Abstract
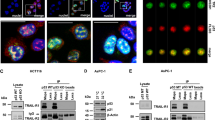
The p53-dependent RR small subunit (p53R2) protein, a newly identified member of the ribonucleotide reductase family, plays a key role in the p53-dependent cellular response to DNA. Several recent studies have suggested that p53R2 also plays an important role in suppressing the invasive potential of human cancer cells. However, the cellular mechanism that regulates invasiveness remains largely unknown. In this study, we show that p53R2 interacts with MEK2 (extracellular signal-regulated kinase (ERK) kinase 2–mitogen-activated protein kinase (MAPK) kinase 2), the molecule immediately upstream of ERK in the Ras–Raf–MAPK signaling cascade. In co-immunoprecipitation and immunofluorescence analyses, we found that p53R2 and MEK2 interact physically in cultured mammalian cells, and that the p53R2 segment comprising amino acids 161–206 is critical for this interaction. Moreover, serum-induced phosphorylation of MEK1/2 and ERK1/2 was greatly augmented in human cancer cells expressing small-interfering RNA against p53R2. On the other hand, phosphorylation of MEK1/2 and ERK1/2 in human cancer cells was markedly attenuated by overexpression of p53R2. Furthermore, MEK2 was required for p53R2 knockdown-induced enhancement of the invasive ability and anchorage-independent growth of human lung cancer H1299 cells. Taken together, these findings show that p53R2 negatively modulates serum-induced MEK–ERK activity and inhibits the MEK–ERK-mediated malignancy potential of human cancer cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- dNTP:
-
deoxyribonucleoside triphosphate
- ERK:
-
extracellular signal-regulated kinase
- MAPK:
-
mitogen-activated protein kinase
- MEK2:
-
ERK kinase 2–MAPK kinase 2
- p53R2:
-
p53-dependent RR small subunit
- ROS:
-
reactive oxygen species
- RR:
-
ribonucleotide reductase
References
Barnes DE, Lindahl T . (2004). Repair and genetic consequences of endogenous DNA base damage in mammalian cells. Annu Rev Genet 38: 445–476.
Cao MY, Lee Y, Feng NP, Xiong K, Jin H, Wang M et al. (2003). Adenovirus-mediated ribonucleotide reductase R1 gene therapy of human colon adenocarcinoma. Clin Cancer Res 9: 4553–4561.
Chabes AL, Pfleger CM, Kirschner MW, Thelander L . (2003). Mouse ribonucleotide reductase R2 protein: a new target for anaphase-promoting complex-Cdh1-mediated proteolysis. Proc Natl Acad Sci USA 100: 3925–3929.
Chen M, Rahman L, Voeller D, Kastanos E, Yang SX, Feigenbaum L et al. (2007). Transgenic expression of human thymidylate synthase accelerates the development of hyperplasia and tumors in the endocrine pancreas. Oncogene 26: 4817–4824.
Devlin HL, Mack PC, Burich RA, Gumerlock PH, Kung HJ, Mudryj M et al. (2008). Impairment of the DNA repair and growth arrest pathways by p53R2 silencing enhances DNA damage-induced apoptosis in a p53-dependent manner in prostate cancer cells. Mol Cancer Res 6: 808–818.
Dhillon AS, Hagan S, Rath O, Kolch W . (2007). MAP kinase signalling pathways in cancer. Oncogene 26: 3279–3290.
Droge W . (2002). Free radicals in the physiological control of cell function. Physiol Rev 82: 47–95.
Enslen H, Davis RJ . (2001). Regulation of MAP kinases by docking domains. Biol Cell 93: 5–14.
Fan H, Huang A, Villegas C, Wright JA . (1997). The R1 component of mammalian ribonucleotide reductase has malignancy-suppressing activity as demonstrated by gene transfer experiments. Proc Natl Acad Sci USA 94: 13181–13186.
Fan H, Villegas C, Huang A, Wright JA . (1998). The mammalian ribonucleotide reductase R2 component cooperates with a variety of oncogenes in mechanisms of cellular transformation. Cancer Res 58: 1650–1653.
Fan H, Villegas C, Wright JA . (1996). Ribonucleotide reductase R2 component is a novel malignancy determinant that cooperates with activated oncogenes to determine transformation and malignant potential. Proc Natl Acad Sci USA 93: 14036–14040.
Gautam A, Bepler G . (2006). Suppression of lung tumor formation by the regulatory subunit of ribonucleotide reductase. Cancer Res 66: 6497–6502.
Gautam A, Li ZR, Bepler G . (2003). RRM1-induced metastasis suppression through PTEN-regulated pathways. Oncogene 22: 2135–2142.
Hakansson P, Hofer A, Thelander L . (2006). Regulation of mammalian ribonucleotide reduction and dNTP pools after DNA damage and in resting cells. J Biol Chem 281: 7834–7841.
Kimura T, Takeda S, Sagiya Y, Gotoh M, Nakamura Y, Arakawa H . (2003). Impaired function of p53R2 in Rrm2b-null mice causes severe renal failure through attenuation of dNTP pools. Nat Genet 34: 440–445.
Kolch W . (2000). Meaningful relationships: the regulation of the Ras/Raf/MEK/ERK pathway by protein interactions. Biochem J 351 (Pt 2): 289–305.
