Abstract
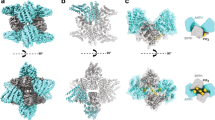
During the assembly of clathrin-coated vesicles, many peripheral membrane proteins, including the amphiphysins, use LLDLD-type clathrin-box motifs to interact with the N-terminal β-propeller domain (TD) of clathrin. The 2.3 Å–resolution structure of the clathrin TD in complex with a TLPWDLWTT peptide from amphiphysin 1 delineates a second clathrin-binding motif, PWXXW (the W box), that binds at a site on the TD remote from the clathrin box–binding site. The presence of both sequence motifs within the unstructured region of the amphiphysins allows them to bind more tightly to free TDs than do other endocytic proteins that contain only clathrin-box motifs. This property, along with the propensity of the N-terminal BAR domain to bind curved membranes, will preferentially localize amphiphysin and its partner, dynamin, to the periphery of invaginated clathrin lattices.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Brodsky, F.M., Chen, C.Y., Knuehl, C., Towler, M.C. & Wakeham, D.E. Biological basket weaving: formation and function of clathrin-coated vesicles. Annu. Rev. Cell Dev. Biol. 17, 517–568 (2001).
Conner, S.D. & Schmid, S.L. Regulated portals of entry into the cell. Nature 422, 37–44 (2003).
Robinson, M.S. & Bonifacino, J.S. Adaptor-related proteins. Curr. Opin. Cell Biol. 13, 444–453 (2001).
Bonifacino, J.S. & Traub, L.M. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 72, 395–447 (2003).
Marks, B. et al. GTPase activity of dynamin and resulting conformation change are essential for endocytosis. Nature 410, 231–235 (2001).
McMahon, H.T. Endocytosis: an assembly protein for clathrin cages. Curr. Biol. 9, R332–R335 (1999).
Slepnev, V.I. & De Camilli, P. Accessory factors in clathrin-dependent synaptic vesicle endocytosis. Nat. Rev. Neurosci. 1, 161–172 (2000).
Lafer, E.M. Clathrin-protein interactions. Traffic 3, 513–520 (2002).
Wendland, B. Epsins: adaptors in endocytosis? Nat. Rev. Mol. Cell Biol. 3, 971–977 (2002).
Korolchuk, V. & Banting, G. Kinases in clathrin-mediated endocytosis. Biochem. Soc. Trans. 31, 857–860 (2003).
Traub, L.M. Sorting it out: AP-2 and alternate clathrin adaptors in endocytic cargo selection. J. Cell Biol. 163, 203–208 (2003).
Evans, P.R. & Owen, D.J. Endocytosis and vesicle trafficking. Curr. Opin. Struct. Biol. 12, 814–821 (2002).
Kirchhausen, T. Clathrin. Annu. Rev. Biochem. 69, 699–727 (2000).
Ybe, J.A. et al. Clathrin self-assembly is mediated by a tandemly repeated superhelix. Nature 399, 371–375 (1999).
ter Haar, E., Musacchio, A., Harrison, S.C. & Kirchhausen, T. Atomic structure of clathrin: a β propeller terminal domain joins an a zigzag linker. Cell 95, 563–573 (1998).
Goodman, O.B. Jr., Krupnick, J.G., Gurevich, V.V., Benovic, J.L. & Keen, J.H. Arrestin/clathrin interaction. Localization of the arrestin binding locus to the clathrin terminal domain. J. Biol. Chem. 272, 15017–15022 (1997).
ter Haar, E., Harrison, S.C. & Kirchhausen, T. Peptide-in-groove interactions link target proteins to the β-propeller of clathrin. Proc. Natl. Acad. Sci. USA 97, 1096–1100 (2000).
Ramjaun, A.R. & McPherson, P.S. Multiple amphiphysin II splice variants display differential clathrin binding: identification of two distinct clathrin-binding sites. J. Neurochem. 70, 2369–2376 (1998).
Slepnev, V.I., Ochoa, G.C., Butler, M.H. & De Camilli, P. Tandem arrangement of the clathrin and AP-2 binding domains in amphiphysin 1, and disruption of clathrin coat function mediated by amphiphysin fragments comprising these sites. J. Biol. Chem. 275, 17583–17589 (2000).
Drake, M.T. & Traub, L.M. Interaction of two structurally-distinct sequence types with the clathrin terminal domain β-propeller. J. Biol. Chem. 276, 28700–28709 (2001).
Owen, D.J. & Evans, P.R. A structural explanation for the recognition of tyrosine-based endocytotic signals. Science 282, 1327–1332 (1998).
Collins, B.M., Praefcke, G.J., Robinson, M.S. & Owen, D.J. Structural basis for binding of accessory proteins by the appendage domain of GGAs. Nat. Struct. Biol. 10, 607–613 (2003).
Miller, G.J., Mattera, R., Bonifacino, J.S. & Hurley, J.H. Recognition of accessory protein motifs by the γ-adaptin ear domain of GGA3. Nat. Struct. Biol. 10, 599–606 (2003).
Smith, T.F., Gaitatzes, C., Saxena, K. & Neer, E.J. The WD repeat: a common architecture for diverse functions. Trends Biochem. Sci. 24, 181–185 (1999).
Orlicky, S., Tang, X., Willems, A., Tyers, M. & Sicheri, F. Structural basis for phosphodependent substrate selection and orientation by the SCFCdc4 ubiquitin ligase. Cell 112, 243–256 (2003).
Scheele, U. et al. Molecular and functional characterization of clathrin and AP-2 binding determinants within a disordered domain of auxilin. J. Biol. Chem. 278, 25357–25368 (2003).
