Abstract
Introduction:
Allergic diseases are common, increasing, and associated with significant morbidity and occasional, but potentially avoidable, mortality. There is widespread public, professional and government agreement about the poor quality of care received by many patients with allergic diseases in the UK. Many remain undiagnosed, few have access to specialist services, and the majority of primary care health professionals have no allergy training. We report an evaluation of a novel, UK NHS-funded primary care-based allergy service in North West London.
Methods:
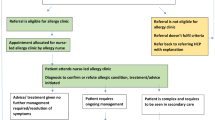
The clinic was run by a specialist allergy nurse and a general practitioner (GP) with a special interest in respiratory disease and allergy. Referrals from local GPs were made on dedicated proformas and handled according to developed and agreed community care pathways.
Results:
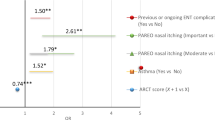
141/151 (93%) of those referred attended the clinic. On average, patients waited 15.7 (SD 15.67) days from referral to an offer of an appointment and were seen 18.11 (SD18.07) daysrfbom the date of referral. The majority of patients were referred for hives or wheals (61;40%), suspected food allergy (28;40%), multiple reasons, (19;12%) or rhinitis (19;12%). Skin prick and specific-IgE tests were performed on 69 (49%) and 19 (14%) patients respectively, with 4 (0.3%) patients having both. We estimated an overall saving of at least £13,580 to the local health economy — through reduced secondary care referrals — during the nine months' duration of the clinic. Assessed using a validated patient satisfaction questionnaire, most patients rated their satisfaction with the service with an overall median score of 75% (IQR 65.64-82.32).
Conclusions:
A primary care allergy service provided by appropriately trained personnel can cater adequately for the majority of primary care referrals. This model provides a pathway of care that supports self-directed care, reduces inappropriate referrals, and encourages care in the local community. The model is one that could easily be developed further into a clinical assessment service for allergy including respiratory based (asthma) problems. This would expand the scope and ensure value for money as well as responding to a clinical need with significant incidence locally. The UK Department of Health should consider rolling out this primary care-based model nationally.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
MLL has accepted sponsorship from GlaxoSmithKline, AstraZeneca, Trinity-Cheisi, Merck Sharpe and Dohme, Merck, Altana Pharma, Novartis, Meda Pharmaceuticals, 3M Pharmaceuticals, Schering Plough for attending conferences. He has accepted lecture fees from Boehringer Ingelheim, GlaxoSmithKline, AstraZeneca and Alk-Abello. He has been on advisory boards or provided consultancy for Schering Plough, Merck Sharp and Dohme, Trinity- Cheisi, Altana Pharma, Ranbaxy, AstraZeneca, 3M Pharmaceuticals and Novartis. In addition, he has received research grants from GSK, Schering Plough, Boehringer Ingelheim, Pfizer, and Astra Zeneca. He has also received an educational grant from Lincoln Medical Ltd for development of an online audit for health professionals on injectable adrenaline. He is a member of ADMIT (The Aerosol Drug Management Improvement Team) which is funded by an educational grant from MEDA Pharmaceuticals.
Rights and permissions
About this article
Cite this article
Levy, M., Walker, S., Woods, A. et al. Service evaluation of a UK primary care-based allergy clinic: quality improvement report. Prim Care Respir J 18, 313–319 (2009). https://doi.org/10.4104/pcrj.2009.00042
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.4104/pcrj.2009.00042
This article is cited by
-
Development and implementation of a nurse-led allergy clinic model in primary care: feasibility trial protocol
npj Primary Care Respiratory Medicine (2019)
-
Complexity of case mix in a regional allergy service
BMC Research Notes (2012)