Liu X, Zhou B, Xue L, Shih J, Tye K, Lin W et al. (2006). Metastasis-suppressing potential of ribonucleotide reductase small subunit p53R2 in human cancer cells. Clin Cancer Res 12: 6337–6344.
Liu X, Zhou B, Xue L, Yen F, Chu P, Un F et al. (2007). Ribonucleotide reductase subunits M2 and p53R2 are potential biomarkers for metastasis of colon cancer. Clin Colorectal Cancer 6: 374–381.
Mathews CK . (2006). DNA precursor metabolism and genomic stability. FASEB J 20: 1300–1314.
McCubrey JA, Steelman LS, Chappell WH, Abrams SL, Wong EW, Chang F et al. (2007). Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochim Biophys Acta 1773: 1263–1284.
Nakano K, Balint E, Ashcroft M, Vousden KH . (2000). A ribonucleotide reductase gene is a transcriptional target of p53 and p73. Oncogene 19: 4283–4289.
Nordlund P, Reichard P . (2006). Ribonucleotide reductases. Annu Rev Biochem 75: 681–706.
Powell DR, Desai U, Sparks MJ, Hansen G, Gay J, Schrick J et al. (2005). Rapid development of glomerular injury and renal failure in mice lacking p53R2. Pediatr Nephrol 20: 432–440.
Rahman L, Voeller D, Rahman M, Lipkowitz S, Allegra C, Barrett JC et al. (2004). Thymidylate synthase as an oncogene: a novel role for an essential DNA synthesis enzyme. Cancer Cell 5: 341–351.
Reddy KB, Nabha SM, Atanaskova N . (2003). Role of MAP kinase in tumor progression and invasion. Cancer Metastasis Rev 22: 395–403.
Tanaka H, Arakawa H, Yamaguchi T, Shiraishi K, Fukuda S, Matsui K et al. (2000). A ribonucleotide reductase gene involved in a p53-dependent cell-cycle checkpoint for DNA damage. Nature 404: 42–49.
Tsai MH, Chen X, Chandramouli GV, Chen Y, Yan H, Zhao S et al. (2006). Transcriptional responses to ionizing radiation reveal that p53R2 protects against radiation-induced mutagenesis in human lymphoblastoid cells. Oncogene 25: 622–632.
Widmann C, Gibson S, Jarpe MB, Johnson GL . (1999). Mitogen-activated protein kinase: conservation of a three-kinase module from yeast to human. Physiol Rev 79: 143–180.
Xu X, Page JL, Surtees JA, Liu H, Lagedrost S, Lu Y et al. (2008). Broad overexpression of ribonucleotide reductase genes in mice specifically induces lung neoplasms. Cancer Res 68: 2652–2660.
Xue L, Zhou B, Liu X, Heung Y, Chau J, Chu E et al. (2007). Ribonucleotide reductase small subunit p53R2 facilitates p21 induction of G1 arrest under UV irradiation. Cancer Res 67: 16–21.
Xue L, Zhou B, Liu X, Qiu W, Jin Z, Yen Y . (2003). Wild-type p53 regulates human ribonucleotide reductase by protein-protein interaction with p53R2 as well as hRRM2 subunits. Cancer Res 63: 980–986.
Xue L, Zhou B, Liu X, Wang T, Shih J, Qi C et al. (2006). Structurally dependent redox property of ribonucleotide reductase subunit p53R2. Cancer Res 66: 1900–1905.
Yamaguchi T, Matsuda K, Sagiya Y, Iwadate M, Fujino MA, Nakamura Y et al. (2001). p53R2-dependent pathway for DNA synthesis in a p53-regulated cell cycle checkpoint. Cancer Res 61: 8256–8262.
Yoon S, Seger R . (2006). The extracellular signal-regulated kinase: multiple substrates regulate diverse cellular functions. Growth Factors 24: 21–44.
Yoshida T, Haga S, Numata Y, Yamashita K, Mikami T, Ogawa T et al. (2006). Disruption of the p53-p53r2 DNA repair system in ulcerative colitis contributes to colon tumorigenesis. Int J Cancer 118: 1395–1403.
Zheng Z, Chen T, Li X, Haura E, Sharma A, Bepler G . (2007). DNA synthesis and repair genes RRM1 and ERCC1 in lung cancer. N Engl J Med 356: 800–808.
Zhou BS, Tsai P, Ker R, Tsai J, Ho R, Yu J et al. (1998). Overexpression of transfected human ribonucleotide reductase M2 subunit in human cancer cells enhances their invasive potential. Clin Exp Metastasis 16: 43–49.
Acknowledgements
This research was supported by a Grant [M 1063901] [M 20706000032] from the Ministry of Science and Technology, the Korean Government and funds from the Chosun University, 2002.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piao, C., Jin, M., Kim, H. et al. Ribonucleotide reductase small subunit p53R2 suppresses MEK–ERK activity by binding to ERK kinase 2. Oncogene 28, 2173–2184 (2009). https://doi.org/10.1038/onc.2009.84
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.84
Keywords
This article is cited by
-
PYCR1 and PYCR2 Interact and Collaborate with RRM2B to Protect Cells from Overt Oxidative Stress
Scientific Reports (2016)
-
Non-enzymatic action of RRM1 protein upregulates PTEN leading to inhibition of colorectal cancer metastasis
Tumor Biology (2015)
-
E2F1 regulates p53R2 gene expression in p53-deficient cells
Molecular and Cellular Biochemistry (2015)