Lichte, B., Veh, R.W., Meyer, H.E. & Kilimann, M.W. Amphiphysin, a novel protein associated with synaptic vesicles. EMBO J. 11, 2521–2530 (1992).
De Camilli, P. et al. The synaptic vesicle-associated protein amphiphysin is the 128-kD autoantigen of Stiff-Man syndrome with breast cancer. J. Exp. Med. 178, 2219–2223 (1993).
Sakamuro, D., Elliott, K.J., Wechsler-Reya, R. & Prendergast, G.C. BIN1 is a novel MYC-interacting protein with features of a tumour suppressor. Nat. Genet. 14, 69–77 (1996).
Leprince, C. et al. A new member of the amphiphysin family connecting endocytosis and signal transduction pathways. J. Biol. Chem. 272, 15101–15105 (1997).
Ramjaun, A.R., Micheva, K.D., Bouchelet, I. & McPherson, P.S. Identification and characterization of a nerve terminal-enriched amphiphysin isoform. J. Biol. Chem. 272, 16700–16706 (1997).
Zhang, B. & Zelhof, A.C. Amphiphysins: raising the BAR for synaptic vesicle recycling and membrane dynamics. Traffic 3, 452–460 (2002).
Ramjaun, A.R., Philie, J., de Heuvel, E. & McPherson, P.S. The N terminus of amphiphysin II mediates dimerization and plasma membrane targeting. J. Biol. Chem. 274, 19785–19791 (1999).
Takei, K., Slepnev, V.I., Haucke, V. & De Camilli, P. Functional partnership between amphiphysin and dynamin in clathrin-mediated endocytosis. Nat. Cell Biol. 1, 33–39 (1999).
Razzaq, A. et al. Amphiphysin is necessary for organization of the excitation-contraction coupling machinery of muscles, but not for synaptic vesicle endocytosis in Drosophila. Genes Dev. 15, 2967–2979 (2001).
Peter, B.J. et al. BAR domains as sensors of membrane curvature: the amphiphysin BAR structure. Science 303, 495–499 (2004).
David, C., McPherson, P.S., Mundigl, O. & De Camilli, P. A role of amphiphysin in synaptic vesicle endocytosis suggested by its binding to dynamin in nerve terminals. Proc. Natl. Acad. Sci. USA 93, 331–335 (1996).
Owen, D.J. et al. Crystal structure of the amphiphysin-2 SH3 domain and its role in the prevention of dynamin ring formation. EMBO J. 17, 5273–5285 (1998).
Howard, L., Nelson, K.K., Maciewicz, R.A. & Blobel, C.P. Interaction of the metalloprotease disintegrins MDC9 and MDC15 with two SH3 domain-containing proteins, endophilin I and SH3PX1. J. Biol. Chem. 274, 31693–31699 (1999).
Lundmark, R. & Carlsson, S.R. The β-appendages of the four adaptor-protein (AP) complexes: structure and binding properties, and identification of sorting nexin 9 as an accessory protein to AP-2. Biochem. J. 362, 597–607 (2002).
Macaulay, S.L. et al. Insulin stimulates movement of sorting nexin 9 between cellular compartments-A putative role mediating cell surface receptor expression and insulin action. Biochem. J., 376, 123–134 (2003).
Lundmark, R. & Carlsson, S.R. Sorting nexin 9 participates in clathrin-mediated endocytosis through interactions with the core components. J. Biol. Chem. 278, 46772–46781 (2003).
Dunker, A.K., Brown, C.J., Lawson, J.D., Iakoucheva, L.M. & Obradovic, Z. Intrinsic disorder and protein function. Biochemistry 41, 6573–6582 (2002).
Owen, D.J. et al. A structural explanation for the binding of multiple ligands by the α-adaptin appendage domain. Cell 97, 805–815 (1999).
Leslie, A.G.W. Recent changes to MOSFLM package for processing film and image plate data. Joint CCP4 and ESF-EACMB Newsletter on Protein Crystallography Vol. 26 (Daresbury Laboratory, Warrington, UK, 1992).
Collaborative Computational Project, the CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeldgaard, M. Improved methods for binding protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997).
Segel, I.H. Biochemical Calculations 2nd edn, 150–159 (Wiley, New York, 1976).
Mishra, S.K. et al. Disabled-2 exhibits the properties of a cargo-selective endocytic clathrin adaptor. EMBO J. 21, 4915–4926 (2002).
Acknowledgements
We thank the staff of Daresbury Synchrotron Radiation Source and M. Ford for help in data collection, and the Wellcome Trust for a traveling research fellowship to A.E.M. and for a senior research fellowship in basic biomedical sciences to D.J.O. L.M.T. was supported in part by US National Institutes of Health grant R01 DK53249.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Miele, A., Watson, P., Evans, P. et al. Two distinct interaction motifs in amphiphysin bind two independent sites on the clathrin terminal domain β-propeller. Nat Struct Mol Biol 11, 242–248 (2004). https://doi.org/10.1038/nsmb736
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb736
This article is cited by
-
Identification of a novel interaction site between the large hepatitis delta antigen and clathrin that regulates the assembly of genotype III hepatitis delta virus
Virology Journal (2022)
-
Integrating protein copy numbers with interaction networks to quantify stoichiometry in clathrin-mediated endocytosis
Scientific Reports (2022)
-
Cryo-EM of multiple cage architectures reveals a universal mode of clathrin self-assembly
Nature Structural & Molecular Biology (2019)
-
Motif co-regulation and co-operativity are common mechanisms in transcriptional, post-transcriptional and post-translational regulation
Cell Communication and Signaling (2015)
-
Clathrin expression in Trypanosoma cruzi
BMC Cell Biology (2014)