Side Chain: Activation Energy Spectrum Analysis. Polym J 39, 1030–1039 (2007). https://doi.org/10.1295/polymj.PJ2007054
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2007054