Abstract
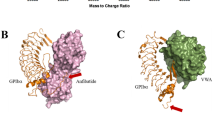
Botrocetin is a snake venom protein that enhances the affinity of the A1 domain of plasma von Willebrand factor (vWF) for the platelet receptor glycoprotein Ibα (GPIbα), an event that contributes to bleeding and host death. Here we describe a kinetic and crystallographic analysis of this interaction that reveals a novel mechanism of affinity enhancement. Using high-temporal-resolution microscopy, we show that botrocetin decreases the GPIbα off-rate two-fold in both human and mouse complexes without affecting the on-rate. The key to this behavior is that, upon binding of GPIbα to vWF-A1, botrocetin prebound to vWF-A1 makes no contacts initially with GPIbα, but subsequently slides around the A1 surface to form a new interface. This two-step mechanism and flexible coupling may prevent adverse alterations in on-rate of GPIbα for vWF-A1, and permit adaptation to structural differences in GPIbα and vWF in several prey species.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Savage, B., Almus-Jacobs, F. & Ruggeri, Z.M. Specific synergy of multiple substrate-receptor interactions in platelet thrombus formation under flow. Cell 94, 657–666 (1998).
Doggett, T.A. et al. Selectin-like kinetics and biomechanics promote rapid platelet adhesion in flow: the GPIbα-vWF tether bond. Biophys. J. 83, 194–205 (2002).
Kumar, R.A. et al. Kinetics of GPIbα–vWF-A1 tether bond under flow: effect of GPIbα mutations on the association and dissociation rates. Biophys. J. 85, 4099–4109 (2003).
Read, M.S., Shermer, R.W. & Brinkhous, K.M. Venom coagglutinin: an activator of platelet aggregation dependent on von Willebrand factor. Proc. Natl. Acad. Sci. USA 75, 4514–4518 (1978).
Brinkhous, K.M., Read, M.S., Fricke, W.A. & Wagner, R.H. Botrocetin (venom coagglutinin): reaction with a broad spectrum of multimeric forms of factor VIII macromolecular complex. Proc. Natl. Acad. Sci. USA 80, 1463–1466 (1983).
Sanders, W.E., Read, M.S., Reddick, R.L., Garris, J.B. & Brinkhous, K.M. Thrombotic thrombocytopenia with von Willebrand factor deficiency induced by botrocetin. An animal model. Lab. Invest. 59, 443–452 (1988).
Fujimura, Y., Holland, L.Z., Ruggeri, Z.M. & Zimmerman, T.S. The von Willebrand factor domain-mediating botrocetin-induced binding to glycoprotein Ib lies between Val449 and Lys728. Blood 70, 985–988 (1987).
Andrews, R.K., Booth, W.J., Gorman, J.J., Castaldi, P.A. & Berndt, M.C. Purification of botrocetin from Bothrops jararaca venom. Analysis of the botrocetin-mediated interaction between von Willebrand factor and the human platelet membrane glycoprotein Ib–IX complex. Biochemistry 28, 8317–8326 (1989).
Rabinowitz, I. et al. von Willebrand disease type B: a missense mutation selectively abolishes ristocetin-induced von Willebrand factor binding to platelet glycoprotein Ib. Proc. Natl. Acad. Sci. USA 89, 9846–9849 (1992).
Ajzenberg, N., Ribba, A.-S., Rastegar-Lari, G., Meyer, D. & Baruch, D. Effect of recombinant von Willebrand factor reproducing type 2B or type 2M mutations on shear-induced platelet aggregation. Blood 95, 3796–3803 (2000).
Fukuda, K. et al. Structural basis of von Willebrand factor activation by the snake toxin botrocetin. Structure 10, 943–950 (2002).
Huizinga, E.G. et al. Structures of glycoprotein Ibα and its complex with von Willebrand factor A1 domain. Science 297, 1176–1179 (2002).
Jenkins, P.V., Pasi, K.J. & Perkins, S.J. Molecular modeling of ligand and mutation sites of the type A domains of human von Willebrand factor and their relevance to von Willebrand's disease. Blood 91, 2032–2044 (1998).
Miura, S. et al. Interaction of von Willebrand factor A1 with platelet glycoprotein Ibα (1–289). Slow intrinsic binding kinetics mediate rapid platelet adhesion. J. Biol. Chem. 275, 7539–7546 (2000).
Emsley, J., Cruz, M., Handin, R. & Liddington, R. Crystal structure of the von Willebrand factor A1 domain and implications for the binding of platelet glycoprotein Ib. J. Biol. Chem. 273, 10396–10401 (1998).
