Abstract
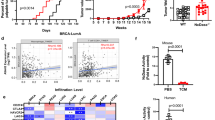
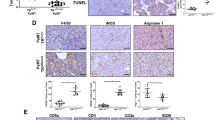
Although cysteine cathepsins have been identified as key regulators of cancer growth, their specific role in tumor development remains unclear. Recent studies have shown that high activity levels of tumor cathepsins are primarily a result of increased cathepsin activity in cancer-promoting tumor-associated macrophages (TAMs). To further investigate the role of cysteine cathepsin activity in normal and polarized macrophages, we established in vitro and in vivo models of macrophage differentiation and polarization and used a novel cysteine cathepsin inhibitor, GB111-NH2, to block the activity of cathepsins B, L and S. Here we show that in vitro, cysteine cathepsin inhibition yields both apoptosis and proliferation of macrophages, owing to increased oxidative stress. Proteomic analysis of cathepsin- inhibited macrophages demonstrates inhibition of autophagy, suggesting a likely cause of elevated reactive oxygen species (ROS) levels. In vivo models of mammary cancer further show that cathepsin inhibition yields TAM death owing to increased ROS levels. Strikingly, apoptosis in TAMs yields a seemingly cell non-autonomous death of neighboring cancer cells, and regression of the primary growth. These results show that cysteine cathepsin inhibitors can specifically trigger macrophage cell death and may function as an effective anticancer therapy in tumors with high levels of TAMs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Condeelis J, Pollard JW . Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 2006; 124: 263–266.
Qian BZ, Pollard JW . Macrophage diversity enhances tumor progression and metastasis. Cell 2010; 141: 39–51.
Galdiero MR, Garlanda C, Jaillon S, Marone G, Mantovani A . Tumor associated macrophages and neutrophils in tumor progression. J Cell Physiol 2013; 228: 1404–1412.
Mantovani A, Biswas SK, Galdiero MR, Sica A, Locati M . Macrophage plasticity and polarization in tissue repair and remodelling. J Pathol 2013; 229: 176–185.
Pollard JW . Trophic macrophages in development and disease. Nat Rev Immunol 2009; 9: 259–270.
White ES, Mantovani AR . Inflammation, wound repair, and fibrosis: reassessing the spectrum of tissue injury and resolution. J Pathol 2013; 229: 141–144.
Sica A, Mantovani A . Macrophage plasticity and polarization: in vivo veritas. J Clin Invest 2012; 122: 787–795.
Vasiljeva O, Papazoglou A, Krüger A, Brodoefel H, Korovin M, Deussing J et al. Tumor cell-derived and macrophage-derived cathepsin B promotes progression and lung metastasis of mammary cancer. Cancer Res 2006; 66: 5242–5250.
Vasiljeva O, Korovin M, Gajda M, Brodoefel H, Bojic L, Krüger A et al. Reduced tumour cell proliferation and delayed development of high-grade mammary carcinomas in cathepsin B-deficient mice. Oncogene 2008; 27: 4191–4199.
Vasiljeva O, Reinheckel T, Peters C, Turk D, Turk V, Turk B et al. Emerging roles of cysteine cathepsins in disease and their potential as drug targets. Curr Pharm Des 2007; 13: 387–403.
Jedeszko C, Sloane BF . Cysteine cathepsins in human cancer. Biol Chem 2004; 385: 1017–1027.
Mikkelsen T, Yan PS, Ho KL, Sameni M, Sloane BF, Rosenblum ML et al. Immunolocalization of cathepsin B in human glioma: implications for tumor invasion and angiogenesis. J Neurosurg 1995; 83: 285–290.
Palermo C, Joyce JA . Cysteine cathepsin proteases as pharmacological targets in cancer. Trends Pharmacol Sci 2008; 29: 22–28.
Gocheva V, Wang HW, Gadea BB, Shree T, Hunter KE, Garfall AL et al. IL-4 induces cathepsin protease activity in tumor-associated macrophages to promote cancer growth and invasion. Genes Dev 2010; 24: 241–255.
