Abstract
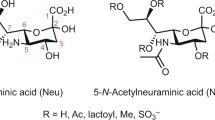
Sialic acid is the most abundant terminal monosaccharide on mammalian cell surface glycoconjugates. The crystal structures of a mammalian sialyltransferase, that of porcine ST3Gal-I, in the apo form and bound to analogues of the donor and acceptor substrate are now described, providing insights into the catalytic mechanism and for inhibitor design.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Harduin-Lepers, A. et al. Biochimie 83, 727–737 (2001).
Tsuji, S., Datta, A.K. & Paulson, J.C. Glycobiology 6, 647 (1996).
Cantarel, B.L. et al. Nucleic Acids Res. 37, D233–D238 (2009).
Paulson, J.C. & Colley, K.J. J. Biol. Chem. 264, 17615–17618 (1989).
Hennet, T., Chui, D., Paulson, J.C. & Marth, J.D. Proc. Natl. Acad. Sci. USA 95, 4504–4509 (1998).
Martin, L.T., Marth, J.D., Varki, A. & Varki, N.M. J. Biol. Chem. 277, 32930–32938 (2002).
Galuska, S.P. et al. J. Biol. Chem. 281, 31605–31615 (2006).
Burchell, J.M., Mungul, A. & Taylor-Papadimitriou, J. J. Mammary Gland Biol. Neoplasia 6, 355–364 (2001).
Datta, A.K., Sinha, A. & Paulson, J.C. J. Biol. Chem. 273, 9608–9614 (1998).
Datta, A.K. & Paulson, J.C. J. Biol. Chem. 270, 1497–1500 (1995).
Datta, A.K., Chammas, R. & Paulson, J.C. J. Biol. Chem. 276, 15200–15207 (2001).
Geremia, R.A., Harduin-Lepers, A. & Delannoy, P. Glycobiology 7, 161 (1997).
Jeanneau, C. et al. J. Biol. Chem. 279, 13461–13468 (2004).
Vallejo-Ruiz, V. et al. Biochim. Biophys. Acta 1549, 161–173 (2001).
Kono, M. et al. Glycobiology 7, 469–479 (1997).
Rearick, J.I., Sadler, J.E., Paulson, J.C. & Hill, R.L. J. Biol. Chem. 254, 4444–4451 (1979).
Unligil, U.M. et al. EMBO J. 19, 5269–5280 (2000).
Chiu, C.P. et al. Nat. Struct. Mol. Biol. 11, 163–170 (2004).
Charnock, S.J. & Davies, G.J. Biochemistry 38, 6380–6385 (1999).
Lairson, L.L., Henrissat, B., Davies, G.J. & Withers, S.G. Annu. Rev. Biochem. 77, 521–555 (2008).
Holm, L., Ouzounis, C., Sander, C., Tuparev, G. & Vriend, G. Protein Sci. 1, 1691–1698 (1992).
Kitazume-Kawaguchi, S., Kabata, S. & Arita, M. J. Biol. Chem. 276, 15696–15703 (2001).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 805 kb)
Rights and permissions
About this article
Cite this article
Rao, F., Rich, J., Rakić, B. et al. Structural insight into mammalian sialyltransferases. Nat Struct Mol Biol 16, 1186–1188 (2009). https://doi.org/10.1038/nsmb.1685
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1685
This article is cited by
-
The vertebrate sialylation machinery: structure-function and molecular evolution of GT-29 sialyltransferases
Glycoconjugate Journal (2023)
-
A universal glycoenzyme biosynthesis pipeline that enables efficient cell-free remodeling of glycans
Nature Communications (2022)
-
Glycosyltransferases as targets for therapeutic intervention in cancer and inflammation: molecular modeling insights
Chemical Papers (2022)
-
Metabolic fate of dietary sialic acid and its influence on gut and oral bacteria
Systems Microbiology and Biomanufacturing (2022)
-
Insights into the role of sialylation in cancer progression and metastasis
British Journal of Cancer (2021)