Dumas, J.J. et al. Crystal structure of the wild-type von Willebrand factor A1–glycoprotein Ibα complex reveals conformational differences with a complex bearing von Willebrand disease mutations. J. Biol. Chem. 279, 23327–23334 (2004).
Lo Conte, L., Chothia, C. & Janin, J. The atomic structure of protein-protein recognition sites. J. Mol. Biol. 285, 2177–2198 (1999).
López, J.A. et al. Cloning of the α chain of human platelet glycoprotein Ib: a transmembrane protein with homology to leucine-rich α2-glycoprotein. Proc. Natl. Acad. Sci. USA 84, 5615–5619 (1987).
Ware, J., Russell, S. & Ruggeri, Z.M. Cloning of the murine platelet glycoprotein Ibα gene highlighting species-specific platelet adhesion. Blood Cells Mol. Dis. 23, 292–301 (1997).
Kenny, D., Morateck, P.A., Fahs, S.A., Warltier, D.C. & Montgomery, R.R. Cloning and expression of canine glycoprotein Ibα. Thromb. Haemost. 82, 1327–1333 (1999).
Marchese, P. et al. Identification of three tyrosine residues of glycoprotein Ibα with distinct roles in von Willebrand factor and α-thrombin binding. J. Biol. Chem. 270, 9571–9578 (1995).
Dong, J.-f. et al. Tyrosine sulfation of glycoprotein Ibα. Role of electrostatic interactions in von Willebrand factor binding. J. Biol. Chem. 276, 16690–16694 (2001).
Uff, S., Clemetson, J.M., Harrison, T., Clemetson, K.J. & Emsley, J. Crystal structure of the platelet glycoprotein Ibα N-terminal domain reveals an unmasking mechanism for receptor activation. J. Biol. Chem. 277, 35657–35663 (2002).
Doggett, T. et al. Alterations in the intrinsic properties of the GPIbα-VWF tether bond define the kinetics of the platelet-type von Willebrand disease mutation, Gly233Val. Blood 102, 152–160 (2003).
Chang, K.-C. & Hammer, D.A. The forward rate of binding of surface-tethered reactants: effect of relative motion between two surfaces. Biophys. J. 76, 1280–1292 (1999).
Chen, S. & Springer, T.A. Selectin receptor-ligand bonds: formation limited by shear rate and dissociation governed by the Bell model. Proc. Natl. Acad. Sci. USA 98, 950–955 (2001).
Alon, R., Hammer, D.A. & Springer, T.A. Lifetime of the P-selectin-carbohydrate bond and its response to tensile force in hydrodynamic flow. Nature 374, 539–542 (1995).
Ramachandran, V. et al. Tyrosine replacement in P-selectin glycoprotein ligand-1 affects distinct kinetic and mechanical properties of bonds with P- and L-selectin. Proc. Natl. Acad. Sci. USA 96, 13771–13776 (1999).
Smith, M.J., Berg, E.L. & Lawrence, M.B. A direct comparison of selectin-mediated transient adhesive events using high temporal resolution. Biophys. J. 77, 3371–3383 (1999).
Bell, G.I. Models for the specific adhesion of cells to cells. Science 200, 618–627 (1978).
Bonnefoy, A. et al. Shielding the front-strand β3 of the von Willebrand factor A1 domain inhibits its binding to platelet glycoprotein Ibα. Blood 101, 1375–1383 (2003).
Cestele, S. & Catterall, W.A. Molecular mechanisms of neurotoxin action on voltage-gated sodium channels. Biochimie 82, 883–892 (2000).
Lerm, M., Schmidt, G. & Aktories, K. Bacterial protein toxins targeting rho GTPases. FEMS Microbiol. Lett. 188, 1–6 (2000).
Matsui, K., Boniface, J.J., Steffner, P., Reay, P.A. & Davis, M.M. Kinetics of T-cell receptor binding to peptide/I-Ek complexes: correlation of the dissociation rate with T-cell responsiveness. Proc. Natl. Acad. Sci. USA 91, 12862–12866 (1994).
Dwir, O. et al. Avidity enhancement of L-selectin bonds by flow: shear-promoted rotation of leukocytes turn labile bonds into functional tethers. J. Cell Biol. 163, 649–659 (2003).
Chen, J., Diacovo, T.G., Grenache, D.G., Santoro, S.A. & Zutter, M.M. The α2 integrin subunit-deficient mouse: a multifaceted phenotype including defects of branching morphogenesis and hemostasis. Am. J. Pathol. 161, 337–344 (2002).