Goulet B, Sansregret L, Leduy L, Bogyo M, Weber E, Chauhan SS et al. Increased expression and activity of nuclear cathepsin L in cancer cells suggests a novel mechanism of cell transformation. Mol Cancer Res 2007; 5: 899–907.
Goulet B, Baruch A, Moon NS, Poirier M, Sansregret LL, Erickson A et al. A cathepsin L isoform that is devoid of a signal peptide localizes to the nucleus in S phase and processes the CDP/Cux transcription factor. Mol Cell 2004; 14: 207–219.
Concha C, Monardes A, Even Y, Morin V, Puchi M, Imschenetzky M et al. Inhibition of cysteine protease activity disturbs DNA replication and prevents mitosis in the early mitotic cell cycles of sea urchin embryos. J Cell Physiol 2005; 204: 693–703.
Zhang Y, Morgan MJ, Chen K, Choksi S, Liu ZG . Induction of autophagy is essential for monocyte–macrophage differentiation. Blood 2012; 119: 2895–2905.
Zavasnik-Bergant T, Turk B . Cysteine proteases: destruction ability versus immunomodulation capacity in immune cells. Biol Chem 2007; 388: 1141–1149.
Larghi P, Porta C, Riboldi E, Totaro MG, Carraro L, Orabona C et al. The p50 subunit of NF-κB orchestrates dendritic cell lifespan and activation of adaptive immunity. PLoS One 2012; 7: e45279.
Pello OM, Andrés V . Role of c-MYC in tumor-associated macrophages and cancer progression. Oncoimmunology 2013; 2: e22984.
Sevenich L, Schurigt U, Sachse K, Gajda M, Werner F, Müller S et al. Synergistic antitumor effects of combined cathepsin B and cathepsin Z deficiencies on breast cancer progression and metastasis in mice. Proc Natl Acad Sci USA 2010; 107: 2497–2502.
Bell-McGuinn KM, Garfall AL, Bogyo M, Hanahan D, Joyce JA . Inhibition of cysteine cathepsin protease activity enhances chemotherapy regimens by decreasing tumor growth and invasiveness in a mouse model of multistage cancer. Cancer Res 2007; 67: 7378–7385.
Schurigt U, Sevenich L, Vannier C, Gajda M, Schwinde A, Werner F et al. Trial of the cysteine cathepsin inhibitor JPM-OEt on early and advanced mammary cancer stages in the MMTV-PyMT-transgenic mouse model. Biol Chem 2008; 389: 1067–1074.
Blum G, Mullins SR, Keren K, Fonovic M, Jedeszko C, Rice MJ et al. Dynamic imaging of protease activity with fluorescently quenched activity-based probes. Nat Chem Biol 2005; 1: 203–209.
Meara JP, Rich DH . Mechanistic studies on the inactivation of papain by epoxysuccinyl inhibitors. J Med Chem 1996; 39: 3357–3366.
Porta C, Rimoldi M, Raes G, Brys L, Ghezzi P, Di Liberto D et al. Tolerance and M2 (alternative) macrophage polarization are related processes orchestrated by p50 nuclear factor kappaB. Proc Natl Acad Sci USA 2009; 106: 14978–14983.
Hawker KM, Johnson PR, Hughes JM, Black JL . Interleukin-4 inhibits mitogen-induced proliferation of human airway smooth muscle cells in culture. Am J Physiol 1998; 275: L469–L477.
Hsu KF, Wu CL, Huang SC, Wu CM, Hsiao JR, Yo YT et al. Cathepsin L mediates resveratrol-induced autophagy and apoptotic cell death in cervical cancer cells. Autophagy 2009; 5: 451–460.
Dennemärker J, Lohmüller T, Müller S, Aguilar SV, Tobin DJ, Peters C et al. Impaired turnover of autophagolysosomes in cathepsin L deficiency. Biol Chem 2010; 391: 913–922.
Goulet B, Truscott M, Nepveu A . A novel proteolytically processed CDP/Cux isoform of 90 kDa is generated by cathepsin L. Biol Chem 2006; 387: 1285–1293.
Katunuma N . Posttranslational processing and modification of cathepsins and cystatins. J Signal Transduct 2010; 2010: 375345.
Pan L, Li Y, Jia L, Qin Y, Qi G, Cheng J et al. Cathepsin S deficiency results in abnormal accumulation of autophagosomes in macrophages and enhances Ang II-induced cardiac inflammation. PLoS One 2012; 7: e35315.
Liao X, Sluimer JC, Wang Y, Subramanian M, Brown K, Pattison JS et al. Macrophage autophagy plays a protective role in advanced atherosclerosis. Cell Metab 2012; 15: 545–553.
Dragin N, Smani M, Arnaud-Dabernat S, Dubost C, Moranvillier I, Costet P et al. Acute oxidative stress is associated with cell proliferation in the mouse liver. FEBS Lett 2006; 580: 3845–3852.
Jacquel A, Obba S, Boyer L, Dufies M, Robert G, Gounon P et al. Autophagy is required for CSF-1-induced macrophagic differentiation and acquisition of phagocytic functions. Blood 2012; 119: 4527–4531.
Turk B . Targeting proteases: successes, failures and future prospects. Nat Rev Drug Discov 2006; 5: 785–799.
Withana NP, Blum G, Sameni M, Slaney C, Anbalagan A, Olive MB et al. Cathepsin B inhibition limits bone metastasis in breast cancer. Cancer Res 2012; 72: 1199–1209.
Elie BT, Gocheva V, Shree T, Dalrymple SA, Holsinger LJ, Joyce JA et al. Identification and pre-clinical testing of a reversible cathepsin protease inhibitor reveals anti-tumor efficacy in a pancreatic cancer model. Biochimie 2010; 92: 1618–1624.
Shree T, Olson OC, Elie BT, Kester JC, Garfall AL, Simpson K et al. Macrophages and cathepsin proteases blunt chemotherapeutic response in breast cancer. Genes Dev 2011; 25: 2465–2479.
Lin Y, Wei C, Liu Y, Qiu Y, Liu C, Guo F et al. Selective ablation of tumor-associated macrophages suppresses metastasis and angiogenesis. Cancer Sci 2013; 104: 1217–1225.
Wisniewski JR, Zougman A, Nagaraj N, Mann M . Universal sample preparation method for proteome analysis. Nat Methods 2009; 6: 359–362.
Wiśniewski JR, Zougman A, Mann M . Combination of FASP and StageTip-based fractionation allows in-depth analysis of the hippocampal membrane proteome. J Proteome Res 2009; 8: 5674–5678.
Rappsilber J, Mann M, Ishihama Y . Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat Protocols 2007; 2: 1896–1906.
Cox J, Mann M . MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat Biotechnol 2008; 26: 1367–1372.
Blum G, von Degenfeld G, Merchant MJ, Blau HM, Bogyo M . Noninvasive optical imaging of cysteine protease activity using fluorescently quenched activity-based probes. Nat Chem Biol 2007; 3: 668–677.
Acknowledgements
We thank Dr Michael Berger (The Hebrew University) for providing reagents and advice regarding the project. We would like to acknowledge Boris Turk (Lubljana, Slovenia) for supplying the recombinant cathepsins. This work was supported by the Israel Cancer Research Foundation (ICRF) (to GB, SJS and TG) and the European Research Council (Marie Curie International Reintegration Grant) (to GB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Salpeter, S., Pozniak, Y., Merquiol, E. et al. A novel cysteine cathepsin inhibitor yields macrophage cell death and mammary tumor regression. Oncogene 34, 6066–6078 (2015). https://doi.org/10.1038/onc.2015.51
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.51
This article is cited by
-
Helicobacter pylori and gastric cancer: a lysosomal protease perspective
Gastric Cancer (2022)
-
Cysteine cathepsins L and X differentially modulate interactions between myeloid-derived suppressor cells and tumor cells
Cancer Immunology, Immunotherapy (2020)
-
Lysosomal peptidases in innate immune cells: implications for cancer immunity
Cancer Immunology, Immunotherapy (2020)
-
Recombinant human arginase I elicited immunosuppression in activated macrophages through inhibiting autophagy
Applied Microbiology and Biotechnology (2019)
-
The role of autophagy in asparaginase-induced immune suppression of macrophages
Cell Death & Disease (2017)