Kahn, M.L. et al. Glycoprotein V-deficient platelets have undiminished thrombin responsiveness and do not exhibit a Bernard-Soulier phenotype. Blood 94, 4112–4121 (1999).
Fujimura, Y. et al. Isolation and chemical characterization of two structurally and functionally distinct forms of botrocetin, the platelet coagglutinin isolated from the venom of Bothrops jararaca. Biochemistry 30, 1957–1964 (1991).
Sekiya, F., Atoda, H. & Morita, T. Isolation and characterization of an anticoagulant protein homologous to botrocetin from the venom of Bothrops jararaca. Biochemistry 32, 6892–6897 (1993).
Hammer, D.A. & Apte, S.M. Simulation of cell rolling and adhesion on surfaces in shear flow: general results and analysis of selectin-mediated neutrophil adhesion. Biophys. J. 63, 35–57 (1992).
Gillespie, D.T. A general method for numerically simulating the stochastic time evolution of coupled chemical reactions. J. Comput. Phys. 22, 403–434 (1976).
Afshar-Kharghan, V., Li, C.Q., Khoshnevis-Asl, M. & López, J.A. Kozak sequence polymorphism of the glycoprotein (GP) Ibα gene is a major determinant of the plasma membrane levels of the platelet GP Ib–IX–V complex. Blood 94, 186–191 (1999).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–327 (1997).
Navaza, J. AMoRe: an automated package for molecular replacement. Acta Crystallogr. A 50, 157–163 (1994).
Sen, U. et al. Crystal structure of the von Willebrand factor modulator botrocetin. Biochemistry 40, 345–352 (2001).
Roussel, A. & Cambileau, C. TURBO-FRODO. In Silicon Graphics Geometry (Silicon Graphics, Mountain View, California, USA, 1991).
Brünger, A.T. et al. Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998).
Laskowski, R.A., MacArthur, N.W., Moss, D.S. & Thornton, J.M. PROCHECK: a program to check stereochemical quality of protein structures. J. Appl. Crystallogr. 26, 283–291 (1993).
Lawrence, M.C. & Colman, P.M. Shape complementarity at protein/protein interfaces. J. Mol. Biol. 234, 946–950 (1993).
Kraulis, P.J. MOLSCRIPT: a program to produce both detailed and schematic plots of protein structures. J. Appl. Crystallogr. 24, 946–950 (1991).
Merritt, E.A. & Bacon, D.J. Raster3D: photorealistic molecular graphics. Methods Enzymol. 277, 505–524 (1997).
Nicholls, A., Sharp, K.A. & Honig, B. Protein folding and association: insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins 11, 281–296 (1991).
Barton, G.J. ALSCRIPT: a tool to format multiple sequence alignments. Protein Eng. 6, 37–40 (1993).
Acknowledgements
We thank to the staff of the Stanford Synchrotron Radiation Laboratory and the US Department of Energy for the use of X-ray data collection facilities. This work was supported by grants from the US National Institutes of Health (to R.C.L and T.G.D), the American Heart Association (T.G.D.) and a postdoctoral fellowship from the PhRMA Foundation (I.J.L.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Intrinsic off-rate for the botrocetin–vWF-A1–GPIb tether bond. (PDF 184 kb)
Supplementary Fig. 2
The effect of botrocetin on the kinetics of the GPIbα–vWF-A1 tether bond. (PDF 330 kb)
Supplementary Fig. 3
The botrocetin-GPIbα interface regulates the off-rate of the ternary complex. (PDF 80 kb)
Rights and permissions
About this article
Cite this article
Fukuda, K., Doggett, T., Laurenzi, I. et al. The snake venom protein botrocetin acts as a biological brace to promote dysfunctional platelet aggregation. Nat Struct Mol Biol 12, 152–159 (2005). https://doi.org/10.1038/nsmb892
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb892
This article is cited by
-
Biomechanical activation of blood platelets via adhesion to von Willebrand factor studied with mesoscopic simulations
Biomechanics and Modeling in Mechanobiology (2023)
-
Staphylococcus aureus iron-regulated surface determinant B (IsdB) protein interacts with von Willebrand factor and promotes adherence to endothelial cells
Scientific Reports (2021)
-
Vwf K1362A resulted in failure of protein synthesis in mice
International Journal of Hematology (2018)
-
Platelet clearance via shear-induced unfolding of a membrane mechanoreceptor
Nature Communications (2016)
-
Conformational Transition of Glycoprotein Ibα Mutants in Flow Molecular Dynamics Simulation
Cellular and Molecular Bioengineering (2011)